The exhibit will include a multimodality review pancreatic pathological processes and examples of each.
Congenital
A wide spectrum of anomalies of the pancreas and the pancreatic ductal system are commonly encountered at radiologic evaluation.
These anomalies may simulate various neoplastic,
inflammatory and posttraumatic conditions and should be part of the differential diagnosis for a variety of abnormalities found at diagnostic imaging.
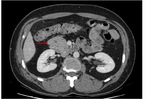
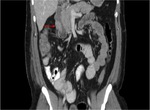
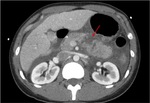
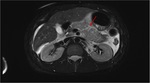
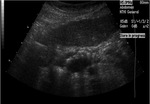
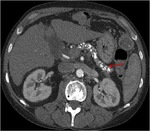
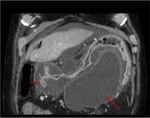
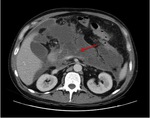
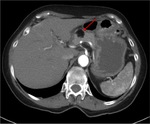
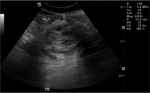
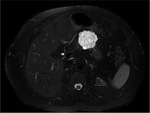
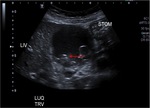
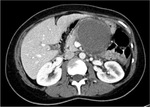
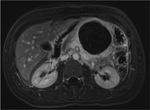
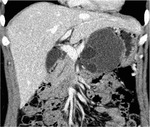
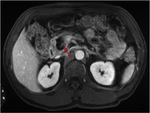
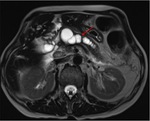
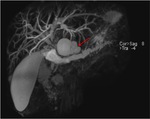
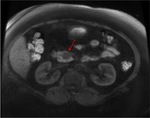
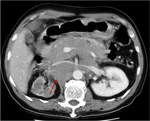
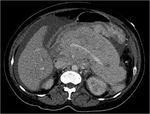
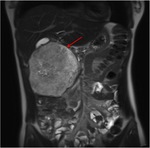
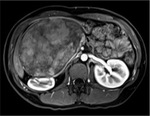
Annular Pancreas Fig 1,
2
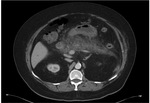
Annular pancreas is a rare congenital anomaly in which incomplete rotation of the ventral anlage leads to a segment of the pancreas encircling the second part of the duodenum.
It has a prevalence of 1 in 2,000 persons and occurs either as an isolated finding or with other congenital abnormalities.
- May present in childhood or as adult.
- Narrowed and stretched descending duodenum.
- Circumferential mass around 2nd portion of duodenum; easily identified on CT,
MR.
- Enhances like rest of pancreas.
- Symptoms of duodenal obstruction and/or pancreatitis.
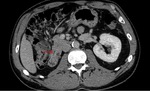
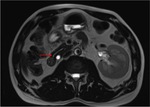
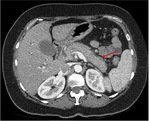
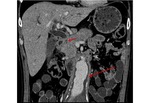
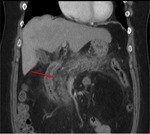
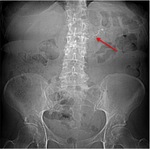
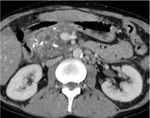
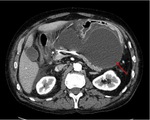
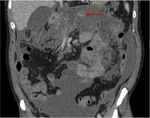
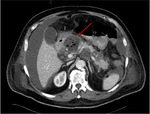
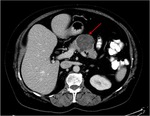
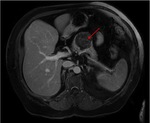
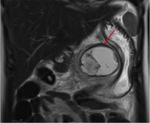
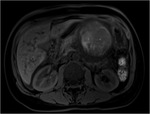
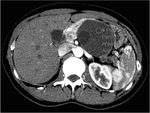
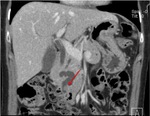
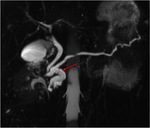
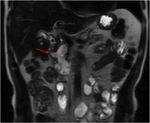
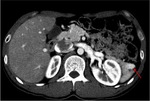
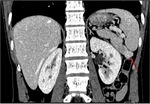
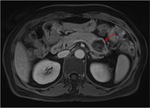
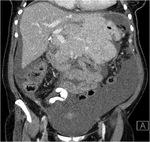
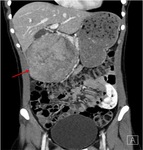
Pancreatic divisum Fig 3,
4
The most common congenital anomaly of the pancreatic ductal system,
being reported in 4%–10% of the population.
