A pictorial review aiming to address theoretical concepts and illustrate some of the imaging findings and differential diagnosis of the most common types of benign adnexal masses based on ultrasound,
CT and MRI scans performed at our institution,
and discussing features pointing towards benign or malignant etiology such as solid components,
vascularity,
accompanying ascites and others.
General approach principles
The first line,
go-to examination for pelvic mass diagnosis and characterization is transvaginal ultrasound.
Advancements in the field and hardware have allowed this radiation free,
low cost exam to evaluate adnexal masses with high precision,
detail and resolution through a pattern recognition approach.
Transabdominal ultrasound can also be useful,
especially with sizeable lesions although lower probe frequencies and higher distance from the mass will limit its resolution and detail compared with TVUS.
It is also easily repeatable allowing for conscientious follow-up,
and allows study of tumor vascularization by use of color,
power and spectral Doppler.
Its main limitation,
besides air and fat interposition,
is known to be its operator dependency,
so proper training and experience are keys to success and will often preclude the need for further imaging.
MRI is traditionally held as a “problem solving” modality,
with its superior soft tissue differentiation and multiple sequences and techniques allowing characterization of many US-indeterminate lesions.
It also allows easier evaluation of lymph nodes and regional pelvic anatomy.
CT has traditionally been reserved for staging of malignant lesions,
having been largely supplanted by MRI for adnexal mass characterization given its inferior soft tissue differentiation,
the use of radiation and literature derived from old single-detector scanners.
However,
"recent" advances in MCDT scanners and the ability for fast,
lower radiation dose imaging,
use of IV contrast and excellent multiplane reconstruction ability have resulted in studies demonstrating similar (though slightly inferior) performance to MRI for the management of adnexal lesions and will therefore be discussed in this work.
Suspicious findings
Though the focus of this work is mostly to discuss benign adnexal lesions and their imaging findings,
one must first acknowledge them as likely benign before attempting to further their characterization.
This section lists some helpful features that raise suspicion of malignancy (Fig.
5) and that should therefore bring about proper consideration for subsequent patient management.
- Mixed solid and cystic lesions are,
generally speaking,
suspicious for malignancy,
especially when intratumoral flow can be demonstrated on Doppler imaging or contrast enhancement on CT/MRI on the solid component; Benign pseudo-nodular irregularities such as adherent clots should be strictly avascular;
- Thickened (>3mm) or irregular/nodular walls and septs, once again especially if Doppler positive or enhancing in CT/MRI; Ascites,
though present in some benign conditions,
should raise suspicion for malignancy and inspire extra care,
particularly in searching for signs of peritoneal carcinomatosis such as implants or omental cake;
- Lymphadenopathy or images suggesting secondary deposits;
- Though not strictly imaging related,
a detailed clinical history and lab work including elevated tumor markers may also contribute to increase our suspicion;
Cystic Masses
Unilocular
Functional (Follicular) Cysts
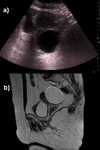
Physiologic or functional ovarian cysts are usually well defined,
smooth thin walled (<3mm) and fluid filled structures located on the ovarian parenchyma.
They should be uniloculated even though they are often multiple and bilateral owing to follicle maturation and development.
They can be large but usually range from a few millimeters to roughly 3cm,
and usually cause no symptoms.When these features are demonstrable,
the main differential diagnosis to be considered is a (serous) cystadenoma.
Follicular cysts’ functional and cyclic nature makes them appear mostly in reproductive age to peri-menopause,
and they will tipically resolve in a few months,
so follow-up imaging is advised,
usually by ultrasound.
Imaging findings correlate well between different modalities,
ultrasound revealing anechoic content,
no septations,
focal thickening or solid components (Fig.
1) ,
CT much of the same with homogeneous low density content and MRI with hyperintense T2w and low intensity in T1w,
no significant contrast enhancement on any modality.
,
CT much of the same with homogeneous low density content and MRI with hyperintense T2w and low intensity in T1w,
no significant contrast enhancement on any modality.
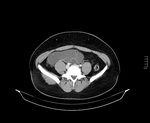
Serous Cystadenoma
Though Serous Cystadenomas can be multilocular,
the majority are simple cystic formations (Fig.
