Numerous criteria are used to select the technique including: a) age; b) physical condition; c) intestinal,
hepatic,
and renal function; d) tumor stage; e) previously abdominal radiation therapy.
There are several complex reconstructive procedures associated with radical cystectomy; in this work we will address the most commonly performed.
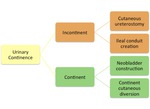
They can be divided according to functional result (figure 1).

Fig. 1: Types of urinary diversion procedures according to functional result.
The percentage of urinary diversion procedures performed are: neobladder 47%,
ileal conduit 33%,
continent cutaneous diversion 8% and incontinent cutaneous diversion 2%.
1) Incontinent diversion
1.1) Cutaneous ureterostomy (Figure 2)
Indications: impossibility to use an intestinal segment - patients with bowel disease or serious coexisting medical condition.
Procedure: Both ureters are anastomosed directly to the anterior abdominal wall.
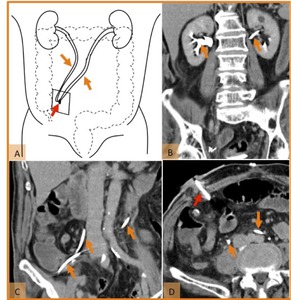
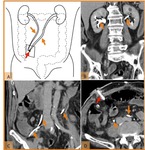
Fig. 2: Normal postoperative anatomy after radical cystectomy and cutaneous ureterostomy. (A) Drawing illustrating both ureters (orange arrows) anastomosed directly to the anterior abdominal wall (red arrow). (B,C) Coronal reformatted CT image and (D) axial CT scan, on the excretory phase, demonstrate both ureters route (orange arrows) and their surgical anastomoses to the right anterior abdominal wall (red arrow) in a patient with bladder trauma.
References: Hospital de São João, Porto
1.2) Ileal conduit creation (figure 3)
Procedure:
1) An ileal segment (≈15–20 cm) is isolated and both ureters are anastomosed to its proximal end.
The ureters may be anastomosed to a proximal end segment of the bowel in a separate (Bricker procedure) or conjoined (Wallace procedure) fashion.
2) The distal end provides urinary drainage to a cutaneous stoma to which an ostomy bag is attached for urine drainage.
The ileal stoma is usually located in the right flank.
An ileal segment,
proximal to the ileocecal junction,
is preserved to maintain adequate absorption of bile salts,
vitamin B12,
and fat-soluble vitamins.
Advantages:
1) Technically easier to perform than continent reconstruction – ideal for patients with medical comorbidities to decrease perioperative complications;
2) Short gut diverting segment limits enteric absorption of urine waste products,
reducing metabolic abnormalities – method of choice for patients with renal insufficiency or hepatic dysfunction;
3) There is no need for occasional catheterization (use of a tube to drain the urine).
Disadvantages:
1) Loosening of continence and voluntary voiding;
2) Body image is not preserved;
3) Requires external appliance and stoma care;
4) Possible stomal complications (including hernias,
stenosis and bleeding/skin irritation).
It is,
also,
important to evaluate the enteroenteric anastomosis,
which is also placed to the right side because of possible complications related to the suture.

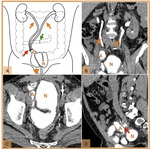
Fig. 3: Normal postoperative anatomy after radical cystectomy and cutaneous ileal conduit construction. (A) Drawing illustrating both ureters (orange arrows) anastomosed (red arrow) to the proximal end of an ileal segment (c) which was previously isolated (green arrow). The distal end of the ileal segment provides urinary drainage to a cutaneous stoma (orange arrowhead). (B,C) Coronal CT reformatted images and (D, E) axial CT scans, on the excretory phase, demonstrates ureters route (orange arrows), uretero-ileal anastomosis (red arrows), ileal conduit (c) and a cutaneous stoma (orange arrowhead) to which an ostomy bag (B) is attached for urine drainage. Note metallic suture material (green arrows) in the ileo-ileal anastomosis and the surgical clips from lymphadenectomy (white arrow), in a patient with bladder cancer.
