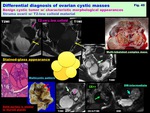
[Conventional and advanced MRI for the differential diagnosis]
- Conventional MRI (T1-/T2WI) ( Fig. 1 )
-Water shows T2-very high and T1-low signal pattern
-Serous fluid shows water-like signal pattern
-Thin mucinous fluid may also show water-like intensities,
however,
thick mucinous material may show T1-increase / T2-decrease pattern
-Fat shows high signal intensity on both T1- and T2WI
-Hemorrhagic fluid may show various signal patterns:
-Acute hemorrhage may show T2-decrease due to deoxyhemoglobin
-Subacute to chronic hemorrhage may show T1/T2 increase due to methemoglobin
-Hematocrit effect may be observed
-Repeated hemorrhage in endometrioma may show T1-high,
and T2-low (shading) due to hyperviscosity,
and high concentration of protein and hemosiderin

Fig. 1: Conventional MRI
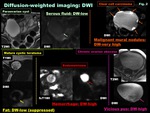
- Diffusion-weighted imaging: DWI ( Fig. 2 )
-DWI visualizes the local microstructural characteristics of water diffusion
-Water /serous fluid show DW-low
-Fat shows DW-low due to fat-suppression
-DW-very high cystic components is suggestive for thick,
viscous material such as pus in abscess,
hyperkeratinous material in mature cystic teratoma,
or thick mucinous materials
-Hemorrhagic material may also show DW-signal increase
-Malignant solid components may appear as DW-very high with low ADC reflecting hypercellularity,
whereas benign solid components tend to show DW-low to intermediate
-Some benign T2-high portion may show DW-high (T2-shine through effect),
and high ADC is suggestive for its benignity
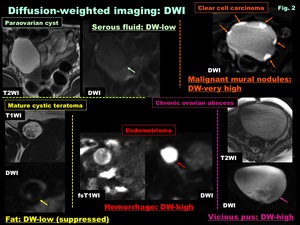
Fig. 2: Diffusion-weighted imaging: DWI
- Susceptibility-weighted imaging (SWI) ( Fig. 3 )
-SWI has been used in imaging of central nervous system,
and recently in body imaging,
too
-SWI combines magnitude and phase information from fully velocity compensated gradient-echo sequence,
and visualizes the magnetic susceptibility effects generated by local inhomogeneity of the magnetic field as signal voids
-To enhance the visibility of signal voids caused by the magnetic susceptibility effects,
post-processing is applied to the magnitude images multiplied with a phase mask generated from the filtered phase data
-SWI has exquisite sensitivity to the blood products and can demonstrate especially deoxyhemoglobin in acute hemorrhage,
and hemosiderin deposits in obsolete hemorrhage as signal voids due to T2* shortening
-Spotty /curved linear signal voids on SWI is specific for endometrioma reflecting repeated cyclic hemorrhage as signal voids along the cyst wall

Fig. 3: Susceptibility-weighted imaging: SWI
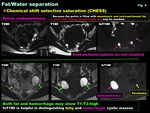
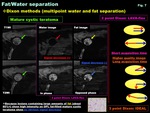
- Fat/Water separation (Fig.
4-7)
-Fat/Water separation by various MR techniques can distinguish fatty and hemorrhagic tumors
-In pelvic region,
fat saturation sequence (chemical shift selective saturation: CHESS) is commonly used: Fat resonance is excited selectively,
and then the signal is spoiled using gradient pulses.
CHESS is sensitive to radiofrequency magnetic induction field inhomogeneities resulting in non-uniform fat suppression ( Fig. 4 )

Fig. 4: Fat/Water separation: Chemical shift selective saturation (CHESS)
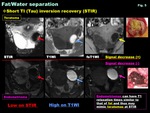
-Short TI (Tau) inversion recovery (STIR): Inversion recovery sequence with the value of inversion time (TI) chosen for the null point for the fat signal.
STIR is not suitable for female pelvic examination,
because not only teratomas but also endometriomas or other hemorrhagic cysts may appear as low intensity ovarian cystic masses ( Fig. 5 )

Fig. 5: Fat/Water separation: Short TI (Tau) inversion recovery (STIR)
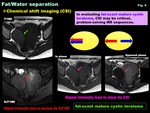
-Chemical shift imaging (CSI),
combination of in-phase image (IPI) and opposed-phase image (OPI),
is useful to prove the existence of fatty components.
Fat can be estimated by comparing IPI with OPI (180 degree phase difference between lipid and water spins because of the difference in the chemical shifts).
CSI is used to assess for the presence of fat and water in a voxel of tissue.
OPI show more signal intensity loss in tissues containing relatively small quantities of fat ( Fig. 6 ).
Lesions containing large amounts of fat (about 80%<) show high intensity on OPI ( Fig. 7 )

Fig. 6: Fat/Water separation: Chemical shift imaging (CSI)
-Dixon methods (multipoint water and fat separation): 3D dual-echo Dixon techniques (2 point: LAVA-flex /3 point: IDEAL) is robust to B0 inhomogeneity,
and eliminate fat signal on the basis of the differences in precessional frequency between water and fat,
which leads to better and more uniform fat suppression,
and may facilitates superior-quality fat suppression in the pelvis compared with conventional fsT1WI (CHESS).
High quality water-only images,
fat-only images,
IPI & OPI can be obtained at once ( Fig. 7 )