7) sharing much in common with the functional cysts described above in terms of imaging findings.
No reliable features exist to clearly set them apart,
and the main differences have to do,
by definition,
with size and lack of resolution,
as serous cystadenomas are typically larger (>3cm) and can grow to large volumes before being detected since they also tend to be asymptomatic.
Imaging differentiation from serous cystadenocarcinoma relies mostly on the general malignancy criteria previously discussed.
Hydrosalpinx
Hydrosalpinx refers to distension of the fallopian tubes due to build-up of fluid,
usually resulting from obstruction of its lateral extremity (ie: adhesions,
scarring,
sequelae from PID,
among others).
It is an important differential diagnosis to consider among other cystic adnexal masses and one of the easiest ways to point towards it is by demonstrating an ipsilateral normal or at least clearly distinct ovary.
It may abut the ovarian border,
complicating the differential diagnosis vs an exophytic cystic tumour but should not be seen producing indentations or the “beak sign” on closer inspection.
Other characteristic findings include tubular,
“S” or “C” shape instead of round or oval as seen with simple cysts or cystadenoma.
Also incomplete “septa” can be seen owing to folding of the tube’s wall.
Cross sectional imaging on multiple planes should prove useful in assessing these findings.
Uncomplicated hydrosalpinx translates into clear,
anechoic content on ultrasound,
low density on CT and hyperintense T2w and hypointense T1w on MRI,
with thin and unremarkably enhancing walls.
Hemorrhagic Cyst
Hemorragic cysts are also usually functional in nature and often result from follicle/corpus luteal bleeding.
Their imaging findings are variable according to timing of the exam,
and so follow up is key to determine their resolution,
usually complete after about 8 weeks.
On ultrasound acute hemorrhagic cysts can display large clots to the point of resembling a solid,
slightly heterogeneous but non shadowing lesion with good transmission,
however will elicit no internal flow when challenged with Doppler,
whereas peripheral flow is often found or even increased.
CT will sometimes depict these cysts with high (>50HU) spontaneous density and will show no real enhancement of the lesion.
The most commonly found stage is the subacute one,
when the clot begins to shrink and/or resolve eliciting characteristic images described as “retracting clot” – solid,
avascular component with convex borders in a cystic mass (Fig.
2)– and lace-like,
multiple fibrin strands – avascular,
very thin echogenic strands,
often incompletely seen that differ from true septs.
MRI findings are very similar to the ones found in endometriosis,
described below,
and differentiating these lesions based exclusively on this modality is difficult.
Clinical history and follow up help guide suspicion,
however,
it’s worth mentioning that hemorrhagic functional cysts tend to be solitary whereas endometriotic cysts can more often be multiple and even bilateral.
Also,
acute hemorrhage results in early peripheral deoxyhemoglobin to methemoglobin conversion as it transitions to its subacute phase causing a peripheral hyperintense rim in T1w images.
Multilocular
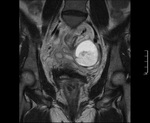
Endometriosis
Endometriosis comes as a consequence of ectopic implant of endometrial tissue,
and while there are multiple theories attempting to explain this phenomenon,
they are beyond the scope of this work.
It is an important cause of recurrent,
chronic pelvic pain and imaging methods play an increasingly important role in its diagnosis and management.
The most common sites of implant are the ovaries,
first and foremost,
and the peritoneum,
however other locations are possible.
On ultrasound endometriomas typically present as a homogeneous nodules,
with low level echoes resembling “ground glass”-like texture,
even though a smaller percentage will have findings roughly overlapping with hemorrhagic cysts,
displaying cystic heterogeneous structure with “lacey” fibrin strands and sometimes associated adherent mural clots.
They can appear isolated but are much more likely than hemorrhagic cysts when multiple and/or bilateral.
Concurrently,
chronic inflammation may lead to scarring and adhesions to surrounding structures such as the fallopian tubes,
small bowel or even contralateral ovary producing a typical sign termed “kissing ovaries”.
(Fig.
3)
CT findings correlate well with ultrasound,
demonstrating (multiple and/or bilateral) cysts with discrete variations in density,
usually fluid-like but in case of fresh hemorrhage can increase up to slightly over 50HU.