Ventral and dorsal pancreatic ducts fail to fuse.
The ventral duct (duct of Wirsung) drains only the ventral pancreatic anlage,
whereas the majority of the gland empties into the minor papilla through the dorsal duct (duct of Santorini). Clinically,
most cases are asymptomatic but may contribute to pancreatitis or recurrent episodes.
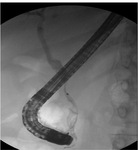
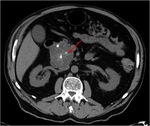
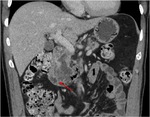
Trauma Fig 5,6
Pancreatic trauma is either blunt or penetrating; 3-12% of all abdominal injuries.
Lacerations usually accompany midline compression injury against the vertebral column and may involve other solid organs.
Presentation: History of trauma,
upper abdominal pain,
post prandial vomiting
Clinically: Raised serum amylase/lipase,
leukocytosis.
Complications include: Recurrent pancreatitis,
pseudocyst,
hemorrhage,
pseudoaneurysm,
fistula,
abscess.
- CT: accurate in detecting extrapancreatic fluid collections,
pancreatic lacerations/fractures.
- Emergency ERCP: Investigate pancreatic injuries when CT positive and status of PD uncertain.
- 24-48 hours delayed scans may uncover findings not present earlier.
Inflammation
Autoimmune pancreatitis (AIP) Fig 7-9
A multisystem disease process classified into two subtypes; lymphoplasmocytic sclerosing pancreatitis (LPSP) and idiopathic duct centric pancreatitis (IDCP).
Asian Consensus diagnostic criteria for AIP (Otsuki 2008):
- Pancreatic parenchymal imaging reveals diffuse/segmental/focal gland enlargement,
occasionally with a mass and/or rim of hypoattenuation.
- Pancreaticobiliary duct imaging reveals diffuse/segmental/focal duct narrowing ,
often with stenosis of the bile duct.
And either of the following
- Elevated serum IgG or IgG4 concentration and detection of antibodies,
or
- Lymphoplasmocytic infiltration of pancreatic tissue with abundant IgG4-postive plasma cells,
Or (optional criterion)
- Response to steroid therapy : due to dramatic positive response to steroids in AIP an optional criterion was included .
This stated that diagnostic trials of steroid therapy could be conducted in those patients fulfilling criterion 1 alone and negative work up for pancreaticobiliary cancer.
Acute pancreatitis Fig 10,11.
Acute inflammatory process of the pancreas with variable involvement of other local tissues and remote organ systems.
It is associated with elevated pancreatic enzyme levels in blood and/or urine.
Imaging:
- CT: focal or diffuse pancreatic enlargement,
inhomogenous enhancement and necrotic foci,
peripancreatic fluid collections and obliteration of fat planes.
Chronic pancreatitis Fig 12-15
The end result of a continuous,
prolonged,
inflammatory and fibrosing process that affects the pancreas.
This results in irreversible morphologic changes and permanent endocrine and exocrine pancreatic dysfunction.
Imaging:
- Atrophy of gland,
dilated main duct,
intraductal calculi,
thickening of peripancreatic fascia.
- Fibroinflammatory mass; clinical and imaging features overlap with cancer and there is an increased risk of adenocarcinoma in chronic pancreatitis (x20).
Biopsy not always helpful.
Look for:
- Pancreatic calcification (chronic pancreatitis).
- Smoothly tapering duct: benign.
- ‘double duct sign’ (stricture of distal CBD and pancreatic duct).
- vascular complications; splenic vein thrombosis and its sequelae and pseudoaneurysms of gastroduodenal or other arteries.
Pancreatic pseudocyst Fig 16,17
A complication of pancreatitis in 10-20% patients. An acute pancreatic fluid can transition into a pseudocyst after 4-6 weeks.
Increased amylase and lipase activity within the cyst due to communication with the pancreatic ductal system (70% cases).
Imaging:
- CT or MR; well circumscribed,
round or oval peripancreatic fluid collection of homogenously low attenuation surrounded by a well defined enhancing wall (capsule consisting of fibrous or granulation tissue).
