Radiologic evaluation is often necessary to confirm the diagnosis,
delineate the extent of the lesions,
assess the flow characteristics and as a therapeutic option.
Plain radiography has today a limited value,
demonstrating bone involvement (asymmetric hypertrophy or atrophy,
osteoporosis,
or lytic lesions).
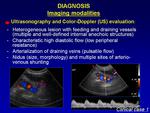
Ultrasonography (US) and colour Doppler evaluations are often performed initially because of their non-invasivity and accessibility and they not require ionizing radiation and a great deal of cooperation of the patient (e.g.
paediatric patients).
Even if they are operator-dependent techniques,
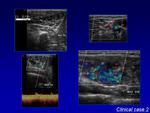
colour Doppler ultrasound is able to confirm vascular nature of the lesion assessing flow characteristics and visualizing multiple internal,
well-defined anechoic structures.
Colour Doppler analysis can also identify both feeding and draining vascular flow patterns.
Spectral waveforms of feeding arteries indicate low peripheral resistance and dilated draining veins show pulsatile flow,
suggesting the presence of direct AV communications without an intervening capillary bed.
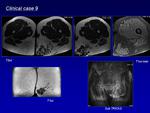
The nidus is characterized by a ‘‘mosaic’’ pattern with a mixture of red and blue colour patterns in the anechoic structures as well as coarse ‘‘rumbling’’ acoustic Doppler representation (Fig.
9,
10).
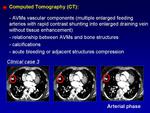
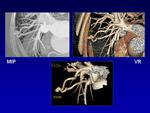
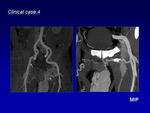
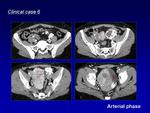
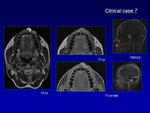
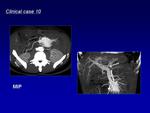
Computed tomography (CT) is important to visualize vascular components and to define the extent of the AVM.
AVMs demonstrate multiple enlarged feeding arteries with rapid contrast shunting into enlarged draining veins without intervening tissue enhancement.
Because of radiation,
CT ideally should be reserved for particular cases (acute bleeding or adjacent structure compression) (Fig.
11,
12,
13,
14,
15,
16,
25).
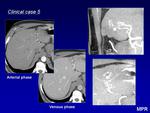
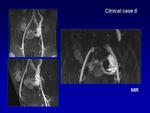
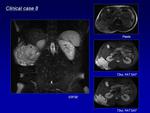
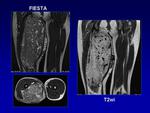
Magnetic Resonance (MR) is a non-invasive technique,
without ionizing radiation,
that provides both anatomic and hemodynamic data.
It is excellent for tissue differentiation and this,
together with its capacity to acquire images in multiple spatial planes,
makes MR the best radiologic technique for demonstrating anatomic relationships of the lesions considering the adjacent structures (muscle and fascial planes),
the involvement of bony structures,
and providing better detail in confined locations (orbit,
lip).
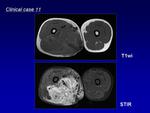
MR shows multiple hypertrophied arteries and dilated veins connected by linear or focal shunting seen as low signal on T1- and T2-weighted spin echo sequences and a characteristic lack of soft tissue component.
On T1- and T2-weighted sequences,
the presence of rapid or turbulent flow decreases the intensity of the signal (flow void phenomenon),
and when the flow is slow or thrombosis is present,
the intensity of the signal increases.
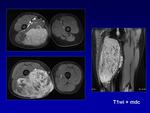
In addition,
gadolinium enhancement delineates feeding arteries and draining veins well.
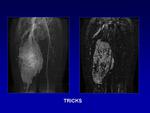
MR using phase-contrast (PC) and time of flight (TOF) techniques can successfully identify abnormal arteries and veins.
PC-MR is usually sufficient to identify a high flow lesion.
Dynamic contrast opacification of the lesion can be performed using time-resolved MR sequences (TRICKS),
but spatial resolution tends to be compromised (Fig.
17,
18,
19,
21,
23,
31,
32,
33,
34).
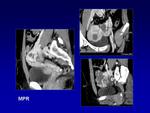
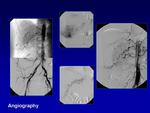
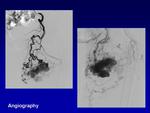
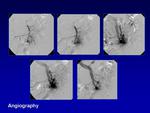
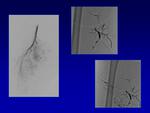
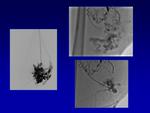
Angiography is necessary both to assess the extent of the lesion before therapeutic intervention and to guide intra-arterial embolization; it is specially recommended when MR examination is equivocal or if vascular intervention is considered.
It is rarely used for diagnostic purposes alone.
The classic angiographic appearance of AVMs demonstrates multiple hypertrophied feeding arteries rapidly shunting into engorged dilated draining veins via a nidus that is the point at which arterial structures first opacify the venous drainage.
No soft tissue enhancement is seen in AVMs,
unlike vascular tumours and haemangioma.
Direct arterio-venous fistulous components and intralesional aneurysms also may be identified (Fig.
20,
22,
24,
26,
35,
36).
Cho et al.
propose an angiographic classification of AVMs based on nidal morphology with implications for therapy and outcomes:
• Type I: arterio-venous fistulae
• Type II: arteriolo-venous fistulae
• Type III a: arteriolo-venulous fistulae with non dilated fistula
• Type III b: arteriolo-venulous fistulae with dilated fistula
AVMs treatment is complicated,
but in all cases the main goal is to obtain complete eradication of the nidus,
that causes the high-flow shunting between arterial and venous system.
Partial treatment usually results in recurrences that may be more difficult to manage than the initial malformation.
Large,
diffuse,
intracavitary and infiltrating lesions with muscle involvement are inoperable or require extensive,
potentially disfiguring,
resection or even amputation.
Transcatheter and percutaneous nidal embolization often is the first therapeutic option and is an effective approach that can be used as a palliative procedure,
giving only a temporary control,
or as an adjunct to a surgical resection.
Interruption of the proximal supplying vessels inevitably results in the development of a collateral arterial supply and an inability to access the feeding vessels for endovascular intervention.
The use of particulate agents (polyvinyl alcohol particles,
PVA),
should be reserved for preoperative embolization because recanalization rates are high.
A selective or superselective nidal access for embolic agent delivery must be obtained to maximally exposed the nidus to chosen agent effects,
minimizing local and systemic complications.
To achieve this goal,
sclerosant agents can be delivered transarterially in close proximity to the nidus,
using a superselective microcatheter or via a retrograde transvenous approach ,
with the assistance of a balloon occlusion device; a
direct percutaneous puncture into the nidus can also be used.
Moreover,
flow reduction techniques,
increase concentration,
and dwell time allow greater control of distribution of embolic agent within the nidus.
Whatever technique is used,
injection of the embolization agent is preceded by contrast injection into the vascular distribution to be embolized to determine the volume and flow rate of the malformation (Fig.
27,
28,
29,
30).