References: Hospital de São João, Porto
2) Continent diversion is the preferred method for young patients with a good prognosis.
Advantages: Allows preservation of body image and a greater quality of life.
Principals: create a reservoir capable of storing a proper volume of urine,
using the shortest possible segment of bowel with an acceptable level of continence.
Selecting the bowel segment:
Advantages small bowel reservoir: metabolic acidosis has less impact with ileal reservoir than with a colonic or jejune reservoir; small intestinal reservoirs can accumulate higher volumes of urine with lower pressures than colonic; postsurgical complications with colon are severer and more difficult to resolve than with the other intestinal segments.
Advantages sigmoid colon: easy access.
2.1) Orthotopic Bladder Replacement (neobladder construction) – The use of ileal neobladder is becoming the preferred procedure with the percentage of patients receiving neobladder increasing to 50-90% (figure 4).
Procedure:
1) Cystectomy,
lymphadenectomy,
and preparation of the urethra;
2) Ileal segment (≈50 cm long) is detubularized to construct an intestinal reservoir:
- Ileal segment (≈40 cm long) is used to create an intestinal reservoir (pouch);
- An isoperistaltic afferent limb is created using a proximal ileal segment (≈10 cm) (normally is located to the right of the reservoir) to reflux prevention;
3) A small opening is made in the distal part of the pouch,
which is then anastomosed to the native urethra;
4) Bowel continuity is restored with ileal anastomosis;
5) Ureters are anastomosed to the afferent limb.
Advantages: voluntary voiding is preserved which improves quality of life: no stoma or external collecting devices,
with preservation of the patient’s body image.
Disadvantages:
1) Longer surgical time;
2) Need for a longer bowel segment for neobladder construction;
3) Requirement of functional urethra or negative urethral margins;
4) Delay before optimal neobladder function is achieved,
requires active patient training/participation.
Contraindications (Table 1)

Table 1
References: adapted from: Nagele U, et al. Urinary Diversion Following Cystectomy. EAU Update Series 2005; 3:129–137.
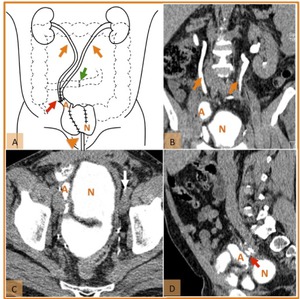
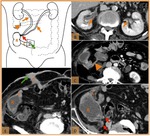
Fig. 4: Normal postoperative anatomy after radical cystectomy and neobladder construction. (A) Drawing illustrating both ureters (orange arrows) anastomosed (red arrow) to the proximal end of an afferent ileal segment (orange A). The intestinal reservoir (pouch - N) created with an ileal segment is anastomosed, in the distal part, with the native urethra (orange arrowhead). Note the ileo-ileal anastomosis (green arrow). (B) Coronal and (D) sagittal reformatted CT images; and (C) axial CT scan, on the excretory phase, demonstrates ureters route (orange arrows), the ileal afferent limb (orange A), uretero-ileal anastomosis (red arrow) and the intestinal reservoir (N) in a patient with bladder cancer. Note surgical clips from lymphadenectomy (white arrow).
References: Hospital de São João, Porto
2.2) Continent cutaneous diversion (Indiana pouch) – currently is not frequently performed (figure 5).
Procedure:
1) A segment of the large or small bowel is used to construct a reservoir;
2) The ureters are anastomosed to the proximal end;
3) A cutaneous stoma is created,
and urine is eliminated during self-catheterization performed by the patient.
Advantages:
1) Enables continence with no need for external appliance;
2) Body image is preserved.
Disadvantages:
1) Voluntary voiding is not possible;
2) Requires patient motivation/education regarding lifelong self-catheterization every four to six hours using sterile technique.