Hydrosalpinx,
adherences and adjacent fat stranding can also be shown,
though there should be no remarkable enhancement.
A slight “hematocrit effect” gradient can seldom be demonstrated.
Small amounts of pelvic free fluid can also be present.
MRI boasts the most specific findings and diagnostic criteria for endometrioma include either (multiple) hyperintense nodules in both T1w and T2w,
with no dropout after FS (differentiating from fat content in a dermoid,
for instance) or hyperintense T1w and “shading” in T2w owing to high viscosity brought on by chronic hemorrhage and blood degradation products.
This shading can also occur in hemorrhagic cysts although it is not as common or as pronounced due to a smaller concentration of the mentioned blood products.
Fibrotic adhesions and other criteria discussed above are also well evaluated and instill further confidence in the diagnosis.
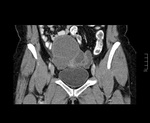
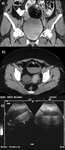
Mucinous Cystadenoma
Though Mucinous Cystadenomas can be uniloculated,
most of them exhibit multiple loci with thin septa as opposed to serous cystadenomas.
Also,
they tend to be smaller than their serous counterpart.
By definition,
there should be no solid component and care must be taken to differentiate between true septal/wall thickening and overlapping walls of a collapsed loca.
(Fig.
4)
On ultrasound they typically present with anechoic content,
though some heterogeneity may result from variable concentration of mucin bringing about homogeneous low level echoes on some or even all of the loci.
This correlates well with other cross-sectional imaging modalities as sometimes it is possible to establish small differences in density (CT) or intensity (MRI) from different loci.
Imaging differentiation from mucinous cystadenocarcinoma relies mostly on the general malignancy criteria previously discussed.
(Fig.5)
Solid Masses
Fibrothecoma
Fibromas and thecomas are some of the most common sex-cord stromal tumors of the ovary and are most often benign.
They are predominantly collagenous nodules formed by varying amounts of spindle,
round and oval cells and so have similar,
overlapping imaging findings and are commonly grouped together,
and will be discussed here as such.
Some of these are hormonally active (estrogenic) and so assotiated uterine enlargement may favor this diagnosis.
On ultrasound they tend to present as solid,
homogeneous hypoechoic nodules with posterior sound attenuation.
These findings,
however,
lack specificity and differentiating these from other entities such as pedunculated leiomyoma may prove difficult.
Rarely,
these may demonstrate internal cystic component and further complicate the differential.
CT shows these lesions as predominantly solid,
tissue density lesions with minimal or absent arterial enhancement,
and larger lesions may just as well demonstrate fluid density areas as observed on ultrasound.
MRI generally demonstrates intermediate/low signal intensity in T1w and low signal intensity in T2w sequences similar to that of muscle,
and likewise some heterogeneity can be accounted for just as well by areas of cystic degeneration paralleling CT and US findings.
Little to no enhancement should occur after gadolinium administration,
although contrast use isn’t generally warranted when faced with typical findings.
Brenner Tumor
Brenner tumors make up a smaller portion of ovarian tumors.
It is a predominantly solid surface-epithelial stromal tumor comprised of transitional epithelial cells,
not unlike those found in urothelial tumors.
They are mostly unilateral.
On ultrasound they present similarly to fibrothecomas,
as a solid hypoechoic and attenuating lesion.
Calcifications are also commonly seen (~50% of cases).
CT findings correlate well and also show frequent gross calcifications,
and may demonstrate mild to moderate enhancement post contrast.
MRI best differentiates these lesions owing to characteristic very low signal intensity in T2w sequences,
some studies reporting it to be the lowest of all solid tumors ovarian,
in fact.
It is also worth mentioning that Brenner tumors are often associated with ipsilateral mucinous cystic tumors,
in which case findings will reflect a mixed solid cystic multilocular lesion.
Mixed Solid and Cystic Masses
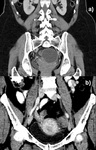
Dermoid
Dermoids,
of which mature cystic teratomas are the most common,
are tumors comprised of cells derived from all germ cell layers.
Hystologically they may show variable amounts of fat (sebum),
hair,
or even teeth and so have variable appearance on imaging that correlates well with its varying composition.