Fig. 7: Chemical shift imaging (CSI): Dixon methods (multipoint water and fat separation)
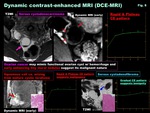
- Dynamic contrast-enhanced MRI (DCE-MRI) ( Fig. 8 )
-DCE-MRI can reveal the angiogenic activity of tumors
-Malignant solid components tend to show hypervascularity due to angiogenesis of tumor cells
-Demonstrating tiny early-enhancing mural nodules on 3D-DCE-MRI is helpful in diagnosing ovarian cancer in early stage
-Rapid & Plateau CE pattern of dynamic curve suggests hypervascular malignant tumor,
whereas Gradual CE pattern suggests hypovascular benign (fibrous) tumor

Fig. 8: Dynamic contrast-enhanced MRI (DCE-MRI)
- High resolution (HR) MR imaging at 3T ( Fig. 9 )
-The increase in SNR at 3T offers high-resolution (HR) MR imaging
-Anatomically detailed structures are visualized on HR MRI and may improve the diagnostic accuracies in differential diagnosis
-HR MR images can demonstrate characteristic imaging features sush as aggregated tiny cysts in lobular endocervical glandular hyperplasia (LEGH)
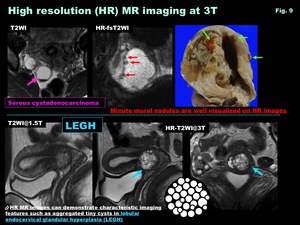
Fig. 9: High resolution (HR) MR imaging at 3T
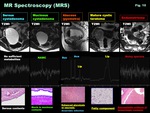
- MR Spectroscopy (MRS) ( Fig. 10 )
-Proton MR spectroscopy (MRS) demonstrates tissue metabolite concentrations along a spectrum based on the chemical shift phenomenon
-MRS provides metabolic information,
which is useful for the differentiation of benign and malignant pathologies,
and in making differential diagnosis,
especially in the estimation of cystic fluid contents
-Susceptibility artifacts due to hemorrhagic contents may cause noisy spectra
-Choline (Cho) (3.2 ppm) peak reflects metabolic activity of cell membrane in solid components,
and may be observed in both benign and malignant solid portions.
High grade malignancy tend to show higher choline peaks
-Lipid (Lip) (1.3 ppm) peak is observed in lesions with fat or mobile lipids.
Necrosis-associated lipid peak may be observed in both solid and cystic portions of malignant tumor.
Lipid detection in cystic portion may also useful in diagnosing mature cystic teratoma with scant fatty component.
Lip peak is also observed in abscess due to enhanced glycolysis and necrosis
-N-acetyl mucinous compounds (NAMC) (2-2.1 ppm) peak,
mimicking NAA peak observed in CNS,
is suggestive for mucinous tumors such as primary mucinous tumors,
and secondary tumors containing mucinous materials such as Krukenberg’s tumor.
This peak is from N-acetyl compounds in mucus glycoproteins.
NAMC peak in peritoneal fluid collection is suggestive for pseudomyxoma peritonei
-Acetate (Ace) (1.9 ppm) and Succinate (Suc) (2.4 ppm) peaks are observed in abscess with anaerobic infection as the result of enhanced glycolysis and the fermentative pathways for energy generation.
These peaks are not observed in aerobic abscess,
or tumors with massive necrosis mimicking abscess

