Introduction:
Under the recent EU directive (Euratom 59/13) it is the responsibility of the practitioner to provide where practicable risk information to the patient prior to the exposure.
It is also the responsibility of the practitioner that the benefit outweighs the risk to the individual and to society. There are a number of risks associated with requested medical radiological procedures other than radiation.
These include the test not being carried out in the first place,
an incorrect or non-contributory diagnosis,
unnecessary or no follow up,
injury from the contrast agent or the test itself.
However radiation risks are the most studied and regulated. The risk will depend on the amount of radiation received and the radio sensitivity of the organ involved. There are two types of radiation risks resultant from radiological procedures: 1.
Deterministic effects such as erythema and hair loss,
which occur above a threshold,
and 2.
stochastic effects which occur at any dose but likelihood increases with dose.
Even in this age of social media and communication the understanding and communication of risk is still difficult and not entirely successful.
One of the reasons for this is that the uncertainties around risk at low level are so large that the radiology community are not able to give a coherent answer to the question of risk from most radiological procedures.
Risks are presented as a range of values,
for example,
in terms of annual background,
bands of radiation symbols.
These are based around typical doses for the procedures.
In recent years auditing and dose tracking have shown that a percentage of exams have very high doses for patients where either deterministic or cancer risk are meaningful.
Quantitative risk assessments based on actual doses to patients combined with individualised risk assessment are becoming more popular with the use of these software tools.
These quantitative risk assessments may be carried out for a number of reasons:
- Investigating an incident
- Advising at-risk groups such as pregnant women or parents about the risk to a foetus or child from their procedures as required by national regulations
- Setting up a research study or new procedure
- Establishing risks from practices within your hospital
This paper looks at the methodology to do such risk assessments,
carries out a number of worked examples and looks at the usefulness of their results.
Materials and Methods:
In conventional risk assessment a scale of probability (Scale of 1-5) multiplied by severity (Scale of 1-5) is used to populate risk registers.
In stochastic radiation risk assessment such a tool may not be appropriate as the probability does not fit in with such a confined subjective scale.
This paper gives a number of worked examples of quantitative risk assessment based on real doses from our hospital group.
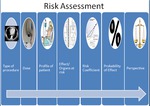
A number of steps are used in quantitative risk assessment.
These include: establishing the dose from the procedure,
profiling the patient,
determination of the organs involved and potential effects,
establishing the probability of the effect occurring from risk coefficients or threshold,
contextualising the risk in terms of other risks,
see Fig 1.
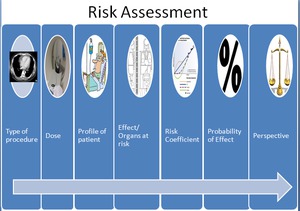
Fig. 1: Outline of the typical steps of a risk assessment
Each of these steps has uncertainty associated with it. In some cases the lack of certainty has led to questioning of the validity of quantitative dose and risk assessment,
especially at low levels.
Two approaches exist; the first estimates the risk from an organ dose, the second establishes a whole body dose and applies a risk coefficient.
In the case studies presented here,
the dose information was returned from the DICOM information direct from the modality or returned from dose tracking systems.
For the interventional cardiology systems and for one of the CT scans,
Care Analytics (Siemens,
Erlangen) was used to access procedure information.
Organ dose estimates were also generated from the Impact CT dose calculator.
The skin dose estimate was based on that outlined in Miller et al.
(2003).
For the high dose CT outlier,
Radimetrics (Bayer,
Berlin) generated the scan information and size specific organ dose estimation.
Risk coefficients and probability were generated from a number of sources based on Beir VII. For organ doses lifetime risks can be calculated using the Radiation risk calculator from the National Cancer Institute in the US.
Pregnancy risks are based on Japanese Bomb survivor data and similar cancer induction risks to neonates of 8% per Gy or Sievert (Wakeford & Little,
2003).
Results:
Results are presented in figures 2 - 6 which outline a number of worked examples.
The risks from the scans are put in perspective by relating to the annual background radiation dose in Ireland of 4 mSv and linking the percentile risk range to the Calman (1996) risk scale.
