Patients
Seventy-five patients (34 women and 41 men; mean age 58.9 yrs),
with sellar/parasellar tumors were prospectively enrolled in the study.
The histopathological examinations obtained after resection of these tumors revealed: 46 macroadenomas,
12 meningiomas (including 2 meningiomas in an intrasellar location and 10 meningiomas in a parasellar location),
10 craniopharyngiomas: 6 adamantinomatous type (4-intrasellar,
2-suprasellar),
4 squamous-papillary type (suprasellar),
as well as 1 intrasellar hemangioblastoma,
3 intrasellar metastases,
2 suprasellar lymphomas and 1 suprasellar glioma.
Among the adenomas there were: 8 prolactin-secreting adenomas,
1 growth hormone-secreting adenoma and 37 nonfunctioning adenomas.
The craniocaudal diameter of tumors included in our study ranged between 1.4 cm and 6.0 cm (mean 2.9 cm).
The study was conducted in accordance with the guidelines of the local University Ethics Committee for conducting research involving humans.
Each patient provided his/her signed consent to participate in the examination.
Imaging protocol
Imaging was performed on a 1.5T Signa Hdx scanner (GE Healthcare,
Medical System) using a 16-channel coil dedicated for head and spine imaging.
Conventional sequences included: coronal and sagittal T1 FSE,
coronal and sagittal T2 FSE,
and post-contrast T1-weighted images in coronal and sagittal planes (slice thickness 3 mm).
Additionally,
axial T2-weighted images covering the whole brain were performed.
Axial DWI EPI (echo planar imaging) was performed with a b value of 0 and 1000; TR = 8000,
TE = 81.5; FOV= 26x20,8; section thickness 4 mm (with no gap).
PWI was performed with a dynamic susceptibility-weighted contrast-enhanced (DSC) method,
using a gradient-recalled T2*-weighted echo-planar imaging sequence.
Parameters used were as follows: TR/TE = 1900/80 ms,
flip angle = 80°,
NEX = 1.0,
matrix size = 192 × 128,
and slice thickness = 5 mm (with no gap).
Perfusion images were obtained in axial slices parallel to anterior commissure – posterior commissure (AC-PC) line.
During the first 10 seconds images were acquired before starting the contrast agent injection to establish a pre-contrast baseline.
Ten seconds after the start of image acquisition,
0.2 mmol/kg of body weight gadopentetate dimeglumine was injected with a power injector (Medrad) at a rate of 5 mL/s through an intravenous catheter placed in the antecubital vein.
This was immediately followed by a bolus injection of saline (total of 20 mL at 5 mL/s).
Total duration of acquisition was 1 minute and 24 seconds.
Image post-processing
Post-processing of perfusion data and perfusion measurements was performed using the commercial GE Healthcare workstation (ADW 4.4).
Diffusion Measurements
Regions of interest (ROIs) were drawn on the grey-scale ADC maps.
Multiple ROIs (size 30–40 mm2) were placed over several dark spots,
and the minimum ADC values of all ROIs were chosen.
Additionally for each transaxial section,
the aligned imaging data set was used to manually draw a single ROI around the entire region of tumor.
Mean ADC values for each transaxial section of tumor were calculated.
For normalization,
another ROI with a size of approximately 30–50 mm2 was placed in the contralateral normal-appearing white matter,
carefully excluding gray matter.
The rADC ratio was then obtained by dividing the lesion ADC by the values obtained from the contralateral normal-appearing white matter.
Perfusion Measurements
To obtain relative (normalised) values,
all perfusion measurements from the lesions were divided by values from the normal appearing white matter in the right hemisphere.
Measurements of cerebral blood volume (CBV)
To obtain mean CBV from the whole tumor we used irregular hand drawn regions of interest (ROIs) outlining the tumors margins on each axial section.
The mean tumor CBV was calculated as the mean value from all those ROIs.
On the other hand,
to obtain maximum CBV values,
several small round ROIs (size 30–40 mm2) were used.
These ROIs were placed over several hot spots on color-coded CBV maps and the ROI with the highest CBV value was chosen as the maximum CBV.
T1- and T2-weighted images and raw data of perfusion images were used to ensure that regions of interest did not include any hemorrhage or apparent blood vessels.
For the best tumor localization we also used CBV maps overlaid on post-contrast T1 images.
In craniopharyngiomas only regions without calcifications were evaluated.
Due to large,
diffused calcifications in one craniopharyngioma to assess CBV measurements,
only a small ROI method was applied.
In this case mean values from the whole tumor were not possible to assess because of artifacts distorting mathematical measurements and perfusion curves.
Measurements of percentage of signal intensity recovery (PSR) and peak height (PH)
The T2*-weighted signal-intensity curves obtained for the ROIs with max.
CBV and mean CBV were analyzed.
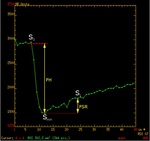
For each perfusion curve 3 major points were established: S0 – a baseline T2*-weighted signal intensity before contrast arrival,
Smin – the highest drop in T2*-weighted signal intensity after contrast arrival (minimum T2*-weighted signal intensity),
S1- T2*-weighted signal intensity recovery after 24 seconds.
The PSR was calculated using the following equation: PSR = (S1-Smin)/(S0-Smin), while PH was calculated with the following equation: PH = S0-Smin (Fig.3).
Statistical analysis
Perfusion measurements: relative cerebral blood volume (rCBV),
relative peak height (rPH),
relative percentage of signal intensity recovery (rPSR) from adenomas and meningiomas were compared using the Student T-test.
Additionally sensitivity and specificity of PWI in distinguishing adenomas from meningiomas were assessed for fixed cut off values of the perfusion parameters showing the most significant differences between these two groups of tumors.
Statistical computations were performed using the Statistica PL software package version 10.0,
and the p < 0.05 was set as a significant level.
Due to the small number of other tumor types statistical analysis between them was not possible.