HIP OSTEOARTHRITIS
Hip osteoarthritis affect 4.4% of adults over 55 years.
Primary hip OA is found as a degenerative joint disease frequently associated with increased loads on this weight-bearing joint,
such as in the case of various grades obesity.
Secondary hip OA can be found in patients with untreated FAI (femoracetabular impingement) or hip dysplasia,
in which the altered biomechanics usually leads to joint damage.
Clinical presentation
The main symptom is joint stiffness that occurs as they are getting out of bed or after sitting for a long time,
variably associated with pain,
swelling,
or tenderness in the hip joint and inability to move the hip to perform routine activities such as exiting from the car or putting on socks.
Sometimes,
a sound or feeling of bone rubbing against bone is reported.
Ultrasound diagnosis
The diagnosis of hip OA requires plain film examination.
However,
US can demonstrate the presence of joint effusion,
thickened and/or calcified capsule and calcifications of the acetabular labrum.
Treatment options
Conservative treatment includes weight loss,
physical therapy,
anti-inflammatory and analgesic medications and oral administration or intra-articular injections of steroids.
At later stage,
total hip replacement can be performed.
Interventional procedure
Indications are diagnostic or therapeutic pain relief,
intra-articular injection of steroids,
primary or secondary OA,
degenerative osteoarthritis associated with articular effusion,
conservative treatment in patients ineligible for total hip arthroplasty.
Contraindicated if suspected or known joint infection,
presence of joint fracture,
coagulopathy,
overlying cellulitis or infection,
hypersensitivity to corticosteroids.
Objective
To deliver anti-inflammatory,
analgesic or viscosupplementant drugs within the joint space.
Equipment
- 1 syringe (5 ml);
- 20G spinal needle;
- Local anesthetic (1-3 ml Lidocaine 2% without adrenaline);
- Long-acting steroid (1 ml,
40 mg/ml) or hyaluronic acid (2-4 ml)
- Plaster
How we do it
Intra-articular injection at the level of the hip is generally more complex for its deep location and the presence of “critical” anatomic structures (femoral neurovascular bundle).
Out-plane approach has its strength in a shorter procedure time,
but suffers from a more difficult visualization of the needle tip.
Lateral (in-plane) approach allows a direct and continuous visualization of the needle along the whole path in soft tissues.
The patient lies in supine position,
with the lower limb slightly intra-rotated (about 15-20°).
The neurovascular bundle can be visualized with an axial scan at the level of the groin in order to locate it,
detect eventual vascular or neural anatomic variations and avoid accidental punctures.
Then,
the probe is rotated 90 degrees and shifted laterally in order to reach an anterior para-sagittal scanning plane at the level of the coxo-femoral joint.
A correct scanning plane should visualize the femoral neck,
the femoral head covered by hyaline cartilage,
the acetabular labrum,
the osseous component of the acetabulum,
the joint capsule and,
superficially,
the ileopsoas muscle belly.
The articular cortex of the femoral head appears as a spherically curved echogenic line,
and the cortical surface of the anterior acetabular rim as a triangular echogenic structure just distal to this line.
The fibrocartilaginous anterior acetabular labrum is seen as a well-defined,
triangular,
and uniformlyechogenic structure.
Lateral approach
- ANAESTHESIA: We inject a small amount of local anesthetic in the intra-articular space (up to 3mL Lidocaine 2% without adrenaline,
20G needle) to confirm the correct intra-articular positioning of the needle tip. According to the patient’s habitus,
the depth of the joint can vary extremely,
and thus the angle of needle insertion has to be adjusted; generally,
the angle of needle insertion ranges from 30 to 60 degrees.
With this approach,
the whole path of the needle can be visualized in real-time and slight corrections of the direction can be made.
There should be no resistance to the injection; if this is not the case,
a short retraction (1–2 mm) of the needle should be considered because the needle tip could be pointed against the femoral cartilage or into the anterior acetabular labrum (Fig.3).
Fig. 3: Video showing the step No. 1 of the infiltrative procedure: anesthesia.
- STEROID OR HYALURONIC ACID INJECTION: We inject 1mL of steroid (1mL of triamcinolone acetonide 40mg/mL; steroid solution has a hypoechoic appearance with many hyperechoic dots inside) or 2-4mL of intermediate molecular weight hyaluronic acid (hyaluronic acid is highly echogenic). The intra-articular injection should determine a distension of the joint capsule according to the amount of fluid injected.