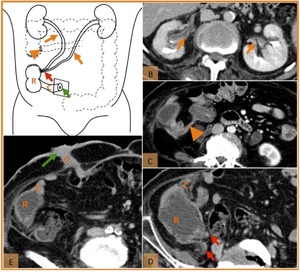
Fig. 5: Normal postoperative anatomy after radical cystectomy and continent cutaneous diversion with right colonic pouch. (A) Drawing illustrating both ureters (orange arrows) anastomosed (red arrow) to right colonic pouch used to construct a reservoir (R). An intestinal segment (c) provides urinary drainage from the reservoir to the cutaneous stoma (green arrow). Note the ileo-colic anastomosis (orange arrowhead). The urine is eliminated during self-catheterization performed by the patient. (B,C,D,E) Axial enhanced CT images demonstrate ureters route (orange arrow), ileo-colic anstomosis (orange arrowhead), uretero-colic anastomosis (red arrows), colonic reservoir (R) and an ileal conduit (c) that provides dranaige to a cutaneous stoma (green arrow), in a patient with a neurogenic bladder.
References: Hospital de São João, Porto
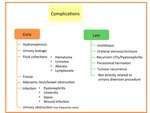
Complications
Complications after urinary diversion procedures are common because of the technique complexity.
They can be divided in early (≤30 days after surgery) or late (≥ 30 days or more after surgery) (Figure 5).
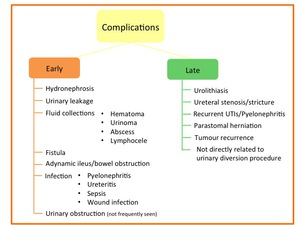
Fig. 6: Early and late complications after urinary diversion procedures.
(A) Early complications (Figure 6)
(A.1) Hydronephrosis (figure 7)
- Is not necessarily indicative of an urinary tract obstruction.
Some degree of hydronephrosis is common finding after urinary diversion either due to prior obstruction or to reflux at the ureteral anastomoses.
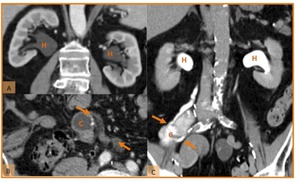
Fig. 7: Hydronephrosis after radical cystectomy. (A) Coronal CT reformatted image, (B) axial CT scan and (C) coronal CT reformatted image in two different patients with reconstruction with cutaneous ileal conduit. These images demonstrate hydronephrosis (H), without any evidente of stricture or obstruction. Note the ureters route (orange arrows).
References: Hospital de São João, Porto
(A.2) Urinary leakage (figure 8)
- Occurs in approximately 4% of patients after urinary diversion;
- The most frequent site of leakage is at the ureteral–reservoir anastomosis.
It must be suspected when there is increased drainage from the catheter or urinary drainage from the wound;
- On CT,
urinary leak may be detected as an extravasation of contrast material into the peritoneal space on the excretory images.
The leakage may emanate from the ileal conduit/neobladder or on the anterior portion of the conduit.
- Treatment includes place a stent over the leakage site to encourage healing of the abnormal communication.
Surgical repair is rarely necessary.
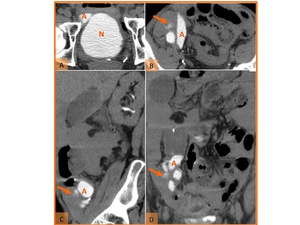
Fig. 8: Urinary leak in a patient with bladder cancer and orthotopic bladder replacement 6 days after surgery. (A, B) Axial, (C) sagittal and (D) coronal reformatted CT images on the excretory phase show contrast extravasation (orange arrows) adjacent to the afferent loop (A) of the neobladder (N) near to uretero-ileal anastomosis.
References: Hospital de São João, Porto
(A.3) Fluid collections
- On CT hematoma is commonly seen as a dense,
heterogeneous,
nonenhancing collection fluid collection that typically decreases in attenuation over time and is usually located in or near the surgical area.
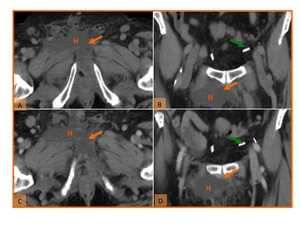
Fig. 9: Hematoma in a patient 5 days after cystectomy and ileal conduit creation for perineal neoplastic mass. (A)Unenhanced axial and (B) coronal reformatted CT images; and (C) axial and (D) coronal reformatted CT images on the nephrographic phase show a nonenhancing heterogeneous fluid collection (H) located in the anterior perineal region and with some spontaneously hyperdense foci compatible with clots (orange arrow). Note also the surgical clips (green arrow).
