This poster is published under an
open license. Please read the
disclaimer for further details.
Keywords:
Sedation, Outcomes, Metastases, Treatment effects, Complications, Chemoembolisation, Ultrasound, CT, Catheter arteriography, Oncology, Liver, Interventional non-vascular
Authors:
G. Scevola1, A. Bruni2, M. Rastelli1, E. Papa1, S. Pacella3, S. Sposato1; 1Roma (RM)/IT, 2Roma/IT, 3Rome/IT
DOI:
10.1594/ecr2014/C-0821
Methods and materials
Between February 2009 and July 2013,
87 patients (47 male and 39 female aged 54 to 79 years) with colorectal liver metastases,
not treatable by surgery,
were treated with the FOLFOX and FOLFIRI chemotherapy regimens.
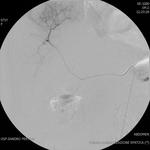
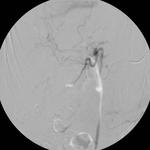
Patients who did not respond to chemotherapy and who were eligible to third-line therapy were treated locally with Irinotecan-loaded Drug-eluting Beads (DEBIRI®).
This technique consists of the intrahepatic embolization of metastases with non-absorbable 75-150 micron beads (DCBeads- Biocompatibles UK Ltd)) pre-loaded with 200 mg of irinotecan.
The periprocedural medications adopted included initially antiemetic (ondansetron hydrochloride) painkilling (tramadol hydrochloride) drugs,
H2-blockers (ranitidine hydrochloride) and 5-days antibiotic prophylaxis,
as commonly used in standard TACE procedures.
However,
this medication strategy often failed to achieve an adequate pain control,
precluding subsequent treatment courses because of patients refusal.
Thus,
evaluation of the efficacy of DEBIRI in these patients was compromised since at least two treatment courses are required for it.
In 2010 we adopted the following periprocedural medication protocol: 4 hours before treatment morphine 2 mg and ondansetron hydrochloride 2 mg,
by i.v.
infusion pump and 1 hour before treatment paracetamol 1,000 mg.
During the procedure,
boluses of morphine 0.2 mg up to a maximum of 2 mg,
and of midazolam hydrochloride up to a maximum 5 mg were administrated.
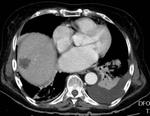
Most of the patients (82 %) had more than one metastasis.
Involvement of more than 50% of the hepatic parenchyma,
evaluated by volumetric CT,
was considered as exclusion criteria.
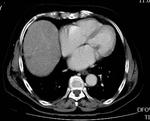
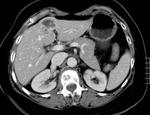
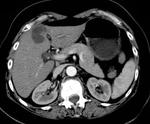
Efficacy of treatment,
denoted by lack of disease progression and by reduction of the size of metastases,
was evaluated after 4,
8,
12 and 24 months performing both CEUS (SonoVue,
Bracco) and CT scan (Lightspeed 16,
GE).