Imaging workup of the Male Breast
Radiologically,
3 patterns of gynaecomastia are described,
namely the nodular pattern (Figure 2),
the dendritic (Figure 3,
Figure 4) and the diffuse glandular (Figure 5,
Figure 6) patterns.

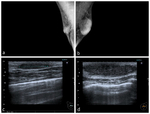
Fig. 2: 41 year old gentleman referred to clinic for work up of gynaecomastia. Patient awaiting liver transplant for liver cirrhosis secondary to Hepatitis C.
Bilateral asymmetrical nodular gynaecomastia on MLO views (a,b). Ultrasound demonstrates hypoechoic retroareolar areas surrounded by fatty elements (c,d). No suspicious masses identified. The nodular phase is described as the early phase of gynaecomastia.
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK

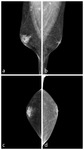
Fig. 3: 82 year old gentleman referred for imaging of a lump in the right breast – Clinical grade 3
Unilateral dendritic gynaecomastia on Right MLO and Right CC views (a,c). The left breast is normal (c,d).
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK

Fig. 4: Ultrasound, of same patient in Figure 3, demonstrates a wedge-shaped opacity with its apex in the subareolar region with irregular margins posteriorly extending into echogenic fibrous breast tissue. In view of P3 clinical grading, patient’s age and unilaterality, ultrasound guided tissue sampling was, at the time, performed confirming gynaecomastia.
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK

Fig. 5: 54 year old gentleman presenting with a 6 month history of left breast swelling and tenderness – Clinical grade 2
Diffuse glandular pattern of gynaecomastia bilaterally on MLO and CC views (a,b,c,d).
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK

Fig. 6: Ultrasound of same patient in Figure 5 confirms diffuse glandularity (a,b,c). Hyperechoic fibrous breast tissue (*) arranged as per sonographic assessment of the female breast.
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK
Other benign breast lesions may be demonstrated in the male breast such as cutaneous conditions,
fat necrosis,
cysts (Figure 7),
abscesses and intramammary lymph nodes may also occur in the male breast.

Fig. 7: 17 year old young man noticed to have a growing lump in the left breast.
Simple cysts – anechoic, thin-walled and demonstrating through transmission – measuring up to 11mm in the lateral part of retroareolar gynaecomastia/glandular elements. No solid, complex elements demonstrated.
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK
The workup and imaging modality of choice for assessment of the male breast varies – with some centres opting for mammography first-line,
and targeted ultrasound being performed as an adjunct assessment when required.
Other breast units opt for ultrasound assessment of the male breast.
Barkeji et al [8] identified that out of 142 male patients imaged over a twelve month period,
44% had a biopsy performed and 5 were found to have a malignancy. 124 of these patients had imaging findings which were classified as normal or benign and 45 of these individuals had a biopsy. None of these biopsies were malignant or altered management.
Correlation of imaging and clinical findings may safely reduce the number of unnecessary breast interventions performed in these individuals.
It is found that,
in both female and male breast cancer,
the biggest risk factor is age.
Nonetheless,
as is true for much of medicine,
a “one size fits all” universal model is not necessarily appropriate or safe. Men younger than 40 years of age – with clinical gynaecomastia – may not need imaging assessment. If imaging is requested,
ultrasound may be considered sufficient. Patients over the age of 40 years may be initially assessed mammographically with a near 100% specificity when both clinical examination and mammographic findings are benign and hence no further assessment (tissue biopsy) warranted [3].
Correlation with clinical findings and any initial imaging findings should guide triple diagnostic assessment as required. Making use of a standardized reporting breast imaging classification is useful to communicate findings with referrers and also outline the level of suspicion for malignancy. The Royal College of Radiologists Breast Group formulated a national classification for use by Breast Imagers in the UK [9].
The main contrasting element between this system and the American College of Radiologists BI-RADS classification lies in Category 3 classification: BI-RADS deems lesions in this group to be most likely benign but recommends follow-up imaging in 6 months than annually for a year or two. The RCRBG’s classification defines category 3 lesions as indeterminate/probably benign but necessitating further investigation (Table 2).