When injecting small amounts (2-4 ml),
the use of color-Doppler module can help in detecting the flow artifact under the hyperechoic line of joint capsule.
At the end of the injection,
the needle can be removed and a plaster is applied at the cutaneous site of approach (Fig.4).
Fig. 4: Video showing the step No. 2 of the infiltrative procedure: steroid or acid hyaluronic injection.
Coaxial approach
The articular joint space is centered on the screen and a 20G spinal needle is inserted laterally and at the level of the middle to the probe,
with a very slight lateral-to-medial angulation (about 5°) to reach the joint space visualized in the scanning plane.
Along its path,
the needle tip is visualized indirectly,
by means of slight movements of superficial soft tissues; when the intra-articular position is reached,
it is visualized as a hyperechoic dot under the articular capsule.
An intra-articular injection of a small amount (1-3ml) of local anesthetic and the use of color-Doppler can help in confirming the correct position of the needle tip.
We inject 1ml of steroid (1mL of triamcinolone acetonide 40mg/mL) or 2-4 ml of intermediate molecular weight hyaluronic acid.
At the end of the injection,
the needle can be removed and a plaster is applied at the cutaneous site of approach.
Post-procedural Care
The patient should be kept under observation for at least 15 minutes after the procedure.
Pain may occur after treatment and is managed with oral NSAIDs.
HIP BURSITIS
Synovial bursae are anatomical structures that are located in critical regions where tenomuscular structures run in close relationship with other musculotendinous or osseous components and act as friction attenuators between such structures during movement.
Several bursal structures are located around the hip: in the anterior compartment lies the iliopsoas bursa,
between iliopsoas distal myotendinous junction and the anterior aspect of the hip joint; in the lateral compartment several bursae are located between the glutei and tensor fasciae latae muscles and tendons and trochanteric cortical bone (peritrochanteric bursa); in the posterior compartment the ischiogluteal bursa lies between the ischial tuberosity and the deep surface of the gluteus maximus.
Iliopsoas bursa
The iliopsoas bursa is a large synovial bursa lying between the posterior aspect of the iliopsoas muscle and tendon and the anterior capsule of the hip joint,
between the medial femoral vessels and the lateral iliopsoas muscle,
and allows a smooth gliding between these structures during hip movements.
It presents a direct communication with the articular space in up to 15% of cases and in normal conditions it can’t be demonstrated because it contains only a small amount of fluid.
Inflammatory or degenerative hip diseases may be complicated by the development of iliopsoas bursitis although it may also represent an isolated primary pathology or a sport-related overuse syndrome.
Also,
idiopathic bursitis is occasionally described.
Clinical presentation
A large distention of the iliopsoas bursitis can lead to symptoms referred in the lower right quadrant of the abdomen,
mimicking appendiculopaty or pelvic disorders.
When giant,
it can develop inside the pelvis and produce mass effect on the pevic structures.
If fever is present,
the diagnosis of septic bursitis must be considered.
Ultrasound diagnosis
Ultrasound can easily detect distention of the bursae around the hip,
demonstrating a well-defined anechoic fluid collection between gliding anatomical structures.
The pressure applied on the probe determines the squeezing of fluid bursal distentions,
helping to differentiate bursitis from other pathological conditions (e.g.
paralabral ganglion cysts,
whose mucinous content is not compressible).
Bursal walls can thicken in chronic cases,
or internal echogenicity can be present in rheumatic patients due to the presence of a synovial pannus.
Iliopsoas bursitis is seen as an ovoidal,
well-defined hypoechoic collection between the medial femoral vessels and the lateral iliopsoas muscle (Fig.5).
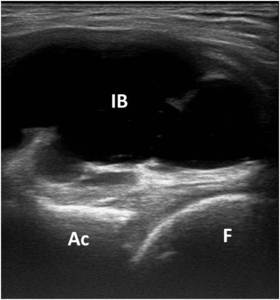
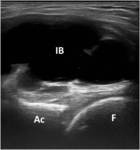
Fig. 5: Short axis US scan of ileopsoas bursa (IB) dimonstrating the fluid bursal distentions. F, femur; Ac, acetabulum.
Treatment options
Conservative treatment includes rest,
weight loss,
physical therapy,
ice,
anti-inflammatory and analgesic medications.
US-guided aspiration and subsequent steroid injection is necessary in the case of large bursal distentions if infection is excluded.
