EXAMINATION TECHNIQUE
Through a revision of the literature we present our case series of examinations acquired with a second generation dual-source 128 slice CT scan (Somatom Definition Flash,
Siemens,
Erlangen,
Germania).
The protocol includes several acquisitions.
After the patient has been elucidated regarding the procedure and an informed consent has been obtained,
a pre-contrast scan is acquired (Calcium Score) in order to plan the exact range for the following steps.
A vasodilator drug able to induce myocardial ischemia is administered for the successive acquisition in stress condition.
The most frequently used vasodilator agents,
both in our case series and in the literature,
are Adenosine and Dipyridamole.
The first one acts directly on adenosine receptors,
while the second elevates Adenosine endogenous levels by reducing its uptake from endothelial cells.
These drugs,
dilating the resistance coronary arterioles,
tend to abolish the auto-regulation mechanism (“transmural coronary steal”) with a subsequent flow reduction downstream the stenosis that is more severe as the stenosis increases.
Both have demonstrated a good sensibility and specificity but Dipyridamole has the advantage to be a lot cheaper and has a more prolonged effect (some minutes respect to the few seconds of Adenosine).
Both can be safely administered with contraindications in patients with asthma,
COPD or high grade AV-block without an implanted pace-maker.
The drug is injected based on body weight with an infusion pump.
The dose is 0.14 mg/kg/min in 3-5 minutes for Adenosine or of 0.56 mg/kg (low dose) - 0.84 mg/Kg (high dose) in 4-6 minutes for Dipyridamole.
Before the examination the patient cannot ingest methylate xanthines such as coffee,
tea,
chocolate,
energetic beverages or to assume bronchodilator drugs that antagonize the effect of the medications used during the examinations.
To inject the drugs and the contrast agent two venous accesses are obtained.
During the examination a 12 derivations electrocardiogram,
blood pressure and oxygen saturation are constantly monitored.
The CT scan room setting is shown in Fig. 1.
The patient is questioned several times for the appearance of symptoms like nausea,
vertigo,
headache,
thoracic pain and general malaise.
These manifestations usually disappear when the drug is suspended or with the administration of an antagonist (Aminophylline 120-240 mg e.v.).
At the end of the infusion the first 30 seconds volumetric angio-dynamic acquisition in stress conditions is performed with prospective tele-systolic ECG-based triggering (about 30% of the R-R interval).
The table moves alternatively back and forth in a “shuttle mode” including a 7.3 cm scan range ( Fig. 2 ).
The administration of contrast agent (≈50 ml of contrast media followed by 50 ml of physiological solution at 5 mL/sec) is temporized based on bolus-triggering with a ROI in the ascending aorta.
When the stress scan is completed,
in case it’s needed,
the antidote is administered (Aminophylline).
After a 15-20 minutes break,
necessary for a complete myocardial wash-out of the contrast agent previously injected and for a complete resolution of the pharmacological effect,
an acquisition in rest conditions can be added for a complete myocardial perfusion evaluation.
During the same examination it’s possible to perform a coronary-CT to match the anatomical data with the perfusion maps.
This is generally done with a prospectively ECG-triggered scan or with a prospective high pitch spiral can (which is possible in second generation dual-source CT scan) to reduce the radiation dose.
If the heart rate is deemed too high,
some beta-blockers can be administered.
An optional sequence can be added at 8-10 minutes,
without further contrast agent injection,
to reveal the presence of late-enhancement areas indicative for necrotic-fibrotic zones; it’s preferable to use low kV protocols (80-100 kV) that enable not only to reduce the radiation dose but also to highlight the difference between vital and ischemic myocardium [2].
EXAMINATION PROTOCOLS
The examination protocols can be the following:
1. An acquisition after pharmacological stress with a subsequent acquisition in rest condition ( Fig. 3 )
2. An acquisition at rest followed by an acquisition under pharmacological stress ( Fig. 4 )
Both protocols present some advantages and some disadvantages ( Fig. 3 ; Fig. 4 ),
[2,3]:
1. With the first protocol there is a higher sensibility in the scan in stress conditions and therefore in the identification of inducible ischemia.
However the contrast agent remained after the first scan could partially contaminate the second scan in rest conditions reducing its sensibility in infarct areas identification.
2. In the second case there is a higher sensibility and therefore a higher accuracy in the identification of infarct areas.
However,
the contrast agent remained after the first scan could partially contaminate the second scan in stress condition reducing its sensibility in the identification of inducible ischemia.