Fig. 10: MR Spectroscopy (MRS)
[5 steps to the diagnosis]
Step 1: Adnexal,
Uterine,
or Non-gynecologic origin?
-Step 1 is the localization of the lesion
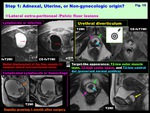
-Normal ovary detection is the very first step (DWI is helpful) ( Fig. 11 )
-Normal ovaries of reproductive age exhibit distinct features of a zonal anatomy:
Thin capsule-like T2-low rim (tunica albuginea -cortex) due to abundant fibrous stroma; T2-very high follicular cysts; T2-Intermediate medulla due to loose stroma
-Normal ovaries after menopause: Ovaries shrink in size w/ follicles replaced by Corpus Albicans,
and may show T2-Homogeneous appearance
-Various extra-ovarian tumors may appear as ovarian tumor-like cystic pelvic masses such as gastrointestinal,
appendiceal,
urachal,
uterine,
peritoneal or extra-peritoneal tumors.
The presence of a normal ipsilateral ovary separate from the cystic mass is suggestive for its extra-ovarian origin
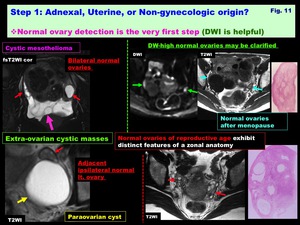
Fig. 11: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Normal ovary detection is the very first step (DWI is helpful)
Localization /Origin of cystic masses
Extra-peritoneal:
> Presacral: Teratoma /Tail gut cyst /Anterior meningocele
> Lateral: Lymphocele,
Mega-ureter
> Presacral /Lateral: Ancient schwannoma
> Anterior (prevesical space of Retzius): Urachal lesions
> Pelvic floor: Urethral diverticulum /Subcutaneous cystic masses
Peritoneal cavity:
> Peritoneal: Lymphangioma /Cystic mesothelioma /Peritoneal inclusion cyst
> Intestinal: Appendiceal mucocele (may be extra-peritoneal) /Duplication cyst
> Others: Endometrial cysts (implants) /Abscesses
Uterus:
> Myometrial: Adenomyotic cyst /Adenomatoid tumor / Degenerated myoma /Leiomyosarcoma w/ massive necrosis
> Cervical: LEGH /Nabothian cysts /Adenoma malignum (MDA)
> Enlarged cavity: Pyometra /Hydrometra /Hematometra
Adnexal:
> Extra-ovarian: Paraovarian cyst /Tubal dilatation (Hydrosalpinx /Pyosalpinx)
> Ovarian:
-Functional cysts /Surface-epithelial inclusion cysts
-Benign enlargement (HL/OHSS)
-Tubo-ovarian abscess /Endometrioma
-Cystic tumors (benign & malignant)
-Localization: Extra-peritoneal lesions ( Fig. 12 )
Centripetal displacement sign: The location of extra-peritoneal tumors could be determined by detecting centripetal displacement of the adjacent structures.
Anterior displacement of the compressed rectum in retroperitoneal tumors arising from the presacral tissue.
Inward displacement of the compressed iliac vessels or ureter in lateral extra-peritoneal tumors
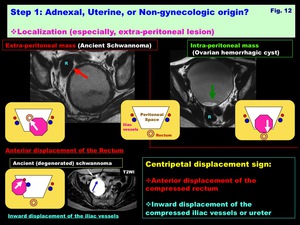
Fig. 12: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Localization (especially, extra-peritoneal lesion)
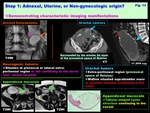
-Localization by demonstrating characteristic imaging manifestations suggesting the origin of the lesions ( Fig. 13 )
Neurogenic tumors:
-Mimicking ovarian tumor,
because asymptomatic and US reveals complex pelvic masses
-Situates at presacral or lateral extra-peritoneal region with the continuity to the nerve root /neural foramen
Urachal tumors:
-Exists in extra-peritoneal region (prevesical space of Retzius)
-Midline situated suprabladder mass with urachal remnant continuing to the navel suggests its urachal origin
-Mucinous carcinoma is common,
and often calcified caused by mucin
-Pseudomyxoma may occur when ruptured
Appendiceal mucocele:
-May often mimic ovarian cyst,
and ruptured mucocele may cause pseudomyxoma peritonei
-Demonstration of tubular cystic structure,
which is the enlarged,
fluid-filled appendix,
continuing to the cecum suggests its appendiceal origin
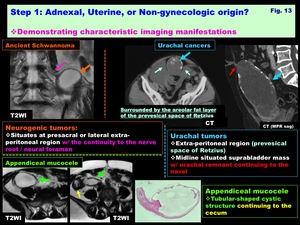
Fig. 13: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Demonstrating characteristic imaging manifestations
Presacral extra-peritoneal lesions: ( Fig. 14 )
-Mature cystic teratoma:
-Extragonadal teratomas derive from totipotential cells of primordial germ cell or early embryonic cell origin,
and commonly locates in presacral region
-Imaging manifestations are similar to those of ovarian teratomas: The presence of fat is diagnostic
-Tail gut cyst:
-Hamartomatous lesion located at retrorectal region arising from the vestiges of the embryonic hindgut
-Usually asymptomatic,
and may cause pain or constipation
-Unilocular or multilocular cystic mass with mucoid contents
-Rarely with malignant transformation
-Water-like pattern on T1/T2WI,
or T1-increase due to mucin,
hyperproteinous material or hemorrhage
-May show DW-very-high due to vicious mucoid contents

Fig. 14: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Presacral extra-peritoneal lesions
Lateral extra-peritoneal /Pelvic floor lesions: ( Fig. 15 )
-Lymphocele:
-Surgical complication seen after extensive pelvic surgery (lymphadenectomy),
usually within 4 months after surgery
-Lymphatic fluid collection due to destruction and disruption of the normal channels of lymph flow
-Usually asymptomatic,
but may be complicated w/ infection
-Urethral diverticulum:
-Urethral protrusion into the urethral-vaginal potential space
-May be asymptomatic,
postvoiding dribbling,
or dyspareunia
-Painful mass in the anterolateral vaginal wall
-Repeated infection w/ duct obstruction leads to Skene gland cyst,
which may rupture into the lumen and form diverticulum surrounding the urethla as “target-like” appearance
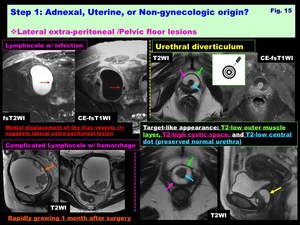
Fig. 15: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Lateral extra-peritoneal /Pelvic floor lesions
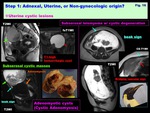
Uterine cystic lesions: ( Fig. 16 )
-Adenomyotic cyst (Cystic adenomyosis):
-Rare variation of adenomyosis appearing as endometrioma-like hemorrhagic cyst surrounded by adenomyotic tissue (T2-low rim)
-May mimic sarcoma w/ massive necrosis and hemorrhage
-May appear as subserosal polypoid cystic mass
-Degenerated myoma:
-Subserosal leiomyoma w/ prominent cystic degeneration may mimic ovarian tumor
-Beak sign & bridging vascular sign are suggestive for uterine origin