References: Hospital de São João, Porto
- Happens when the postoperative drainage catheter does not collect the leaked urine.
- Symptoms include: abdominal pain,
signs of peritoneal irritation,
and fever.
- CT excretory phase is essential (10–20 minutes after contrast injection) to make an accurate diagnosis.
Urinoma on CT appears enhanced because of contrast material accumulation.
If the diagnosis remains uncertain,
percutaneous aspiration and analysis of the fluid may be done.
- Treatment: Percutaneous drainage is usually required.

Fig. 10: Urinoma. (A) Axial CT image on the excretory phase, in a patient after radical cystectomy and ileal conduit reconstruction show extravasation of contrast adjacent to the left ureter (orange arrow) into a collection, compatible with urinoma (U). Note the normal right ureter (green arrow). (B) Axial and (C) sagittal reformatted CT image on the excretory phase in a patient with neobladder construction (N), show a fluid collection (U) located between the abdominal wall and the neobladder with opacification with contrast (red arrow) on the excretory phase.
References: Hospital de São João, Porto
- Can occur after lymphadenectomy usually in patients with malignant bladder condition.
- CT findings: homogeneous fluid collection with a very thin wall,
without enhancement,
in a location near to the site of previous lymphadenectomy (near the surgical clips).
- Treatment: large or symptomatic may require percutaneous drainage.
Commonly resolve spontaneously or may be asymptomatic.

Fig. 11: Lymphocele in two patients with lymphadenectomy after radical cystectomy for bladder carcinoma. (A,B) Axial enhanced CT images show a fluid collections with thin walls (L), without enhancement, located near to the lymphadenectomy site (surgical clips - white arrows).
References: Hospital de São João, Porto
- Abscesses (figure 12 and 13)
- Fluid collections such as an urinoma,
hematoma,
or lymphocele may become infected,resulting in an abscess.
- CT findings: Air bubbles in a fluid collection with a thickened and enhancing wall with densification of adjacent fat.
Nevertheless,
attention must take because air bubbles,
may be iatrogenic,
frequently secondary to the placement of a drainage catheter.
- In some cases it may be difficult to differentiate an infected and no infected collections.
Therefore,
if an abscess is suspected,
percutaneous or surgical drainage is usually necessary.

Fig. 12: (A,B) Axial enhanced CT show fluid collections with thickened and enhancing walls (green arrow), associated with densification of the adjacent fat (white arrow) in patients with infected hematoma (H) and urinoma (U).
References: Hospital de São João, Porto
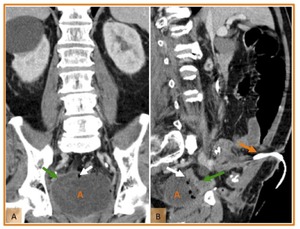
Fig. 13: Abscess 6 days after radical cystectomy for bladder carcinoma and reconstruction with ileal conduit. (A) Coronal and (B) sagittal reformatted enhanced CT images show a heterogeneous fluid collection (A) located in the pelvis with air bubbles (white arrows) and with thickened enhancing walls (green arrow). Note the ileal conduit construction (orange arrow).
References: Hospital de São João, Porto
(A.4) Fistula (figure 14 and 15)
- Types of fistulas seen after urinary diversion surgery are: entero-urinary (in 2.2% of the patients and occur,
most commonly,
at the intestinal anastomosis),
enterogenital,
or enterocutaneous.
- Treatment:conservative management if there is good drainage and no sepsis; otherwise,
surgical revision is required.
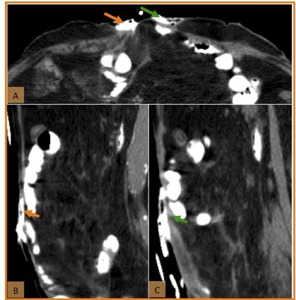
Fig. 14: Enterocutaneous fistula. (A) Axial CT scan and coronal (B,C) reformatted CT images obtained after oral contrast material administration. The images show two enterocutaneous fistulous tracks draining into the surgical wound, one in the left (green arrow) and the other in the right (orange arrow).