When infected,
US-guided aspiration and microbiological analysis of the bursal fluid is required before starting the antibiotical therapy.
Surgical drainage and removal is usually reserved for unresponsive septic bursitis.
Interventional procedure
Indications are diagnostic aspiration or therapeutic pain relief,
intra-bursal injection of steroids and/or viscosupplementant drugs.
Contraindicated if suspected or known bursal infection or in the presence of overlying cellulitis or infection,
hypersensitivity to corticosteroids.
Objective
To deliver anti-inflammatory,
analgesic or viscosupplementant drugs within the affected bursal space.
Equipment
- 1 syringe (20 ml);
- 16-18G needle or a larger shielded cannula;
- Local anesthetic (1 mL Lidocaine 2% without adrenaline);
- Long-acting steroid (1 ml,
40 mg/ml) and/or hyaluronic acid (2-4 ml);
- Plaster.
How we do it
- Patient lies in supine position with the lower limb slightly extra-rotated and a parasaggital US scan is made to identify the enlarged bursa and to assess its anatomical extension. The needle is inserted with an in-plane caudo-cranial approach to reach the bursal space.
- A needle connected to a syringe is inserted with an in-plane approach until the needle tip enters the bursa.
Sometimes the bursal content is very dense and drainage could be extremely challenging.
In these cases,
a larger shielded cannula and application of a manual compression over the bursa may be helpful.
A biopsy handle may also be used to obtain a more effective vacuum effect.
- When the bursa is completely drained,
a small amount of steroid (1 ml) and local anesthetic (1ml) is then injected inside the bursa.
A viscosupplementant drug can also be used together with the steroid to reduce friction between bursal walls.
The needle is then removed and a plaster applied at the cutaneous puncture site (Fig.6) .
Fig. 6: Video showing the drenaige of ileopsoas bursa and the steroid injection with a parasaggital US scan.
Post procedure care
The patient is kept in observation for at least 10 minutes.
Pain occurrence after treatment may occasionally occur and it could be managed using oral NSAIDs.
HIP INSERTIONAL TENDINOPATHY
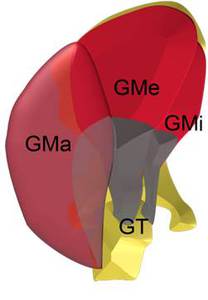
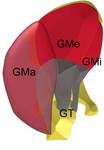
Fig. 7: Anatomical scheme of gluteal muscles and tendons. GT, greater trochanter; GMi,gluteus minimus; GMa, gluteus maximus; GMe, gluteus medius.
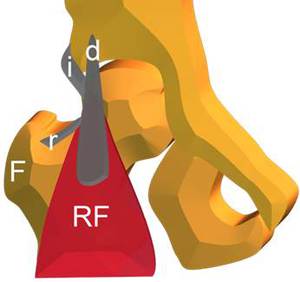
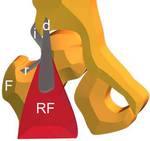
Fig. 8: Anatomical scheme of Rectus Femoris (RF) tendons. F, femur; i, indirect tendon; d, direct tendon; r, reflected tendon.
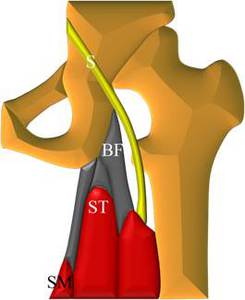
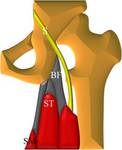
Fig. 9: Anatomical scheme of proximal insertion of Hamstrings muscles on ischial tuberosity. BF, biceps femoris; SM, semimembranosus; ST, semitendinosus.
Tendinous structures around the hip can develop degenerative changes at the site of insertion as a result of chronic microtraumatism due to overload or previous poorly-healed acute injuries.
Accurate anamnestic data collection and clinical examination are essential to differentiate this condition from acute or subacute traumatic injuries and other periarticular or intra-articular pathologies.
Tendinous insertions in the antero-medial and posterior compartments such as adductors,
rectus femoris and hamstrings are more frequently affected in active individuals doing sports such as running,
cycling or football.
On the other hand,
chronic tendinopathy of the lateral compartment is most commonly found in middle-aged to elderly women and is generally referred as to “greater trochanteric pain syndrome” since cannot be distinguished clinically from trochanteric bursitis,
which condition is often associated (Figg.7-8-9).
