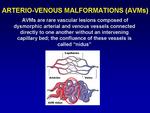
AVMs are rare vascular lesions (ranging from 0.3% to 0.5% of the population) representing errors in vascular development resulting in dysmorphic arterial and venous vessels connected directly to one another without an intervening capillary bed (Fig.
1).
They are present at birth (60%) or become so during the first few weeks of life (30%) with no difference between males and females.
Contrary to haemangioma,
they never regress and may grow during lifetime.
AVMs may develop during early fetal period,
because of the failure of regression of arterio-venous channels in the primitive retiform plexus.
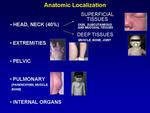
This theory explains the predominance of AVMs in the head and neck region,
since the early embryo is composed mainly of cephalic structures,
having the higher surface area-to volume ratio than other facial structures.
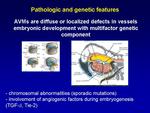
AVMs are frequently sporadic,
but they may be associated with underlying disease or systemic anomalies whose molecular genetics have been correlated with AVMs (e.g.
hereditary hemorrhagic telangiectasia).
Sporadic lesions may include a Transforming Growth Factor b (TGF-b) (it is involved in the induction of apoptotic endothelial cell death) and the tyrosine kinase receptor tunica internal endothelial cell kinase-2 (Tie-2) (it is essential for early vessel development and its increased activity can lead to abnormal growth of the primary vascular plexus) mutation (Fig.
2).
In descending order of frequency they also involve lower and upper limbs,
trunk,
and viscera (e.g.
liver,
chest,
pelvis) (Fig.
3).
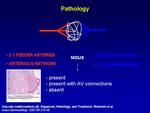
Histologically,
AVM is composed of multiple dysplastic feeder arteries and arterialized veins creating a vascular nidus without a capillary network.
Endothelium cultures show increased growth and reduced apoptosis,
suggesting an intrinsic cellular defect.
Generally,
their histological extension exceeds the visible extension,
with microscopic infiltration of the underlying tissue that favours relapse after partial removal (Fig.
4).
Identification and classification of vascular anomalies are very difficult and confused; the use of confusing nomenclature continues to persist in the Literature.
The accepted classification including AVMs is the International Society for the Study of Vascular Anomalies (ISSVA)/Mulliken’s classification modified in 1996.
Moreover,
vascular malformations can be subdivided on the basis of their vascular components and flow characteristics (slow-flow capillary,
venous,
or lymphatic channels,
fast-flow arterial channels,
or a combination of each) and from this point of view,
AVMs are considered fast-flow lesions.
They are also frequently classified by eponyms when they are components of syndromes (Fig.
5).
The classification is useful to recognize and to study the lesions; in fact AVMs are diagnosed by clinical findings (patient’s medical history and a physical examination) and radiologic features (as a complementary tool specially when there is doubt about the nature of the lesion and as an integral part of treatment).
Clinical presentation is extremely variable: asymptomatic lesions,
warmer and sometimes pulsatile macules,
ulcerated,
painful and bleeding lesions,
high blood flow lesions; hypertrophy of the bone underlying the lesion is also common.
They can be located in critical sites having systemic repercussions on diagnosis and treatment of these lesions.
In fact,
a proximal arterio-venous malformation with high blood flow may increase cardiac load and lead to congestive heart failure even if there is usually a compensation for years; if the malformation is distal,
there is a propensity to lower flow and peripheral ischemia (Fig.
6).
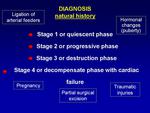
The natural history of AVMs can be divided into different stages based on Schobinger staging system (Fig.
7):
- Quiescent phase: asymptomatic lesions or pink-violaceous marks sometimes with a bruit or a thrill if there I a fast-flow component.
- Expansive phase: as in stage I,
but clinically pulsatile,
with tortuous vessels and tight turns,
sometimes invading deep structures.
- Destruction phase: as in stage II with dystrophic skin changes,
ulceration,
bleeding,
and continuous pain.
- Decompensate phase: similar to stage III,
with heart failure.
Stages progression seems to be favoured by hormonal changes (puberty),
pregnancy,
traumatic injuries creating a local ischemia,
ligation of arterial feeders and partial surgical excision.
Local or diffuse soft-tissue deformity,
dysmetria,
compression,
invasion and/or destruction of deep structures,
chronic venous deficiency with interstitial edema,
cutaneous gangrene can complicate all the lesions (Fig.
8).