References: Hospital de São João, Porto
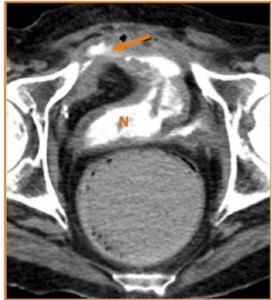
Fig. 15: Axial CT image obtained on the excretory phase in a patient with neobladder construction (N) for neurogenic bladder. The image shows extravasation of contrast throughout an enterocutaneous fistula (orange arrow).
References: Hospital de São João, Porto
(A.5) Adynamic ileus/bowel obstruction (figure 16)
- Bowel function is normally seen within 5 days after surgery.
If normal function is not restored,
the most likely cause is either adynamic ileus or mechanical obstruction.
- Adynamic ileus affects 18%–23% of patients after urinary diversion surgery and is the most usual bowel complication.
CT: uniformly dilated loops of small and large bowel with gas-fluid levels without an evident cause of obstruction.
- Mechanical obstruction (3% of cases): bowel loops with gas-fluid levels are visible proximal to the site of obstruction,
where an abrupt change in intestinal caliber is seen.
Adhesive small-bowel obstruction near to the enteroenteric anastomosis is the most common type.
- Treatment:Partial obstruction and adynamic ileus can be managed conservatively.
On the other hand,
complete or high-grade obstruction requires immediate surgery.
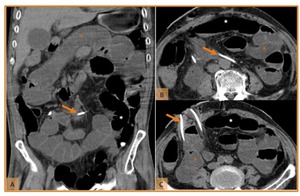
Fig. 16: Adynamic ileus 6 days after radical cystectomy and cutaneous ureterostomy in a patient with neurogenic bladder. (A) Coronal reformatted CT image adn (B,C) axial CT scans show dilated fluid-filled small-bowel loops (orange *) and large bowel, with gas-fluid levels (white *). No identifiable cause of obstruction was found. Note the catheters inside the ureters draining into the skin (orange arrow).
References: Hospital de São João, Porto
(A.6) Infection (figure 17)
- Urinary tract infection may occur as either an early or a late complication.
- CT findings: Pyelonephritis: focally wedge-shaped or striated areas of decreased parenchymal enhancement; loss of normal sharp corticomedullary differentiation; renal enlargement; asymmetric stranding or obliteration of the perinephric fat and thickening of the renal fascias.
Ureteritis: enhancement of the ureter and renal pelvis.
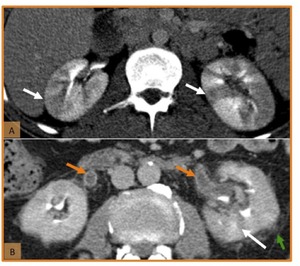
Fig. 17: Pyelonephritis and ureteritis. (A, B) Axial enhanced CT images show striated areas of decreased parenchymal attenuation (white arrows) and loss of normal sharp corticomedullary differentiation in both kidneys (image A) and in the left kidney (image B). Renal enlargement, stranding of the perinephric fat (green arrow) and enhancement of the ureter and renal pelvis walls (orange arrow) are also seen (image B).
References: Hospital de São João, Porto
(B) Late complications (figure 6)
(B.1) Urolithiasis (figure 18 and 19)
- Patients who have undergone urinary diversion have an increased risk of developing urolithiasis,
with an incidence of approximately 10%.
- Stone formation is more common in patients with an ileal conduit.
- The etiology of urinary stone formation is multifactorial (metabolic,
structural,
infectious,
and idiopathic factors) with most stones being primarily of struvite (probably secondary to infection).
- Calculi can be seen within the kidney,
ureter,
and/or ileal conduit/neobladder.
- Unenhanced CT images improve detection of urinary tract calculi.