Clinical presentation
The most common symptom is pain at the corresponding site of affected tendinous insertion,
worsened by and the execution of active movements against resistance,
digital compression or when lying on the affected side,
without limitations in the hip range of motion.
Attention must be paid to exclude abdominal causes of pain that can mimick adductor or rectus femoris tendinopathy.
Hamstrings enthesopathy can also mimick radiculopathy or be associated with sciatic nerve irritation.
Ultrasound diagnosis
Ultrasound can assess with high accuracy the several tendinous structures that take insertion on different bony attachment sites around the hip.
In the presence of tendinopathy,
a variable grade of swelling and loss of fibrillar tendinous echotexture can be demonstrated (Fig.10),
as well as the presence of tiny hyperechoic calcifications at the site of insertion.
In severe cases,
partial or full thickness tendon tears can be seen as focal anechogenic areas with loss of the normal fibrillar pattern.
In patients with trochanteric enthesopathy the presence of a fluid-distended peritrochanteric bursa can be an associate finding.
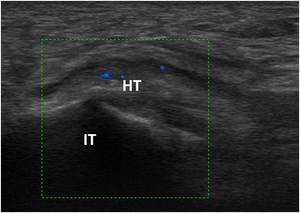
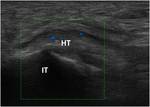
Fig. 10: US longitudinal scan to evaluate the hamstrings’ insertion (HT) into the ischiatic tuberosity (IT) that shows swelling and hypoechogenity of the tendon and increased vascularity on color Doppler.
Treatment options
Conservative treatment includes rest,
weight loss,
physical therapy,
ice,
anti-inflammatory and analgesic medications.
Pelvic imbalance (e.g.
leg-length discrepancy) should always be excluded or corrected.
In unresponsive cases,
percutaneous ultrasound-guided procedures or surgical tendinous debridement can be considered.
Interventional procedure
Indication is symptomatic insertional enthesopathy in one or more tendons around the hip.
Objective
To cause local hyperemia and bleeding into the tendon,
thus promoting post-procedural platelet-induced recovery phenomena.
Equipment
- 1 syringe (5-10 ml);
- 1 syringe (1-2 ml);
- 20G spinal needle;
- Local anesthetic (5-10 mL Lidocaine 2% without adrenaline);
- Long-acting steroid (1 ml,
40 mg/ml) and/or hyaluronic acid (2-4 ml);
- Plaster.
How we do it
1- The patient is positioned in a different way according to the tendon to treat (see below).
A US scan is made to identify the insertional portion and to assess the most confortable percutaneous approach.
- Adductors enthesopathy: the patient lies in supine position with the lower limb in slight extrarotation.
The proximal tendinous insertion is assessed with a longitudinal scan and the needle is inserted with an in-plane cranial-caudal approach.
- Rectus femoris enthesopathy: the patient lies in supine position with the lower lib in neutral position.
The proximal tendinous insertion is assessed by means of both transverse and longitudinal scans and the needle is inserted with an in-plane medial-to-lateral or caudo-cranial approach.
- Trochanteric enthesopathy: the patient lies on the controlateral side.
The affected tendon is demonstrated with an axial scan over the trochanter avoiding to apply excessive pressure to detect if trochanteric bursitis is associated.
The needle is then inserted with a posterior-to-anterior,
medial-to-lateral,
in-plane approach.
- Hamstrings enthesopathy: the patient is positioned prone and tendinous insertions are demonstrated by means of a parasagittal US scan.
Discrete pressure on the probe could be necessary according to patient’s habitus.
The needle is then inserted with an in-plane,
caudal-to-cranial approach.
2- Anesthetic is injected along the path of the needle,
in the peritendinous soft tissues and in the degenerated portions of the affected tendon.
A series of 15-20 repeated punctures (dry-needling) on the insertional portion of the tendon is then performed,
hitting also the periostum (Fig.11).
3- 1ml of steroid is injected in the peritendinous soft tissues,
superficially to tendon enthesis.
When treating trochanteric enthesopathy,
a small amount of steroid and/or viscosupplementant drug can be injected in the peritrochanteric bursae if effusion present.
The needle is then removed and a plaster applied at the cutaneous puncture site.
Fig. 11: Video showing the dry needling procedure on the hamstrings' insertion on the ischial tuberosity.
Post procedure care
The patient is kept in observation for at least 10 minutes.