Fig. 16: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Uterine cystic lesions
-Enlarged cavity (Pyometra /Hydrometra /Hematometra):
( Fig. 17 )
-Fluid collection in the uterine cavity may appear as thick-walled cystic mass.
-Obstruction of cervical canal or lower portion of the cavity due to a cervical mass or post-inflammatory adhesion causes this condition
-Serous fluid collection (hydrometra) shows water-like intensity
-Hemorrhagic fluid collection (hematometra) may cause T1-signal increase w/ Hematocrit effect
-DW-very high viscous pus collection (pyometra) due to infection may have T1-high wall due to free radical
-Pyometra is the pus collection in the uterine cavity resulting from cervical canal occlusion with infection
-Usually occurs in elderly postmenopausal women
-Typically,
lower abdominal pain,
purulent vaginal discharge,
postmenopausal bleeding.
However,
asymptomatic in half of cases
-Rarely,
spontaneous perforation may subsequent diffuse peritonitis
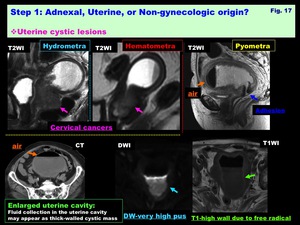
Fig. 17: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Uterine cystic lesions
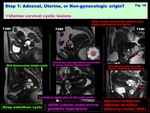
Uterine cervical cystic lesions: ( Fig. 18 )
-Nabothian cysts:
-Nabothian cysts are retention cysts of endocervical glands,
which may extend deeply into the cervical wall as deep nabothian cysts w/wo cervical enlargement
-Lobular endocervical glandular hyperplasia (LEGH):
-LEGH is benign hyperplastic lesion w/ cystic dilatation of endocervical glands w/o solid components
-Adenoma malignum (Minimal deviation adenocarcinoma):
-Adenoma malignum is rare variant of cervical well differentiated adenocarcinoma.
-Prominent watery discharge is suggestive symptom
-May often invade beyond the endocervical region and involve the portio
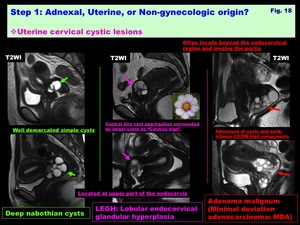
Fig. 18: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Uterine cervical cystic lesions
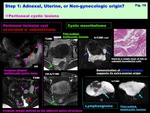
Peritoneal cystic lesions: ( Fig. 19 )
-Peritoneal inclusion cyst:
-May appear as a cystic lesion w/ irregular margin defined by the adjacent pelvic structures
-Extensive pelvic adhesions due to surgical procedure,
trauma,
inflammation or endometriosis may trap the intra-peritoneal fluid and form the pseudocystic lesion including normal ovaries
-May mimic multilocular cystic tumor,
however,
w/o true cystic wall,
and drainage is required for the treatment
-Cystic mesothelioma
-Rare neoplasm occurs predominantly in women of reproductive age
-Extra-ovarian intraperitoneal cystic mass,
typically appear as thin-walled,
multicystic lesions with septal CE-walled,
multicystic lesions
-Lymphangioma
-Lymphangioma is a congenital anomaly and occurs in children,
and may appear as extra-ovarian,
thin-walled,
multicystic lesions
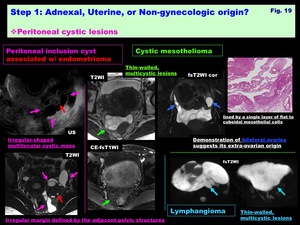
Fig. 19: Step 1: Adnexal, Uterine, or Non-gynecologic origin?
Peritoneal cystic lesions
Step 2: Evaluation of cystic components: Serous,
Mucinous,
Colloid,
Hemorrhagic,
Keratinous,
Fatty or Pus? (Fig.
20-24)
Differentiation by using advanced MR techniques
Serous: ( Fig. 20 )
> T1 low/ T2 high/ DW low (water-like)
> Occasionally thin mucinous or hemorrhagic intensity
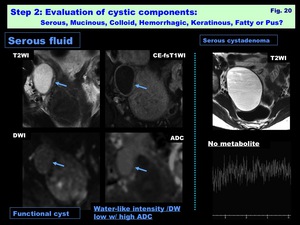
Fig. 20: Step 2: Evaluation of cystic components: Serous, Mucinous, Colloid, Hemorrhagic, Keratinous, Fatty or Pus?: Serous fluid
Mucinous: (depending on the mount of proteinous material) ( Fig. 21 )
> T1 low to high/ T2 high to low/ DW low to high
> N-acetyl mucinous compounds (NAMC) peak is specific
> May various signal intensities (stained-glass) in multilocular (mucinous tumors)
Colloid:
> T2 very low/ T1 low
> May various signal intensities (stained-glass) in multicystic (struma ovarii)
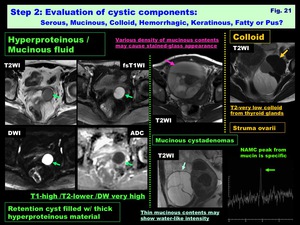
Fig. 21: Step 2: Evaluation of cystic components: Serous, Mucinous, Colloid, Hemorrhagic, Keratinous, Fatty or Pus?: Hyperproteinous /Mucinous fluid, Colloid
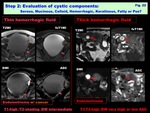
Hemorrhagic: (depending on the oxidation state of hemoglobin) ( Fig. 22 )
> T1 low to high (Met Hb in subacute: high)
> T2 high to low (Deoxy Hb in acute/Hemosiderin in obsolete: low)
> DW low to high/ SWI low to high
> May form hematocrit effect (fluid level)
> Suboptimal MRS (noisy spectra) due to susceptibility artifact