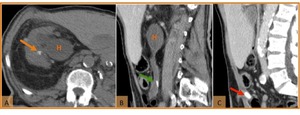
Fig. 18: Urolithiasis 2 years after cystectomy and urinary diversion with ileal conduit construction. (A) Axial and (B,C) sagittal CT images show hydronephrosis (H) and nonobstructive renal calculi (orange arrow) and ureteral calculi (green arrow); an obstructive calculi in the ileal conduit is shown in image C (red arrow).
References: Hospital de São João, Porto
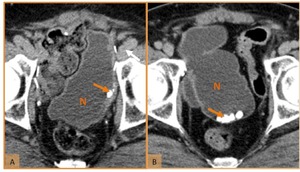
Fig. 19: Axial CT images in two patients after cystectomy and urinary diversion with neobladder construction (N) show calculi (orange arrow) in the reservoir.
References: Hospital de São João, Porto
(B.2) Ureteral stenosis/stricture (figure 20)
- Incidence of ureteral stricture after radical cystectomy varies from 3-10% and occurs most often within the first 1–2 years postoperatively.
- The most common location of ureteral stricture is at the ureteroenteric anastomosis,
with the left distal ureter being at higher risk because of its angulation (the ureter needs to reach the urinary conduit/reservoir usually located in the opposite side of the abdomen).
- Ureteral ischemia,
and subsequent fibrosis,
is the most common cause of stenosis.
Other causes include improperly fashioned anastomosis and recurrence of tumor in the ureter.
- Frequently clinically silent and is commonly detected on the follow-up imaging studies and/or by an increase in the serum creatinine level.
- On CT,
ureteral stenosis is commonly associated with hydronephrosis and delayed contrast excretion.
The ureteral wall may also be thickened,
and it may be difficult to differentiate between benign and malignant strictures.
The latter is usually associated with an enhancing soft-tissue mass on CT (see below); endoscopic and cytologic correlation is necessary to reach the correct diagnosis.
- Treatment includes the use of minimally invasive procedures (balloon dilation or endoscopic incision) and open surgical revision,
the latter with higher long-term success rate (90%).
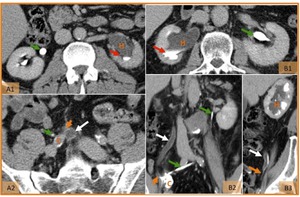
Fig. 20: Benign ureteral strictures in two patients 1 year after cystectomy and ileal conduit diversion for bladder cancer. (A1, A2 and B1) Axial, (B2) coronal and (B3) sagittal CT images on the excretory phase show moderate hydronephrosis (H), dilated ureter on the obstructed side (white arrow) with delayed excretion of the contrast product (red arrow), thickening and tapering of distal ureter near anastomosis (orange arrow) to ileal conduit (C). Note the normal size and excretion of contrast of the opposite ureter (green arrow). Benign cause of strictures was confirmed at ureteroscopy.
References: Hospital de São João, Porto
(B.3) Recurrent UTIs/Pyelonephritis (figure 21)
- Infection may occur as either an early or a late complication.
In patients with an ileal conduit,
bacteriuria was found in approximately 85%,
clinically evident UTI occurred in 23% and urosepsis in 3.8%.
- Pyelonephritis occurs more common in patients with a history of ureteral obstruction or urinary reflux.
- The diagnosis is commonly based on clinical and laboratory findings but typical features of pyelonephritis or perirenal abscess may be observed at CT.
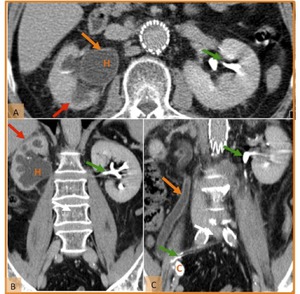
Fig. 21: Recurrent UTIs in a patient submitted to radical cystectomy and ileal conduit diversion (C) for bladder cancer with a benign ureteral stricture. (A) Axial and (B and C) coronal reformatted CT images on the excretory phase show hydronephrosis (H), with diffuse parenchymal thickness reduction (red arrow) indicating a chronic process. These images also demonstrate a thickened and dilated ureter on the right side (orange arrow) indication inflammation. Note the normal size and excretion of contrast of the left ureter (green arrow). An UTI was was confirmed in the analysis of urine.