These can be underestimated also because of beta-blockers employment for the first scan.
The advantage of this approach is that if no significant stenosis are identified the examination can be interrupted with radiation dose and contrast media reduction for the patient.
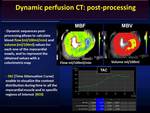
DATA POST-PROCESSING
Acquired images are elaborated with a dedicated software that allows a quantitative assessment of perfusion parameters (contrast agent flow and volume).
Volumetric angio-dynamic images dataset (≈10-15 datasets) are elaborated with two compartments (intra and extra vascular spaces) parametric deconvolution algorithms [4].
The software for the analysis requires a complex multi-step intervention from the radiologist (movement correction,
noise reduction,
volumetric manual and based on density values myocardial segmentation) ( Fig. 5 ; Fig. 6 ; Fig. 7 ).
Having obtained time attenuation curves (TAC),
for each voxel constituting the myocardium blood flow (expressed as ml/100 ml/min) and volume (expressed as ml/100 ml) values are extracted.
These values are then represented with a 3D color map ( Fig. 8 ).
For each voxel it's also possible to visualize how the attenuation values (density) change during time (TAC) with a graph ( Fig. 8 ).
The software automatically calculates the values and draws the TAC of the whole cardiac muscle; the radiologist can assess the regional perfusion by manually defining a region of interest (ROI) ( Fig. 8 ).
A semiautomatic software with a “bull's eye” representation that would allow a quicker,
easier and more accurate and standardized analysis is being tested.
IMAGES INTERPRETATION
Quantitative data regarding the myocardial enhancement can be obtained through images post-elaboration.
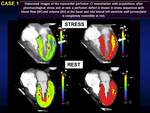
During early arterial phase myocardial segments with reduced vascularization show a reduced contrast agent content and therefore reduced blood flow and volume values.
These areas are defined as perfusion defects and represent inducible ischemia or infarct zones ( Fig. 9 ).
If the blood flow and volume reduction can be identified only in acquisitions performed during stress,
it's indicative of inducible ischemia; whereas if the defect persists also at rest it's due to infarct [3] ( Fig. 9 ).
TAC analysis enables to distinguish ischemic and necrotic areas.
It has been demonstrated that ischemic areas have reduced wash-in with delayed peak enhancement and preserved wash-out compared with remote myocardium ( Fig. 10 ; Fig. 11 ; Fig. 12 ; Fig. 13 ) [5].
On the other side infarct areas have reduced wash-out with delayed peak enhancement and reduced wash-out [6] ( Fig. 12 ).
Iodine based contrast agents,
as those based on Gd,
accumulate in the irreversibly damaged myocadiocytes intracellular space in case of acute myocardial infarction or in the expanded extracellular space in the event of fibrous scar: these necrotic/fibrotic areas can therefore be revealed as hyperdense zones (iper-enhancement) at a late scan 10-15 minutes after contrast agent injection,
Fig. 9 [8].
DOSE
Dynamic scans are performed with a kilo-voltage as low as possible based on patients BMI (80 kV for patients with BMI < 25 and 100 kV for patients with BMI > 25) [2].
By reducing the kilo-voltage not only the dispensed dose is lower,
but also the maximum attenuation difference between areas with different contrast agent concentration is obtained [2].
The reported administered dose for the dynamic perfusion CT varies greatly in the literature (from 3.8 mSv to 10 mSv) and is strictly dependent on the protocol optimization [2,
5,
8-16].
According to these data the method is very promising compared with myocardial perfusion assessment with SPECT (dose= about 10 mSv for 99mTc and 20-40 mSv for 201TI) [17,
18],
adding the advantage to combine in the same examination anatomical and functional informations.
ACCURACY
Data obtained with animal models regarding diagnostic accuracy in comparison with fluorescent microsphere and invasive measurement of the fractional flow reserve (FFR),
have been very encouraging ( Fig. 14 ) [5,
19-22].
Also preliminary data derived from last years human studies are very favorable (Fig.
15) [2,
8-16].
The technique presents good sensibility,
specificity,
PPV,
NPV respect to SPECT [11,
13,
16],
perfusion MRI [12] and invasive measurement of the FFR during coronarography [9-10,
14-15] ( Fig. 15 ).
Bamberg et al [9] and Rossi et al [10] have demonstrated the added value of dynamic perfusion CT in stress conditions to interpret intermediate stenosis (30-70% of lumen restriction).
The perfusion study has increased the specificity and overall accuracy of the coronary CT if compared with FFR.