A fully validated Monte Carlo framework,
previously developed in our group,
was used to model a Planmeca Promax 3D Max CBCT (Planmeca,
Helsinki,
Finland) system1,2.
Simulations were performed with the Zubal and the ICRP reference female voxel phantoms3,4.
Three different protocols were simulated to calculate the dose delivered to the eye lenses under different exposure conditions.
A. Monte Carlo framework
Our EGSnrc based MC framework was initially customized towards dosimetric applications3,4.
It was adapted to enable the calculation of the energy imparted into predefined voxels and therefore to estimate the absorbed organ doses in pre-segmented voxel phantoms.
We further advanced the framework by skipping the x-ray tube simulation part of the framework chain and by applying a method,
already employed and tested for Multislice CT scanners,
for generating equivalent source models5.
The Half Value Layer (HVL) of the beam at the operating kilovoltage of the three protocols (96kV),
was measured with a farmer type ion chamber (IC) (IBA Dosimetry,
Schwarzenbruck,
Germany),
to specify an equivalent spectrum.
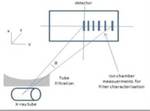
Air kerma measurements across the detector were also carried out,
in order to specify an equivalent filter which attenuates the equivalent spectrum in the same manner as the actual filter does to the actual spectrum (figure 1).The equivalent source model consists of a table with the relative number of photons per keV energy bin (equivalent spectrum) and a table with attenuation factors for each photon which crosses the tube filters from different angles.
Both of them are included into an input file which is used during the simulation.
B.
Voxel models
The manually segmented head CT dataset (from the neck to the crown of the head) of a human adult were used to generate the Zubal phantom (figure 2).The original in-plane and z-axis resolution were preserved (table 1).
The phantom was reorganized by merging certain organs together and it finally consists of sixteen major organs which are listed in table 2.
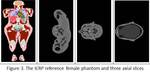
The ICRP reference female computational phantom (figure 3) was also employed in the study.
The initial phantom was modified such that only the last 59 images (head and neck region) were used.
The number of voxels along with the in-plane and z axis resolution are illustrated in table 1.
The customized phantom consists of twenty two major organs,
listed in table 3.
The elemental composition of each organ was obtained by ICRP 89 and Woodard et al 6,7.
To account for variations in bone density,
thickness or pathology across population,
a series of simulations were carried out by using different bone densities.
A method for defining the elemental composition of artificial bones of various densities was employed and six,
different in bone density,
versions of each phantom were created8 (bone density ranges from 1.2 to 2.2 g/cm2).
C.
Implementation
Organ doses were calculated for three,
different in FOV size and in referral criteria,
protocols; A jaw protocol 130x90 mm2 (diameter x height); A face protocol 130 x160 mm2 (diameter x height) and a skull protocol 230 x 260 (diameter x height).
We simulated the normal reconstruction scanning mode which corresponds to a reconstruction voxel size of 400 µm.
The tube current is 7.1 mA and the rotation time is 18 seconds.
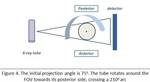
The first two protocols implement an in-plane symmetrical scanning mode; the detector is positioned symmetrically with respect to the FOV and the rotation angle is equal to 210 degrees (figure 4).
Thereby,
the eyes and hence the lenses are not included in the scanning volume.
The skull protocol is carried out in two consecutive 360 degree-rotations such that the whole head is imaged.
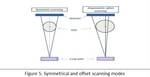
This protocol implements an offset axial scanning geometry; the axial beam coverage at each projection is slightly larger than the radius of the FOV (figure 5).
Due to its offset geometry a whole scanning rotation is required such that the full FOV is imaged; therefore the eyes and hence the lenses are exposed to primary photons as the x-ray tube crosses the anterior side of the patient during the rotation.
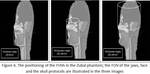
Figure 6 illustrates the positioning of the three FOVs in the head.
It is only scattered photons which reach the eye lenses in the case of the jaw protocol,
whereas in the case of the face protocol,
the lenses may also be exposed to primary x-rays as the tube crosses the posterior side of the head.
Though,
the beam is heavily attenuated by the organs in the head which are found behind the eye bulb.
In the case of the 360-degree rotation employed by the skull protocol,
the lenses are also exposed to primary radiation as the tube crosses the anterior side of the head.