Fig. 22: Step 2: Evaluation of cystic components: Serous, Mucinous, Colloid, Hemorrhagic, Keratinous, Fatty or Pus?: Thin hemorrhagic fluid, Thick hemorrhagic fluid
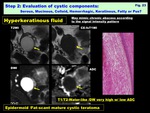
Keratinous: ( Fig. 23 )
> T1/T2 water-like
> DW very high

Fig. 23: Step 2: Evaluation of cystic components: Serous, Mucinous, Colloid, Hemorrhagic, Keratinous, Fatty or Pus?: Hyperkeratinous fluid
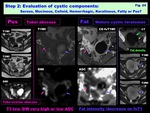
Fatty: ( Fig. 24 )
> T1 high/ T2 high/ DW low/ Signal decrease by fat suppression
> Small amount of fat can be detected by CSI / MRS (lipid peak)
Pus:
> T1/T2 water-like,
DW very high
> Lipid peak / Acetate and Succinate peaks (anaerobic infection)

Fig. 24: Step 2: Evaluation of cystic components: Serous, Mucinous, Colloid, Hemorrhagic, Keratinous, Fatty or Pus?: Pus, Fat
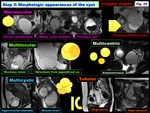
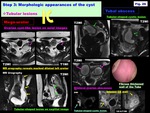
Step 3: Morphologic appearances of the cyst: Monolocular,
Multilocular,
Multicystic,
Tubular,
or Irregular shaped? ( Fig. 25 )
-Monolocular:
> Non-neoplastic ovarian cystic masses
> Various ovarian /extra-ovarian cystic tumors
-Multilocular:
> Mucinous tumors (primary /secondary)
-Multicystic:
> Struma ovarii
> Benign ovarian enlargement: Hyperreactio luteinalis (HL) /OHSS
> Lymphangioma /Cystic mesothelioma
> Uterine cervical cystic lesions (LEGH /Nabothian cysts)
-Tubular: ( Fig. 26 )
> Extra-ovarian: Fallopian tube/ Appendix /Ureter
Mega-ureter:
-Anomaly whereby the ureter is abnormally dilated
-Functional obstruction at the lower end of the ureter leads to progressive dilatation
-May mimic ovarian cyst,
but tubular-shaped structure continuing to the bladder is suggestive
Tubal abscess:
-Ovarian involvement in PID usually takes a form of bilateral tubo-ovarian abscess secondary to salpingitis
-Subclinical patients are common (especially in Chlamydia trachomatis infection)
-Tortuous,
fluid-filled tubal lumen with/without thickened wall suggests hydro/pyosalpinx
-Ill-defined adnexal mass with thick,
irregular walls containing fluid suggesting tubo-ovarian abscess
-Wall and pus show DW-very high
Appendiceal mucocele: see above ( Fig. 13 )
-Irregular shaped:
> Peritoneal inclusion cyst
-Multicentric:
> Endometrioma (multiplicity)
> Lymphocele (solitary or multiple)
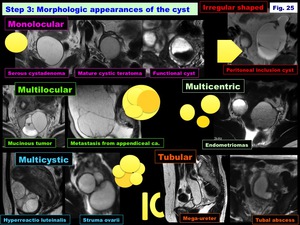
Fig. 25: Step 3: Morphologic appearances of the cyst

Fig. 26: Step 3: Morphologic appearances of the cyst: Tubular lesions
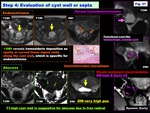
Step 4: Evaluation of cyst wall or septa:
Mural nodules /thickened septa: ( Fig. 27 )
> Probable malignancy
> DW-very high w/ low ADC,
Early CE suggests malignancy
> DW-low (benign serous nodules /fibrous nodule in endometrioma) or DW-high w/ high ADC (decidualized endometrioma),
weak –no CE suggest benignity
> Benign dermoid nipple may show DW-very high /CE
> Intervening ovarian stroma in HL may mimic malignant foci
Hemosiderin deposits:
> Spotty /curved linear signal voids on SWI,
specific for endometrioma:
Endometrioma may appear as non-specific hemorrhagic mass: T1/T2-high,
and SWI reveals hemosiderin deposition reflecting repeated cyclic hemorrhage as signal voids along the cyst wall
Free radical:
> T1-high due to Paramagnetic effect of free radical in abscess wall