A rest period of the affected structure and associated physical therapy are suggested.
Pain occurrence after treatment may occur and it could be managed using ice and oral NSAIDs.
PATELLAR TENDINOPATHY
Patellar tendinopathy,
also called “jumper’s knee” is considered the result of multiple micro-tears that do not heal because of poor tendon vascularity.
It is a degeneration of the tendon enthesis caused by a repetitive and/or chronic overloading that exceeds the adaptive capacity of the tendon.
This disease mostly affects young athletes and is associated with running,
jumping,
soccer,
and volleyball.
Some intrinsic factors (e.g.
gender,
age and overweight) and extrinsic factors (e.g.
functional overload and training errors) play an important role in the development of chronic tendinopathy.
Clinical presentation
Patellar tendinopathy prsents a progressive development over a period of weeks or months.
The most common symptom is stabbing or burning pain in the patellar tendon,
pain and stiffness in the patellar region in the morning or after rest,
tenderness or lumps on palpation. May be acute if partial tear.
Ultrasound diagnosis
Ultrasound can assess with high accuracy the patellar tendon.
In the presence of tendinopathy we can see swelling and loss of fibrillar tendinous echotexture,
as well as the presence of tiny hyperechoic calcifications at the site of insertion (Fig.12).
In severe cases,
partial or full thickness tendon tears can be seen as focal anechogenic areas with loss of the normal fibrillar pattern.
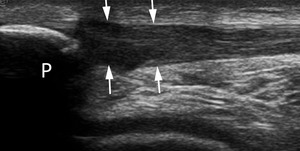
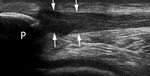
Fig. 12: US longitudinal scan of the patellar tendon (arrow).Note the swelling and loss of fibrillar tendinous echotexture at the tendinous insertion on the lower patellar pole (P).
Treatment options
Conservative treatment includes therapeutic exercises (eccentric training),
extracorporeal shock wave therapy,
and different steroid and platelet-rich plasma injection treatments.
Surgical treatment may be indicated in motivated patients if carefully followed conservative treatment is unsuccessful after more than 3-6 months.
Open surgical treatment includes longitudinal splitting of the tendon,
excision of abnormal tissue (tendonectomy),
resection and drilling of the inferior pole of the patella.
Interventional procedure
Indication is symptomatic insertional enthesopathy of patellar tendon.
Objective
To cause local hyperemia and bleeding into the tendon,
thus promoting post-procedural platelet-induced recovery phenomena and introduce PRP (Platelet rich plasma) that is a concentrate of platelets,obtained centrifugating a sample of fresh blood.
Equipment
- 1 syringe (5-10 ml);
- 1 syringe (1-2 ml);
- 21G needle;
- Local anesthetic (5-10 mL Lidocaine 2% without adrenaline);
- PRP;
- Plaster.
How we do it
1- With the patient in supine position,
perform an axial US scan of the patellar tendon. After thorough disinfection of the skin and subcutaneous administration of local anesthesia (4 ml of 2% Lidocaine),
US-guided scarification of the degenerated region of the tendon is performed using a 21G needle in order to produce a small amount of bleeding (Fig.13).
The latero-lateral direction of the needle allow its correct and continuous visualization in the soft tissues.
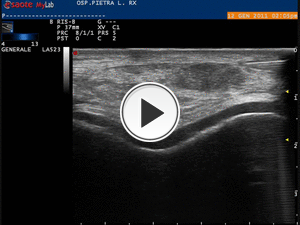
Fig. 13: Video showing the injection of local anhestetic with latero-lateral direction of the needle.
2- Afterward,
using the same needle,
6 ml of autologous PRP prepared from a previously collected sample (110 ml of venous blood,
collected from the patient a few hours before the procedure,
is centrifuged and activated in the laboratory) are injected into the degenerated region of the tendon under US-guidance (Fig.14).
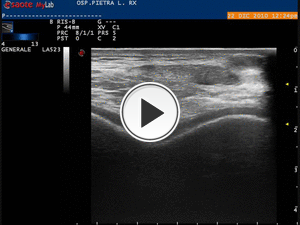
Fig. 14: Video showing the PRP injection with latero-lateral direction of the needle.
Post procedure care
The patient is kept in observation for at least 10 minutes.
A rest period of the affected structure and associated physical therapy are suggested.
ACHILLES TENDINOPATHY
Achilles tendinopathy is a condition thath affects 2.35/1000 in the adult populationwith age beetween 21 and 60 years.