Table 2: RCRBG and BI-RADS Imaging Classification Systems Compared
References: Adapted from: Maxwell A, Ridley N, Rubin G et al. (2009) The Royal College of Radiologists Breast Group breast imaging classification. Clin Radiol 64(1):624‑627
Male Breast Cancer
Male breast cancer is uncommon representing circa 0.6% of all breast cancers.
It constitutes less than 1% of all malignancies in men [10] and male breast cancer has an annual incidence in Europe of 1 in 100000 [5]. In 2012,
the National Cancer Institute in the US,
estimated 226,870 newly diagnosed female breast cancers and 2190 newly diagnosed male breast cancers.
Several risk factors for male breast cancer have been identified (Table 3)[11].
Although some of these risk factors may also be those for gynaecomastia,
the latter does not infer an increased risk of breast cancer development. Gynaecomastia is a much commoner encountered clinical entity. In 20% of men with breast cancer there is a family history of breast cancer in first degree female relatives with mutations of BRCA2 being commonest.

Table 3: Risk factors for Male Breast Cancer
References: Adapted from: Ruddy KJ, Winer EP. (2013) Male breast cancer: risk factors, biology, diagnosis, treatment and survivorship. Annals of Oncology 24:1434-1443
The peak incidence of male breast cancer occurs a decade or so later than does female breast cancer – with patients presenting at a median age of 67. Patients tend to present at a more advanced stage of disease. This may be due to less breast awareness in male patients and a relative lack of clinical suspicion by physicians [10]. The clinical presentation is typically that of a nontender eccentric subareolar mass.
(Figure 8 and Figure 9).

Fig. 8: 75 year old gentleman presenting with a 4 month history of a lump in the right breast. Clinical grade 3/4. Father had breast cancer at age 75.
Mammogram demonstrates a well-defined mass and some variable calcification at 9 o’clock in the right breast. Background gynaecomastia noted bilaterally. Right breast (a,c): M4. Left breast (b,d): M2.
Ultrasound guided core performed: G3 Invasive Ductal carcinoma. Axilla normal at ultrasound assessment.
At surgery, G3 IDC confirmed with associated high-grade DCIS. No lymphovascular invasion. Normal axillary nodes.
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK
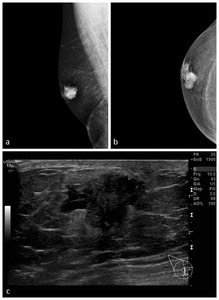
Fig. 9: 42 year old male presenting with a right P3 lump.
Right MLO and CC views (a,b) demonstrate an irregular mass with microcalcifications behind and superomedial to the nipple: M5.
Sonographically a U5 irregular hypoechoic mass (c) is demonstrated. Core biopsy performed. Histology: Grade 2 Invasive Ductal Carcinoma. Macrometasis demonstrated at surgery - pT2,pN1
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK
Also,
the less standardized use of breast imaging in male patients implies a lack of familiarity with the imaging characteristics of male breast cancer.
Pathologically,
the vast majority of breast cancer in males is invasive ductal carcinoma. Ductal carcinoma in situ is not frequently diagnosed as most patients present with a symptomatic lump as opposed to screen detected findings.
Male breast tissue is predominately composed of undeveloped ductal components encircled by connective,
adipose,
and subcutaneous tissues. Lobular carcinoma is therefore rare. Simple breast cysts are uncommon and complexity must be excluded as neoplastic papillary lesions may manifest as complex cystic lesions. Fibroadenomas are also very rare in men – as these are also of lobular origin [12,13].
The imaging appearances of breast carcinoma in males are variable. Even well-defined lesions – which perhaps would be considered benign in the female breast (Figure 10) – merit further investigation in the male breast.

Fig. 10: 87 year old male presenting with a 2cm firm, not very mobile lump in the left breast and vague skin dimpling – P4.
Bilateral MLO views only performed (a,b) – Left M4 opacity. Ultrasound demonstrated a well-defined, hypoechoic, gently lobulated lesion with through-transmission. In the female breast this would not be regarded as highly suspicious (M3) however in a male this is still most in keeping with a carcinoma (M4) - US guided biopsy performed confirming Grade 3 Invasive Ductal Carcinoma.
References: The Park Centre for Breast Care, Brighton and Sussex University Hospitals, Brighton, UK
NICE guidance for Familial Breast Cancer [14] outlines that individuals with a first degree male relative with breast cancer should be offered referral to Secondary Care. Referral to a Specialist Genetic Clinic may be recommended if there are additional family members with breast cancer and a male relative with breast cancer.
The diagnosis of a male breast cancer – rare as it may be – therefore has broader repercussions.