Fig. 27: Step 4: Evaluation of cyst wall or septa
Calcification: ( Fig. 28 )
> Calcification induced by mucin is frequent in mucinous tumors
> Psammoma body in mural nodule of serous tumors
> Common in mature cystic teratoma
> Obsolete degeneration in benign tumors (ancient schwannoma)
Granulation:
> Thick abscess wall with intense CE,
T2/DW-signal increase
Fibrosis:
> T2-low with delayed CE,
in endometrioma /chronic abscess

Fig. 28: Step 4: Evaluation of cyst wall or septa (cont'd)
Step 5: Evaluation of Clinical information: Age,
hormonal state,
pregnancy,
symptoms…
Age:
> Reproductive era only: Ovarian functional cysts
> Common in Reproductive age: Endometrioma,
Adenomyotic cyst
> Common in postmenopausal: Surface-epithelial inclusion cyst
> Malignant lesions tend to affects elder women
Hormonal state /pregnancy:
> Pregnancy related: Decidualized endometrioma /HL
> During ovulation induction therapy: OHSS
Postsurgery:
> Lymphocele,
Peritoneal inclusion cyst (also occur in post-inflammation or endometriosis,
due to adhesion)
Symptoms:
> Usually no symptom,
Large cyst may cause abdominal fullness
> Intracystic hemorrhage causes temporal pain (Corpus luteum cyst)
> Symptoms associated w/ PID (Abscess)
> Symptoms associated w/ endometriosis (Endometrioma)
> Abdominal fullness,
Nausea,
Vomiting,
Oliguria… (OHSS)
> Paraneoplastic limbic encephalitis (Mature cystic teratoma)
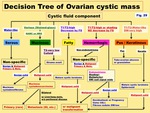
[Differential diagnosis of ovarian cystic masses]
Decision Tree of Ovarian cystic mass ( Fig. 29 )
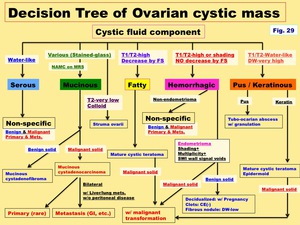
Fig. 29: Decision Tree of Ovarian cystic mass
Benign cystic mass w/o solid portion (Fig.
30-34)
-Functional cysts (Follicular cyst /Corpus luteum cyst): ( Fig. 30 )
-Retention cyst associated with ovulation
-Never occur after menopause
-Spontaneously shrunk after 1-3 months
-Follicular cyst: Unruptured follicle at ovulation
-Corpus luteum cyst: Repaired after ovulation,
Often hemorrhagic,
May mimic endometrioma (no shading)
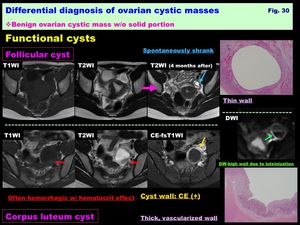
Fig. 30: Differential diagnosis of ovarian cystic masses:
Benign ovarian cystic mass w/o solid portion:
Functional cysts
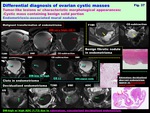
-Surface-epithelial inclusion cyst: ( Fig. 31 )
-Arise from cortical invaginations of the ovarian surface epithelium
- Observed in all ages,
particularly common in postmenopausal ovaries
-May appear as follicle-like cysts,
may be multiple
-Small cyst:1cm> (Larger lesion is identical to serous cystadenoma)
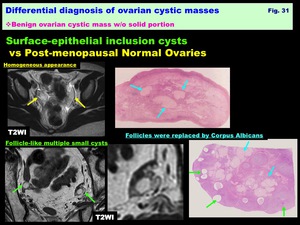
Fig. 31: Differential diagnosis of ovarian cystic masses:
Benign ovarian cystic mass w/o solid portion:
Surface-epithelial inclusion cysts vs Post-menopausal Normal Ovaries
-Serous cystadenomas: ( Fig. 32 )
-Common benign cystic tumor
-Usually monolocular,
occasionally multilocular
-Thin-walled w/ serous fluid contents
-Benign small mural nodules w/wo psamommatous calcification may be seen (No CE /DW-low)
-No sufficient metabolites on MRS