It is condition sport-related in 59% of cases,
among all sports running involved in 53%; affects more males than females with a rate from 4:1 to 9:1.
It is thought to be caused by repeated tiny injuries,
microtrauma to the Achilles tendon.It is important to remember that middlethird of the Achille's tendon is more exposed to repetitive microtraumas,
leading to cronic degenerative changes without signs of peritendinous inflammation.
This condition can lead to major tendinous injuries and rupture.
The intinsic risk factor are drug assumption,
systemic diseases (autoimmune and inflammation condictions),
anatomical and biomechanical factors ( poor gastrosoleus flexibility,
malalignment,
overpronation of foot on heelstrike,
muscular imbalance,
overweight and obesity).
The estrinsic factors are training errors or not correct sports equipments.
Clinical presentation
Achille tendinopaty is a condiction with a progressive development over a period of weeks or months.
Interests mainly the middle third of Achilles region,
causing stabbing or burning pain in the Achilles tendon,
pain and stiffness in the Achilles region in the morning or after rest,
tenderness or lumps on palpation, constant pain with all exercises.
May be acute if partial tear.
Ultrasound diagnosis
The diagnosis is essentially clinical,
but has to be demonstrated with imaging techniques (plain films,
US,
MRI).
Ultrasound can easily detect fusiform swelling>7mm,
loss of fibrillar echotexture of the Achille's tendon and the increased vascularity on color Doppler.
US is a low cost tecnique that allows a High-resolution visualization and is an important guidance for treatment.
Treatment options
Conservative treatment includes rest,
weight loss,
physical therapy,
stretching, night splints,
ice,
decrease of sport activity,
orthotics supports,
anti-inflammatory and analgesic medications.
Interventional procedure include shockwaves,
steroid injection and surgical debridement.
Interventional procedure
Indication is symptomatic insertional enthesopathy of Achilles tendon.
Objective
To cause local hyperemia and bleeding into the tendon,
thus promoting post-procedural platelet-induced recovery phenomena.
Equipment
- 1 syringe (2–5 ml);
- 21G spinal needle;
- Local anesthetic (5-10 mL Lidocaine 2% without adrenaline);
- Long-acting steroid (1 ml,
40 mg/ml);
- Plaster.
How we do it
With the patient in prone position,
with the foot hanging over the edge of the table, perform an axial US scan of the Achille's tendon.
1) ANAESTHESIA: before a preliminary accurate disinfection,
that avoids infective complications,
we inject a small amount of local anesthetic in the peritendineous soft tissues (retrocalcanear bursa if low-sited degeneration) and in the tendon (up to 5mL Lidocaine 2% without adrenaline because is not needed since vascularity is quite poor,
with a 21G needle).
The axial scan of Achilles tendon and the latero-lateral direction of the needle allow its correct and continuous visualization in the soft tissues (Figg.15-16).
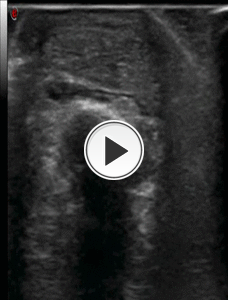
Fig. 15: Video showing the step No. 1 of the infiltrative procedure: retrocalcanear bursa injection of local anesthetic with axial scan of Achilles tendon and the latero-lateral direction of the needle.
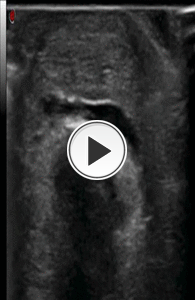
Fig. 16: Video showing the step No. 1 of the infiltrative procedure: peritendinous injection of local anesthetic with axial scan of Achilles tendon and the latero-lateral direction of the needle.
2) DRY NEEDLING: We perform with about 15-20 punctures of the tendon, a dry needling on the degenerated portion of Achilles tendon (Fig.17). Dry-needling leads to local hyperemia and slight bleeding,
that stimulates platelet activation and reparative phenomena induced by leukocytes.
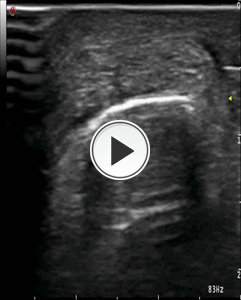
Fig. 17: Video showing the step n° 2 : dry needlind procedure on the Achille’s tendon with axial scan and the latero-lateral direction of the needle.