References: Hospital de São João, Porto
Deterioration of renal function may occur because of recurrent urinary tract infection,
stone formation,
obstruction or reflux.
Imaging (shrunken kidney or hydronephrosis) or functional (serum creatinine greater than 1.7 mg/dL) deterioration was observed in 27% of patients after ileal conduit urinary diversion.
(B.4) Tumour recurrence (figure 22,23,24,25,26 and 27)
- Transitional cell carcinoma (TCC) is the most common primary neoplasm of the bladder and it is often multifocal.
So,
tumour recurrence in the upper tract remains a major concern in patients submitted to radical cystectomy for bladder cancer.
- Tumour relapse may occur locally in the pelvis or manifest as distant metastasis (lymphatic or haematogenous).
- Local tumour recurrence incidence varies from 3% to 16% within 5 years,
depending on the disease stage.
It may manifest with various patterns: a) pelvic soft-tissuemass,
b) an obstructing ureteral stricture (commonly in association with a soft-tissue mass),
c) pelvic lymphadenopathy,
or d) a stricture of the urethro-intestinal anastomosis.
CT urography can be useful in the detection and assessment of upper urinary tract tumours .
At CT tumour is seen as: a) a focal intraluminal mass,
b) urothelial wall thickening with luminal narrowing or/and c) infiltrating mass.
- Up to 70% of the patients with local recurrence also have distant metastasis.
Lymph nodes,
bones,
lung,
liver and peritoneum are the most common sites of distant metastasis from bladder cancer.
- In patients who have undergone pelvic lymphadenectomy for treatment of a bladder tumour,
distant lymphaticnodes such as those in the inguinal and aortocaval regions should always be evaluated.
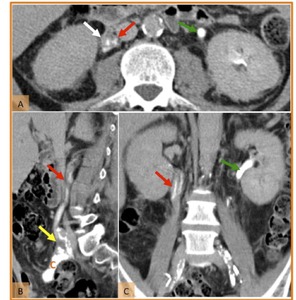
Fig. 22: Tumour recurrence in a patient submitted to radical cystectomy and ileal conduit diversion (c) for bladder cancer. The patient developed hematuria and positive urine cytologies. (A) Axial, (B) sagittal and (C) coronal reformatted CT images on the excretory phase show marked thickening of the right ureter (red arrows) with densification of the adjacent fat (white arrow) and narrowing of the lumen. The figure (B) also demonstrate abnormal thickening at uretero-ileal anatomosis (yellow arrow) which determines ureteral stenosis with filiform passage of contrast. Note the normal size and excretion of contrast of the left ureter (green arrows).
References: Hospital de São João, Porto
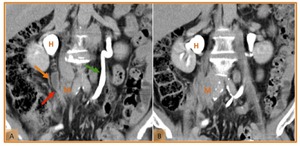
Fig. 23: Tumour recurrence in a patient submitted to radical cystectomy and ileal conduit diversion (c) for bladder cancer. (A and B) Coronal reformatted CT images on the excretory phase demonstrate moderate hydronephrosis (H), dilated right ureter (orange arrow) with an abrupt thickening and tapering of distal ureter (red arrow). This obstructing ureteral stricture is caused by a soft-tissue mass (M). Note the normal size and excretion of contrast of the opposite ureter (green arrow).
References: Hospital de São João, Porto

Fig. 24: Lymph node metastasis in a patient submitted to ileal conduit diversion for bladder cancer. (A and B) Axial enhanced CT images show the presence of pelvic lymphadenopathy (orange arrow) and bilateral inguinal adenomegaly (green arrows). Note ileo-ileal anastomosis (green arrow).
References: Hospital de São João, Porto
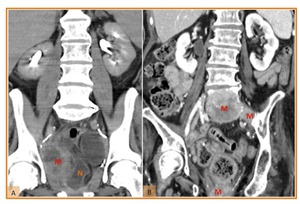
Fig. 25: (A) Coronal reformatted CT image in a patient submitted to radical cystectomy and neobladder construction (N) for bladder cancer. The figure demonstrates a pelvic heterogeneous soft tissue mass (M) representing tumour recurrence. (B) Coronal reformatted CT image in a patient submitted to radical cystectomy and ileal conduit diversion for bladder cancer. The figure exhibits soft tissues masses representing bone metastasis (M).