Fig. 32: Differential diagnosis of ovarian cystic masses:
Benign ovarian cystic mass w/o solid portion:
Serous cystadenoma
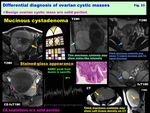
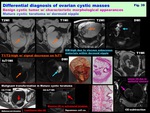
-Mucinous cystadenomas: ( Fig. 33 )
-Usually multilocular,
rarely monolocular
-Various contents in the loculi may show variable signal intensity as “stained-glass appearance”
-Thick mucinous contents within the daughter cyst may show soft tissue density on CT mimicking solid mural nodules
-Borderline malignancy and adenocarcinomas have a larger number of loculi than benign cystadenomas
-May be calcified along the wall –septa due to mucin
-N-acetyl mucinous compounds (NAMC) peak from mucin is specific on MRS
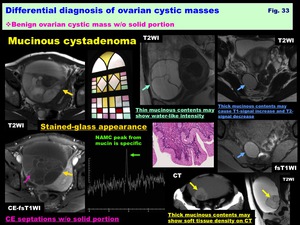
Fig. 33: Differential diagnosis of ovarian cystic masses:
Benign ovarian cystic mass w/o solid portion:
Mucinous cystadenoma
-Endometrioma (Chocolate cyst): ( Fig. 34 )
-Endometriosis is a condition characterized by the presence of ectopic functioning endometrial tissue outside the uterus
-Endometriosis is a common disease of women in their reproductive era,
and usually affects ovaries as endometriomas
-Thick fibrous capsule of the cystic tumor containing highly viscous,
dark hemorrhagic chocolate-like material
-"Shading" on T2WI,
and "multiplicity" on T1WI are diagnostic
-"Shading" is T2-shortening in T1-high ovarian cyst,
"Shading" may appear as complete loss of signal intensity or dependent layering with T2-low fluid level,
The cause of T2-low is complex; hyperviscosity,
and high concentration of protein and hemosiderin from recurrent cyclical bleeding may contribute to T2-shortening
-Spotty /curved linear signal voids on SWI is specific for endometrioma reflecting repeated cyclic hemorrhage as signal voids along the cyst wall
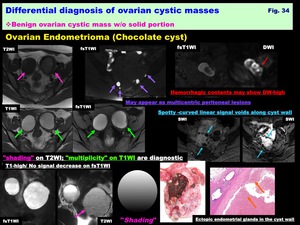
Fig. 34: Differential diagnosis of ovarian cystic masses:
Benign ovarian cystic mass w/o solid portion:
Ovarian Endometrioma (Chocolate cyst)
Tumor-like lesion w/ characteristic morphological appearances:
-Cystic mass containing ovarian parenchyma:
Hyperreactio luteinalis / OHSS: ( Fig. 35 )
-Caused by endogenous or pregnancy-related hormonal stimulation
-Usually associated with gestational trophoblastic disease or polycyesis,
less commonly with normal pregnancy
-Usually bilateral,
occasionally unilateral
-Regresses in size after delivery or removal of causative factors
-May mimic mucinous tumors,
and misdiagnosis may lead to unnecessary surgery
-Marked bilateral ovarian enlargement with uniform multiple cysts (theca lutein cysts caused by increased serum hCG level) separated by intervening parenchyma as “spoke-wheel” appearance
-Edematous,
luteinized stroma of residual ovarian parenchyma shows T2-high; DW-high with relative high ADC (T2-shine through)
-ADC: Hyperreactio luteinalis: 1.86 +/- 0.37 vs Ovarian cancers: 1.04 +/- 0.21 (Takeuchi M et al.
JCAT 35; 2011)
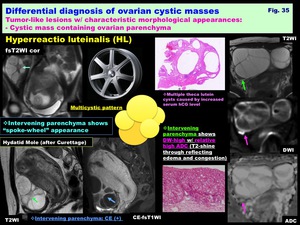
Fig. 35: Differential diagnosis of ovarian cystic masses:
Tumor-like lesions w/ characteristic morphological appearances:
- Cystic mass containing ovarian parenchyma:
Hyperreactio luteinalis (HL)
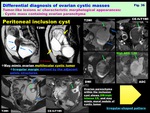
Peritoneal inclusion cyst: ( Fig. 36 )
-May appear as a cystic lesion with irregular margin defined by the adjacent pelvic structures
-Accurate preoperative diagnosis allows application of more conservative therapy
-Extensive pelvic adhesions due to surgical procedure,
trauma,
inflammation or endometriosis may trap the intra-peritoneal fluid and form the pseudocystic lesion including normal ovaries
-Ovarian parenchyma within the inclusion cyst shows DW-high/intense CE,
and may mimic mural nodule of cystic tumor,
however,
the ADC of ovarian parenchyma may be higher than that of cancers.

Fig. 36: Differential diagnosis of ovarian cystic masses:
Tumor-like lesions w/ characteristic morphological appearances:
- Cystic mass containing ovarian parenchyma:
Peritoneal inclusion cyst
-Cystic mass containing benign solid portion:
Endometriosis-associated mural nodules: ( Fig. 37 )
Appearance of mural nodules in endometrioma is suggestive for malignant transformation,
however,
some benign lesions may appear as solid portion in endometrioma and may mimic malignancy
-Decidualized Endometrioma
-With the hypertrophy of the endometrial stromal cells,
the normal uterine endometrium may thicken and transform to the decidua induced by progesterone during pregnancy
-Decidual change during pregnancy in ectopic endometrial tissue of endometrioma may cause mural nodules,
typically multiple,
broad-based,
small nodules,
but occasionally polypoid
-Signal intensity is similar to that of the placenta: T2-very high; DW-high with high ADC (T2-shine through),
whereas malignancy shows low ADC
- ADC measurement is useful for differentiating from malignant transformation: Decudual nodules: 2.10 +/- 0.32 vs Cancer: 1.05 +/- 0.13 (Takeuchi M et al.
JCAT 32; 2008)
-Clots or benign fibrotic mural nodule in Endometrioma
-Clots may show various signal intensity: T2-low to inhomogeneous high,
DW-high
-CE-subtraction image is useful for the diagnosis,
because T1-high hemorrhagic contents may mask CE nodules on usual CE-T1WI
-SWI may also be helpful in demonstrating hemosiderin deposits in clots
-Benign fibrotic nodule may appear as CE mural nodule.
DW-low may be suggestive for its benign nature,
however,
hemorrhagic contents may show DW-high and mask small nodule