No enhancement of pseudocyst contents.
- Gas within pseudocyst = infection or communication with gut wall.
- Pseudoaneurysm can be caused by or simulate a pseudocyst.
Pancreatic necrosis: (parenchymal necrosis,
peripancreatic necrosis,
or both) Fig 18,
19
- Enlarged pancreas,
indistinct borders,
lack of or no parenchymal enhancement(<30HU).
- Peripancreatic necrosis; most often around body and tail of pancreas,
anterior pararenal space,
lesser sac.
- Parenchymal and peripancreatic necrosis most common (75-80% of patients with acute necrotizing pancreatitis.
- Associated necrotic collections which contain non liquefied material; Acute non encapsulated necrotic collections (<4 weeks ) after acute episode of pancreatitis or walled off necrosis and encapsulated (>4 weeks).
Pancreatic abscess Fig 20
- Infections that contain non liquefied material are more likely to become infected.
- Imaging; gas pockets suspended within well circumscribed round,
oval or irregular fluid collections with enhancing walls/rind of inflammatory tissue.
- Definitive proof by FNA (+ve for gas forming organisms).
Benign neoplasm
Pancreatic lipoma Fig 21
Mesenchymal tumors exceedingly rare (1%).
Imaging:
• CT; homogenous distribution of fat density with no central or peripheral contrast enhancement,
Housefield units of -80 to -120 and a sharp demarcation with no evidence of intra- and extra-pancreatic adjacent structures infiltration.
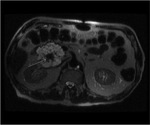
Serous cystic neoplasm (Formerly called serous cystadenoma) Fig 22-28
• Usually benign (<3% probability of degeneration).
• Mainly women,
peak age 65,
10% of cystic tumors.
• Imaging;
• Microcystic; body/tail,
no connection with pancreatic duct,
numerous microcysts cluster around central scar (calcification up to 30%),
lobulated shape,
radiate fibrous scar with delayed enhancement.
• Spongelike: Increasing size of cysts along periphery.
• Honeycomb like; Cysts of equal size in periphery and center.
• Oligocystic mainly pancreatic head unilocular or macrocystic (thin walls no unlike MCN,
no central scar or calcification).
Malignant neoplasm
Mucinous cystic neoplasm (MCN) (formerly mucinous cystadenoma) Fig 29-35
- Exclusively women ( cyst comprises subepithelial ovarian stroma),
premenopausal (peak age 47),
8% of cystic tumors premalignant,
Male: female 1:9.
- Lab data; Increased CEA,
increased cyst fluid levels of ca 19-9 ,
mucinous cyst content.
- Imaging;
- Singular or lobulated tumor 3-23cm; Unilocular or multilocular cysts,
body/tail pancreas,
no connection with pancreatic duct,
singular cysts in small numbers (greater in size than in SCN),
‘cyst in cyst’,
enhancement of cyst wall and septae (<2mm thick),
+/- hemorrhagic cyst content.
- Suspicion of malignancy; solid mural nodules,
thick enhancing septations,
cyst size >6cm,
peripheral ‘eggshell’ calcification.
Intraductal papillary mucinous neoplasm (IPMN) Fig 36-42
Mucinous transformation of the pancreatic ductal epithelium.
• Men>women 3:2,
peak age 60-70,
24% of cystic tumors,
premalignant.
• Main pancreatic duct,
sidebranch or mixed type.
Main duct type
- Mainly body,
tail,
also head,
connection with pancreatic duct,
macrocystic,
diffuse or segmental duct dilatation with cyst formation.
- Suspicion of malignancy: duct dilatation >1cm ,
enhancing mural tumor nodules,
calcifications,
cyst size >3cm,
extrapancreatic extension.
Branch duct type
- Mainly uncinate process,
multifocality in 30%,
connection with pancreatic duct.
- Macrocystic or unilocular,
bulbous dilated minor ducts,
cysts clustered or bundled together like clover leaves.
Mixed type
Neuroendocrine tumor (NET) Fig 43-47
Functioning or non functioning
- 1-5% all pancreatic tumors,
equal gender distribution ,
51-57yrs.
- Sporadic,
association with MEN 1,
Von Hippel Lindau syndrome,
neurofibromatosis type 1,
tuberous sclerosis.
- Imaging:
- Functioning (85%); small ,
+/-calcification (20%),
mostly hypervascular in arterial phase and portal venous phase; solid iso,
hypo or hyper,
Solid or ring enhancement (insulinoma).