References: Hospital de São João, Porto
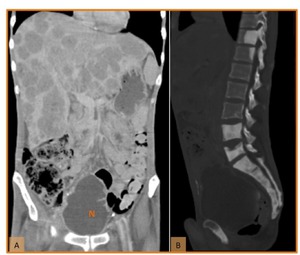
Fig. 26: Liver and bone metastasis in a patient submitted to neobladder construction (N) for bladder cancer. (A) Coronal reformatted enhanced CT image show multiple hepatic hypodense lesions developed during follow-up, compatible with metastases. (B) Sagittal reformatted enhanced CT image demonstrate sclerotic lesions involving multiple vertebral bodies, sacrum and pubic bone, representing bone metastasis.
References: Hospital de São João, Porto
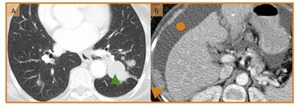
Fig. 27: (A) Axial CT image shows multiple small nodules in both lungs with the larger lesion adjacent to the descending aorta (green arrowhead). These lesions represent pulmonary metastasis. (B) Axial enhanced CT image demonstrates nodular peritoneal implants in the right subdiaphgragmatic space (arrowhead) and ascites (circle) in a patient with recurrence of bladder cancer.
References: Hospital de São João, Porto
(B.5) Parastomal herniation (figure 28)
- Occur in 10%–22% of patients who were submitted to ileal conduit creation.
Obesity may be a contributing factor,
particularly in the elderly patients.
- CT is useful for detecting such hernias in obese patients (in which such hernias are not so easily visible or palpable) and allows the demonstration of any associated complications.
- Most patients are asymptomatic,
however about 10% of these hernias require surgical repair.
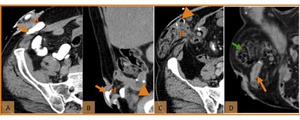
Fig. 28: Paraestomal hernia with no signs of complication in two patients submitted to ileal conduit construction. (A) Axial CT scan and (B) coronal reformatted CT image on the excretory phase show normal small bowel (orange arrow) after parastomal herniation of an ileal conduit (H). (C) Axial and coronal (D) reformatted enhanced CT image show normal small bowel (orange arrow) and colon (green arrow) after parastomal herniation of an ileal conduit (H). Note the location of ileo-ileal anastomosis (orange arrowhead).
References: Hospital de São João, Porto
(B.6) Late complications not directly related to urinary diversion
- Late complications unrelated to urinary diversion procedures and that lead to open surgery were,
most commonly,
incisional hernia (figure 29) (1.5%) and small-bowel obstruction (1.6%) (figure 30).
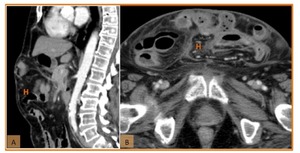
Fig. 29: (A) Coronal reformatted CT show incisional hernia with no signs of complication in a patient submitted to radical cystectomy and ileal conduit diversion for bladder cancer. (B) Axial contrast enhanced CT scan showing strangulated incisional hernia of the anterior abdominal wall in a patient submitted to neobladder construction for neurogenic bladder. Note the bowel wall thickening, severe fat stranding and mesenteric engorgement, findings that suggest strangulation.
References: Hospital de São João, Porto
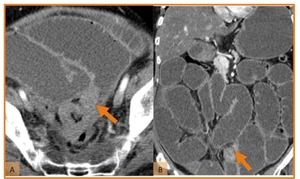
Fig. 30: Small bowel obstruction due to adhesions. (A) Axial and (B) coronal reformatted images showing dilated fluid-filled small bowel loops due to obstruction. Note transition zone (orange arrow), where only collapsed small bowel loops are visualized. The diagnosis of adhesive bands was further confirmed.
References: Hospital de São João, Porto