Fig. 37: Differential diagnosis of ovarian cystic masses:
Tumor-like lesions w/ characteristic morphological appearances:
Cystic mass containing benign solid portion:
Endometriosis-associated mural nodules
Abscess w/ granulomatous tissue: ( Fig. 38 )
-Ovarian involvement in pelvic inflammatory disease (PID) usually takes a form of bilateral oophoritis/tubo-ovarian abscess secondary to salpingitis due to infection
-Symptoms are typically lower abdominal pain,
and less consistently fever,
vaginal discharge or bleeding,
and urinary symptoms,
however,
patients are often asymptomatic
-Ill-defined adnexal mass with thick,
irregular walls containing DW-very high fluid reflecting heavily impeded water mobility of pus related to the hyperviscosity suggests tubo-ovarian abscess.
-Thick fibrous wall to surrounding inflammation show prominent CE,
and may contain intensely CE granulation tissue
-T1-high inner cyst wall due to free radical is specific for abscess
-Lipid peak is observed in abscess due to enhanced glycolysis and necrosis
-Acetate (Ace) and Succinate (Suc) peaks are observed in abscess w/ anaerobic infection,
and may be helpful for the selection of appropriate antibiotics

Fig. 38: Differential diagnosis of ovarian cystic masses:
Tumor-like lesions w/ characteristic morphological appearances:
Cystic mass containing benign solid portion:
Abscess w/ granulomatous tissue
Benign cystic tumor w/ characteristic morphological appearances
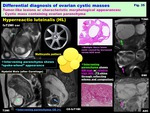
-Mature cystic teratoma w/ dermoid nipple: ( Fig. 39 )
-Most common benign ovarian neoplasm
-Derives from the primitive germ cells of the embryonic gonad
-Detecting fat is diagnostic
-Benign Rokitansky protuberance (dermoid nipple) in mature cystic teratoma is covered with whitish cheese-like material (desquamated,
degenerated epithelium) and may show DW-very high mimicking malignancy reflecting viscous materials
-CE due to epithelial elements may be observed,
however,
massive,
nodular,
or Cauliflower-like CE are suggestive for malignant transformation

Fig. 39: Differential diagnosis of ovarian cystic masses:
Benign cystic tumor w/ characteristic morphological appearances:
Mature cystic teratoma w/ dermoid nipple
-Struma ovarii w/ T2-low colloid material: ( Fig. 40 )
-Composed of mature thyroid tissue and accounts for 3% of mature teratomas
-Multi-lobulated complex mass with various signal as "stained glass appearance”
-Colloid cysts within the tumor may show CT-high and T2-very low
-Solid portion show intense CE / DW-low to intermediate like thyroid glands or struma

Fig. 40: Differential diagnosis of ovarian cystic masses:
Benign cystic tumor w/ characteristic morphological appearances:
Struma ovarii w/ T2-low colloid material
Malignant cystic tumor w/ characteristic morphologic appearances
-Borderline to malignant mucinous tumor w/ aggregated tiny loculi: ( Fig. 41 )
-Aggregated tiny loculi in borderline to malignant mucinous tumors
-Aggregated tiny loculi filled with very viscous fluid in mucinous tumors may appear as T2-low /DW-slight high /CT-soft tissue density solid components
-Reticular CE pattern formed by CE tiny septa with unenhanced cystic spaces is characteristic
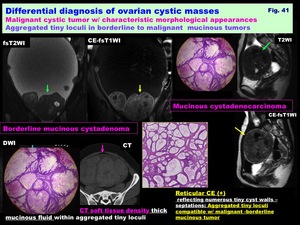
Fig. 41: Differential diagnosis of ovarian cystic masses:
Malignant cystic tumor w/ characteristic morphological appearances:
Aggregated tiny loculi in borderline to malignant mucinous tumors
Malignant cystic tumor: DWI and DCE-MRI features: ( Fig. 42 )
-Malignant solid components show DW-very high with low ADC due to hypercellularity,
and intense early CE reflecting angiogenesis
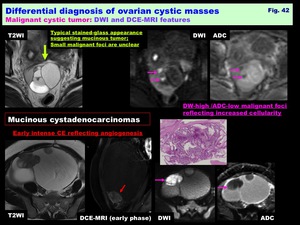
Fig. 42: Differential diagnosis of ovarian cystic masses:
Malignant cystic tumor: DWI and DCE-MRI features