Worked example 1:
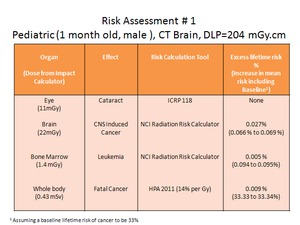
Fig. 2: Worked example 1: Risk assessment for paediatric CT Brain
Worked example 2:
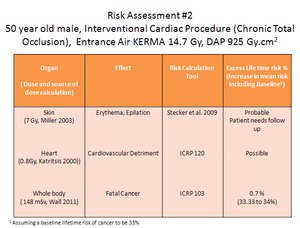
Fig. 3: Worked example 2: Risk assessment for an interventional cardiac procedure
Worked example 3:
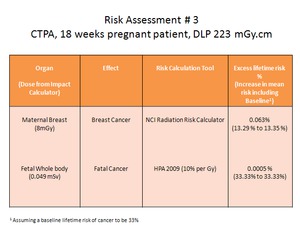
Fig. 4: Worked example 3: Risk assessment for CTPA of 18 week pregnant patient
Worked example 4:

Fig. 5: Worked example 4: Risk assessment for pregnant patient, 8 weeks gestation, CT pelvis
Worked example 5:
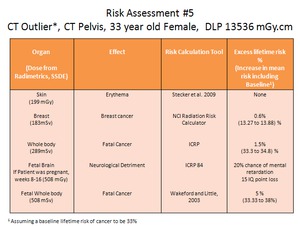
Fig. 6: Worked example 5: Risk assessment for a high dose CT pelvis. This is considered an outlier as it is approximately 15 times the DRL. It should be noted that these outliers constitute <0.5% of the total CT exams.
Discussion:
Quantitative risk assessment is possible using existing information and tools. Such risk assessments provide useful information to patients and referring physicians in following up on deterministic effects and the risk of stochastic effects.
The effect of low doses on large patient populations is not fully understood in terms of absolute number of cancers induced by medical radiation.
In the absence of such knowledge either overplaying or under estimating the risks may have serious public health consequences by denying access to diagnostic tests or treatments or exposing patients to unnecessary risk. In the meantime,
the radiology community should encourage radiation risk research and maintain the optimum risk benefit ratio using dose optimisation and quality assurance programs. Hospitals need access to individuals such as medical physicists,
who can carry out meaningful quantitative dose and risk assessment.
These individuals must also be aware of the limitations of such risk assessments. In most cases the risk assessment shows that the risks to the individual are small,
even using a conservative linear no threshold model for stochastic effects.
For typical CT scans there are no risk of deterministic effects.
If scans are carried out within normal ranges foetal risk is not significant.
Dose tracking systems have shown there are small numbers of high dose scans where individual risks are meaningful. There may be a case for an alerting level for CT scanning suggested by radiation protection initiatives such as Eurosafe.
Exceeding the alerting level would require justification.
Risk assessment in high dose interventional procedures is necessary to identify patients who require follow up for potential deterministic effects.
Analysis of incidents we have come across in our institutions have shown that in some cases these can be reduced by more judicious use of safety technology such as automatic exposure control.
In these outliers (which comprise <0.5% of total exams),
comparison to background or number of cigarettes smoked,
which is often used for risk comparison,
is not meaningful. Risk mitigation should require extra alerts for these outliers.
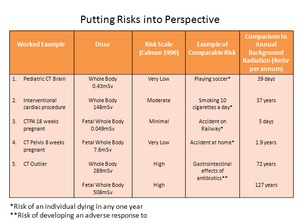
Fig. 7: Putting Risks into Perspective: Comparison of risks from the worked examples in terms of the Calman scale and comparison to annual background radiation. However, this is not always a meaningful method for explanation of risk.
Conclusions:
- There is an increasing requirement for medical professionals to provide risk information from procedures involving ionising radiation.
- Quantitative risk assessment based on estimated dose is possible for a number of types of radiolgical procedure using readily available techniques from literature and software tools.
- There are uncertainties associated with the risk assessments.
- They do identify which deterministic effects are likely and give ranges for stochastic effects.
- These are useful for follow up and communication with patients especially for high dose procedures in radiosensitive groups.