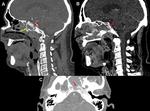
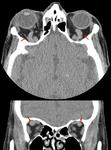
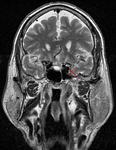
CT and MRI are the preferred non-invasive imaging techniques in diagnostic approach of CCF.
CT scan can depict proptosis,
enlargement of the extraocular muscles,
dilatation and tortuosity of ophthalmic veins (mainly superior ophthalmic vein -SOV-),
and enlargement of the ipsilateral cavernous sinus,
with bulging of its lateral wall.
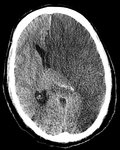
A noncontrast cranial CT scan must be done in order to search skull fractures or intracranial haemorrhages [3].
MR images show similar findings to those seen on CT; also depicts orbital edema,
enlarged pituitary gland (due to venous congestion) and abnormal flow voids in CS that correspond to shunted blood.
Furthermore,
is useful in screening and follow-up.
In the setting of a high-flow fistula and retrograde cortical venous reflux,
MR or CT studies may reveal dilatation of leptomeningeal and cortical veins [7,
9,
10].
However,
CT and MRI cannot exclude totally the diagnosis,
and therefore cerebral angiography is required to confirm CT/MRI findings prior to treatment.
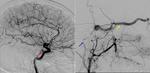
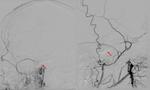
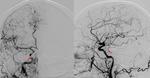
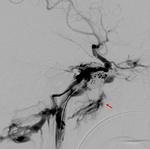
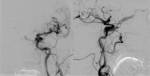
Cerebral angiography is the gold standard for diagnosis,
classification (depicting the anatomy and estimating shunt dynamics) and treatment in CCF [3,
7,
9,
10].
Digital substraction angiography (DSA) is used to obtain the following information:
- Size and location of the fistula.
- Characterize them as direct or indirect.
- To identify associated cavernous carotid aneurysms.
- Presence of complete or partial steal phenomena.
- Identification and confirmation of patency of outflow pathways of the CS.
- Assessment of cortical arterial circulation and collateral flow through circle of Willis.
- Identification of high-risk features (cortical venous drainage,
pseudoaneurysm,
CS varix).
- To depict venous drainage patterns,
therapeutic route,
associated vascular injuries and evaluation of carotid bifurcation before compression therapy.
Endovascular treatment options
In the last years,
it has been developed several therapeutic options in management of CCF.
The endovascular approach has become the first-line option in clinical emergencies,
being used in symptomatic direct CCF´s and indirect fistulas with progressive symptoms (visual loss,
dangerous drainage pathways) [11].
The treatment is chosen according to the type and anatomy of the fistula,
size of arterial defect and operator/institution preferences.
The goal of treatment in direct CCF is to occlude the tear betweenICAand CS,
preserving the patency of ICA,
whereas in indirect CCF the goal is to interrupt fistulous communications and reduce CS pressure [3].
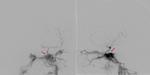
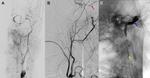
- Balloon Occlusion: This procedure requires that the CS must be large enough to put the balloon for embolization and the size of fistula must be smaller than the inflated balloon,
but large enough to allow a deflated balloon.
The balloon has the advantage of being able to be flow-directed through the fistula and CS,
and must be inflated to a volume larger than the fistula orifice to prevent its retrograde migration into ICA [3].
Later,
angiography is repeated to ensure closure of the fistula and patency of the ICA.
- Transarterial embolization: In the last years it has become the mainstay of treatment in high-flow direct CCF´s [12,
13].
Also,
it’s an alternative when residual AV shunt remains in dural CCF.
Embolization can be made with detachable platinum coils and liquid embolic agents (n-butyl cyanoacrylate,
ethylene-vinyl alcohol copolymer); the first are preferred because of their reliable and controlled deployment into CS.
Complications of this procedure includes thromboembolus and ICA dissection [7].
- Covered stent graft placement: Can be useful to obliterate direct CCF´s,
while preservates ICA patency.
Actually their use is limited.
- Transvenous embolization: Is the current method of choice in treatment of indirect CCF´s.
the goal of this technique is to catheterize the abnormal CS superselectively and occlude the fistula without rerouting venous drainage to cortical structures [7,
9,
10].
This can be made in several routes; the most used is through inferior petrosal sinus (IPS) [9,
10,
14].
There are other alternative routes,
including facial vein and SOV,
trans-contralateral CS,
superficial middle cerebral vein and sphenoparietal sinus,
pterygoid plexus and direct transorbital puncture of CS via the superior orbital fissure.