135 patients (86 men,
49 women,
mean age : 64) treated with RASP between January 2007 and December 2011,
were retrospectively evaluated.
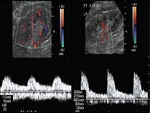
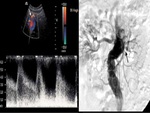
Diagnosis of ASRA was suggested by colour Doppler ultrasound (ECD)findings (Fig. 1 Fig. 2) and confirmed by CT or MRI (Fig. 3).
All patients had indications for RASP according to our inclusion criterions:
- renal artery stenosis > 70%
- resistant hypertension to a 3 drug regimen
- altered renal function
- stenosis < 70% in solitary functioning kidney patients with reduction of renal bipolar diameter
The exclusion criterions were :
- longitudinal diameter of the involved kidney < than 8 cm
- patients affected by stenosis > 70% without hypertension or altered renal function
- alterations at preliminary coagulation test
Before each procedure we evaluated the risk of iodate contrast nephropathy (CIN) based on the values of renal glomerular filtration and creatinaemia; in patients with risk of CIN we applied the guide lines of our Hospital’s protocol which provides:
- the suspension,
the day before the procedure,
of these drugs categories: Non Steroidal Anti-Inflammatory Drugs,
ACE inhibitors,
Angiotensin Receptor Blockers (ARBs),
nephrotoxic antibiotics,
metformin.
- the suspension of anticoagulant/antiaggregant therapies
- hydration with isotonic sodium chloride solution (0,9%),
sodium bicarbonate (1,4 %) and acetylcysteine.
All the RASPs were performed in the angiography suite by an experienced interventional radiologist.
Ballon-expandable stents (Palmaz Blue peripheral 0,14 stent system) were used and recanalisation was usually performed after a percutaneous puncture of the femoral artery (trans-brachial approach was performed only in particular conditions: tortuosity of the abdominal aorta,
severe angle at the origin of renal artery from the aorta,
stenosis of the iliac arteries).
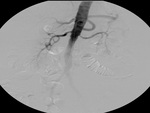
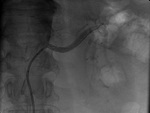
Aortography was performed through a pig tail catheter in order to evaluate the anatomy of the renal arteries and the stenotic lesions (Fig. 4).
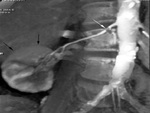
The occlusion were usually crossed using a 0,035-in hydrophilic guidewire supported by a 6F guiding catheter positioned at the renal ostium.
At this point the stent was introduced and an angiographic control was performed (Fig. 5) in order to evaluate its correct position (the markers of the stent have to cover completely the stenosis; furthermore at least 2 mm of the stent surface have to protrude into the aortic lumen).
The stent was finally implanted and the last angiogram was performed to evaluate the result (Fig. 6).
At the end of the procedure the artery puncture was sealed trough a vascular closure device.
All patients received 3,000 to 5,000 (according to the patient’s weight) UI heparin intra-arterially during the procedure.
Immediate technical success of the procedure (residual stenosis <30%) and procedure-related complications were evaluated in all 135 patients.
94/135 (69.6%) patients were studied by ECD after 12 months from RASP to define restenosis incidence (significant hemodynamic restenosis was defined as more than 50% diameter stenosis) .
Blood pressure and serum creatinine levels were measured before the procedures.
The same parameters were evaluated after 1 month,
12 months and 24 months in 74 patients (RASP performed between january 2007 and december 2010) and after 1 month and 12 months in 20 patients (RASP performed during 2011).
41 patients were not evaluated in long term follow up after RASP because they were not followed by the Nephrology Department of our Hospital anymore.
Data analysis was based on the two following statistical tests: ANOVA (for repeated measurements and variance analysis) and Bonferroni’s (for the analysis of multiple confrontations).