Ultrasound will reveal a heterogeneous cystic and solidmass with posterior echo attenuation relating to the fat content and to hair clumps or strands,
which both absorb and reflect echoes.
If dispersed,
hair follicles will sketch a reticular pattern often described as “dermoid mesh”,
roughly similar to fibrin strands observed in hemorrhagic cysts.
When clumped,
they will often form an echogenic nodule with posterior dirty shadowing usually described as “tip of the iceberg” sign.
It’s worth noting that sebum is often fluid at body temperature and so will commonly be seen as hypoechoic material with internal echoes.
Teeth will generally present themselves as stonglyhyperechoicstrutures with marked,
“clean” posterior shadowing.
A nodular mural protuberance is commonly identified and is termed “Rokitansky nodule” or “dermoid plug”.
CT also shows very characteristic findings and facilitates the diagnosis when multiple different densities can be demonstrated,
often showing material with fat density,
tecidular and even calcified density corresponding to fat,
hair and teeth,
respectively.
(Fig.
6)
MRI is usually unnecessary for the diagnosis,
but excels in demonstrating intra-lesional fat by exposing hyperintense material in T1w paralleling subcutaneous fatty tissue and subsequent comparison to T1w FS sequence.
Fat-fluid levels with floating debris,
and Rokitansky nodules are also characteristic and correlate well with other imaging modalities.
Other lesions worth discussing
Ovarian Torsion
A special mention to adnexal torsion is warranted here,
as even though it may not be an adnexal mass in itself,
it is an important differential diagnosis which often presents itself with,
among other findings,
an enlarged ovary owing to congestion and edema caused by initial compromise of its venous drainage.
It is a gynecological emergency requiring prompt surgical attention and if left unattended will likely progress to hemorrhagic necrosis.
It is also important to note that adnexal torsion,
while commonly related to hypermobility of the ovary,
can also often be associated with an ovarian mass or tumor,
most commonly a dermoid.
Doppler evaluation is,
as expected,
fundamental when faced with a possible adnexal torsion.
Typical findings include diffusely decreased vascularity on color and power Doppler when compared to the contralateral ovary,
and when possible,
spectral analysis commonly demonstrates lack of venous blood flow,
can often still detecthigh resistance arterial waveforms with absent or residual diastolic flow or,
less commonly,
total lack of flow.
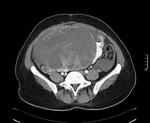
CT evaluation correlates well with ultrasound findings and can show a complex structure comprised of edematous enlarged ovary,
distended pedicle and eventually an underlying lesion.
It may also demonstrate fat stranding adjacent to the ovary and some amount of pelvic free fluid is usually also found.
When compared with contralateral ovary it will often enhance less or not at all after contrast administration.
If possible,
demonstrating a twisted or ”knotted” pedicle is highly specific,
so multiplanar reconstructions may prove useful.
(Fig.
7)
Owing to the emergent nature of this condition,
MRI is usually not indicated.
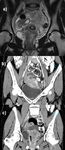
Tubo-ovarian abscess
Tubo-ovarian abscess,
and/or pyosalpinx usually occur following a bout of Pelvic Inflammatory Disease and can be either acute or chronic,
with subtle changes to the imaging findings.
There is also often a suspicious clinical context with fever and pelvic pain or discomfort,
dyspareunia,
and/or other symptoms.
It translates to an enlarged,
heterogeneous and often ill-defined adnexa with solid and multilocular cystic components.
The fallopian tube can also be distended with fluid,
hematic or purulent content and so will present itself with hypoechoic content often with internal echoes or debris,
slightly higher density on CT compared to uncomplicated hydrosalpinx and occasionally prominent,
thickened and strongly enhancing walls with some degree of adjacent fat stranding attributable to local inflammatory changes.
(Fig.
8)
Ultrasound and MRI,
in particular,
might also show pseudo-papillary projections on the mucosa resulting from edematous intrasalpyngeal folds,
thicker and more prominent in acute cases,
that earned this feature the term - “cogwheel sign”; Chronic cases may also show these folds,
though less prominent and thinner.
Small amounts of pelvic fluid can be accounted for but care and cross sectional imaging should be considered to rule out peritoneal implants and other findings that may point towards underlying malignancy.