- Non functioning; larger and complex,
cystic or necrotic areas +/- calcifcation,
can be hypervascular,
early tumor thrombus invasion of portal vein,
hypervascular ring metastases common.
- Longer T1 and T2 relaxation times,
therefore most are T1 hypo intense and T2 hyperintense.
- T1 fat sat; Homogenous avid arterial enhancement <2cm size, heterogeneous for larger tumors.
- Portal venous phase; hyper,
iso,
hypoenhancing.
- Hypervascular ring metastases are common.
- Higher sensitivity (up to 100%) with combined endoscopic USS and triple phase CECT.
- SPECT/CT; better attenuation correction and increased specificity.
- FDG/PET; role in poorly differentiated octretide negative NETs (high uptake due to high proliferative rate of tumor cells)
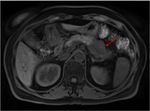
Pancreatic adenocarcinoma Fig 48,
49
85-95% of all pancreatic malignancies.
Malignancy that arises from ductal epithelium of exocrine pancreas.
- Male: Female 2:1
- CT or MR imaging;
- 60-70% pancreatic head,
Irregular,
heterogeneous,
poorly enhancing mass with abrupt obstruction of pancreatic ± common bile duct ("double duct" sign). 10% isodense/isointense and primary tumor not visualized. Calcification rare (<2%).
- 10% cystic/necrotic degeneration.
- T1/T2 hypointense,
hypovascular +/- thin peripheral rim enhancement.
- Parenchymal atrophy and ductal dilation upstream from tumor,
contiguous vascular ± organ invasion,
mesenteric vascular invasion (SMV ‘tear drop sign’),
Involvement = encasement of > 1/2 circumference of vessel,
narrowing,
or occlusion.
- Peritoneal and liver metastases common.
- Endoscopic USS high resolution imaging method for small tumors (0.2-0.3cm).
- FDG/PET avid tumor.
Pancreatic lymphoma Fig 50
• Most commonly B-cell NHL.
• Secondary lymphoma – most common,
30% NHL pts with widespread disease,
usually direct spread from peripancreatic lymph nodes.
• Primary rare,
<2% extranodal disease,
more common in immunocompromised patients.
• Imaging: Two morphological patterns.
• Focal well circumscribed vs.
diffuse form.
Features to help distinguish from adenocarcinoma
- +/-peripancreatic and other lymph node enlargement ( below renal veins).
- +/- encasement of peripancreatic vessels.
- Less likely to cause MPD dilatation (CBD dilatation >MPD ).
- Invasive tumor growth that does not respect anatomical boundaries,
infiltrates retroperitoneal or upper abdominal organs and GI tract.
Focal type
- Pancreatic head (80% cases).
- CT; uniform low attenuation.
- MR; T1 hypo,
T2 intermediate,
faint contrast enhancement.
Diffuse type
- Enlargement of pancreas with infiltrating tumor ± peripancreatic fat involvement; may simulate acute pancreatitis.
- CT; Homogenous soft tissue mass,
little enhancement.
- MR; T1 and T2 hypointensity,
homogenous contrast enhancement.
Pancreatic plasmacytoma Fig 51,
52
- 25 cases in the literature.
- An extramedullary neoplastic proliferation of plasma cells outside the confines of the osseous system.
- Typically presents as a solitary plasmacytoma of the paranasal and pharyngeal soft tissues (80%) or a secondary manifestation of systemic disease/ multiple myeloma.
- Involvement of GI tract 10% cases.
- Imaging:
- CT; Focal multilobulated solid hypodense mass with homogenous post contrast enhancement,
or rarely diffuse enlargement (+/- biliary duct dilatation).
Solid and pseudopapillary tumor (SPT) Fig 53,
54,
55
- 1-2% all pancreatic tumors,
9:1 female predilection,
average age 25yrs
- Large ~9cm,
slow growing,
well encapsulated mass,
cystic or hemorrhagic foci,
calcification (30%),
head>tail,
metastases uncommon (7-10% cases).
- Displaces rather than invades surrounding structures.
- Imaging:
- Peripheral heterogeneous enhancement arterial phase,
progressive non uniform enhancement thereafter,
enhancement usually less than normal pancreas
- CT; Well defined heterogeneous large mass +/- calcification (5-10%),
low density areas within,
thick enhancing capsule (solid component).
- MR; T1 large demarcated mass with mixed hypo and hyperintensity (hemorrhagic areas),
low intensity fibrotic capsule.