The radiological appearances of mucosal o submucosal ureter wall thickening are different,
correlated by pathology.
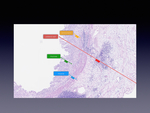
The ureteral wall is thin and is composed by mucosa,
lamina propria,
muscle.
(Fig. 1)
Usually,
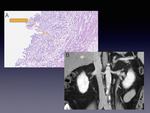
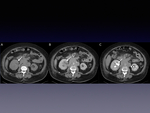
mucosal thickening is caused by cell proliferation at the mucosal and lamina propria (Fig. 2).
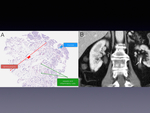
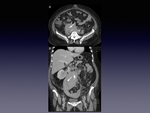
Submucosal thickening is caused by cell proliferation at the lamina propria (Fig. 3).
We studied different patients with radiological alterations of the ureter wall on CT and correlated them with the clinical evolution and/or pathology.
There are some diseases that causes usually mucosal thickening like urothelial neoplasm or pyeloureteritis and other that causes usually submucosal thickening like chronic inflamatory catheter-related or lymphoma (Table 1).
INFLAMATORY AETIOLOGY
- Chronic inflamatory catheter-related
Some patients with chronic double J catheter in the urinary system,
developes ureteral thickness because of chronic inflamatory (Fig. 3).
INFECTIOUS AETIOLOGY
- Pyeloureteritis
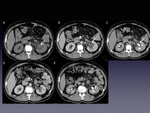
Alone pyeloureteritis is a rare entity that can simulates an urothelial neoplasm.
The clinical symptoms and laboratory items as the clinical evolution can help to make the correct diagnosis.
After medical treatment of antibiotics,
the symptoms disappeared.
(Fig. 4 and Fig. 5).
NEOPLASM AETIOLOGY
- urothelial neoplasms
Transitional cell carcinoma (TCC) is commonly encountered in the urinary bladder and is usually diagnosed at cystoscopy.
Five percent of urothelial tumors arise from the ureter or the renal pelvis or calices,
accounting for approximately 10% of upper tract neoplasms.
Patients with TCC typically present with hematuria,
which may be frank or microscopic.
Up to one-third of patients present with flank pain or acute renal colic,
symptoms more typically associated with calculi.
Occasionally,
tumors may manifest with distant metastases or be discovered incidentally at radiologic examination.
Renal TCC most frequently arises in the extrarenal part of the pelvis,
followed by the infundibulocaliceal region.
The distribution is equal between the left and right kidneys,
with 2%–4% of cases occurring bilaterally.
Twenty-five percent of upper tract tumors occur in the ureter (Fig. 2),
where 60%–75% of cases are found in the lower third,
with no side predominance.
Tumor spread occurs by mucosal extension or local,
hematogenous,
or lymphatic invasion.
The most common sites for metastases are the liver,
bone,
and lungs.
The tumor stage at diagnosis influences the development of local recurrence and metastases and hence overall survival.
Multicentric TCC is common and is associated with poor survival.
Synchronous or metachronous tumor of the ipsilateral or contralateral collecting system is also common,
necessitating vigilant urologic and radiologic follow-up.
Upper tract TCC typically occurs in the 6th and 7th decades of life,
with males affected three times more often than females.
Besides increasing age and male gender,
the most important risk factor is smoking,
with smokers being two to three times more likely to develop TCC than nonsmokers.
Chemical carcinogens (aniline,
benzidine,
aromatic amine,
azo dyes),
cyclo-phosphamide therapy,
and heavy caffeine consumption are also associated with TCC,
and all predispose to synchronous and metachronous tumor development.
These substances are metabolized and excreted in the urine as carcinogenic substances that act locally on the urothelium.
Stasis of urine and structural abnormalities such as horseshoe kidney are also associated with increased prevalence.
These tumors are usually small at diagnosis,
grow slowly,
and follow a relatively benign course.
Pedunculated or diffusely infiltrating tumor is less common,
accounting for approximately 15% of upper tract TCCs,
but tends to behave more aggressively and be more advanced at diagnosis.
Infiltrating tumors are characterized by thickening and induration of the ureteric or renal pelvic wall.
If the renal pelvis is involved,
there is often invasion into the renal parenchyma.
However,
this infiltrative growth pattern preserves renal contour and differs from renal cell carcinoma,
which is typically expansile.
- Lymphoma
Renal lymphoma has a wide variety of CT appearances.
In many cases,
diagnosis is not difficult because patients present with a known lymphoma at the time of imaging.
Lymphoma typically involves the kidney in one of several recognizable patterns.
These patterns include multiple renal masses,
solitary masses,
renal invasion from contiguous retroperitoneal disease,
perirenal disease,
renal sinus involvement and diffuse renal infiltration.
No specific correlation has been found between the exact type of lymphomatous involvement and the pattern or prevalence of renal involvement.Direct renal invasion from contiguous retroperitoneal disease is another common pattern of involvement in renal lymphoma and is seen in approximately 25%–30% of patients with documented disease.
These patients typically present with a large,
bulky retroperitoneal mass that envelops the renal vasculature and invades the renal hilum.
Lymphoma can preferentially affect the renal sinus,
although this is an uncommon occurrence.
In most patients,
the renal arteries and veins remain patent despite tumor encasement,
a finding that is characteristic for lymphoma.
However,
contiguous extension of retroperitoneal involvement in the renal collecting system can often cause obstruction,
and affected patients will commonly present with hydronephrosis (Fig. 6).
Transitional cell carcinoma is usually associated with a greater degree of obstruction of the collecting system (Fig. 7).
Displacement of the kidney can also be seen.
Following treatment of larger masses,
residual fibrosis is often seen and can be mistaken for recurrent or residual disease.