3) STEROID INJECTION: The needle is retracted to reach the peritendinous soft tissues and a small amount of steroid (1mL of triamcinolone acetonide 40mg/mL) is injected,
avoiding the tendon. Steroid is injected around the tendon and in the retrocalcanear bursa (Figg.18-19).
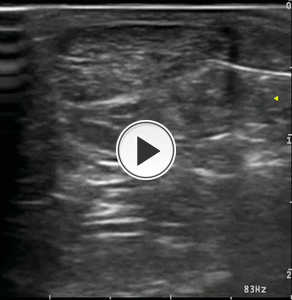
Fig. 18: Video showing the step No. 3 of the infiltrative procedure: peritendinous steroid injection.
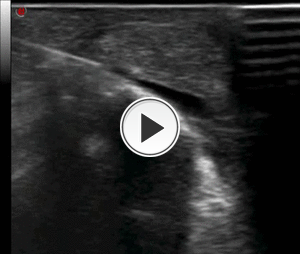
Fig. 19: Video showing the step No. 3 of the infiltrative procedure: retrocalcanear bursa steroid injection.
Post procedure care
The patient is kept in observation for at least 10 minutes.
A rest period of the affected structure and associated physical therapy are suggested.
PLANTAR FASCITIIS
Plantar fascitiis is a condiction that affects about 2 million people in the world (76% are runners) with an equal incidence between males and females,
especially in an age between 40 and 60 years,
it is reported to be the most common cause of inferior heel pain.
It is caused by repetitive microtrauma of the plantar fascia,
that plays an important role in the foot arch.
In 66% of cases is monolateral and in 74% of cases affects the medial band of the fascia.
The etiology of plantar fasciitis is poorly understood.
It is well known that
plantar fasciitis can occur in association with various arthritides, but in approximately 85% of cases the etiology is unknown.
Several risk factors have been hypothesized : anatomical (pes planus,
pes cavus,
shortened Ahille's tendon,
leg lenght discrepancy) biomecanical ( ecessive subtalar joint pronation,
weak muscles of the foot,
limited dorsiflexion of the ankle joint) enviromental (trauma,
hard surface,
prolonged weight bearing).
The principle risck factors are the overweight and the obesity certainly.
Clinical presentation
Patients with plantar fasciitis present pain when they take their first steps after they get out of bed or sit for a long time that improve after the first steps, pain and tenderness in the area of the medial tubercle of the calcaneus and pain that is generally worse while weight-bearing.
Ultrasound diagnosis
The diagnosis is essentially clinical,
but has to be demonstrated with imaging techniques (plain films,
US,
MRI).
Ultrasound can easily detect fascial thikening > 4mm,
loss of fibrillar echotexture and the increased vascularity on color Doppler.
US is a low cost tecnique that allows a High-resolution visualization and is an important guidance for treatment.
Treatment options
Conservative treatment includes rest,
weight loss,
physical therapy,
stretching,
ice,
decrease of sport activity,
orthotics supports,
anti-inflammatory and analgesic medications.
Interventional procedure include shockwaves,
steroid injection and surgical debridement.
Interventional procedure
Indication is symptomatic insertional enthesopathy of plantar fascia.
Objective
To cause local hyperemia and bleeding into the tendon,
thus promoting post-procedural platelet-induced recovery phenomena.
Equipment
- 1 syringe (2–5 ml);
- 21G needle;
- Local anesthetic (5-10 mL Lidocaine 2% without adrenaline);
- Long-acting steroid (1 ml,
40 mg/ml);
- Plaster.
How we do it
With the patient in prone position,
with the foot hanging over the edge of the table, perform an axial US scan of the plantar fascia.
1) ANAESTHESIA before a preliminary accurate disinfection that avoids infective complications We inject a small amount of local anesthetic in the perifascial soft tissues and in the fascia (up to 5mL Lidocaine 2% without adrenaline,
21G needle). The axial scan of plantar fascia and the medium-lateral direction (less painful approach) of the needle allow its correct and continuous visualization in the soft tissues (Fig.20).
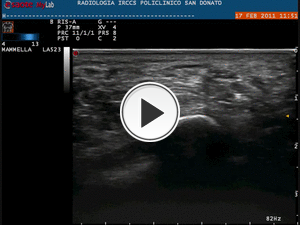
Fig. 20: Video showing the step No. 1 of the infiltrative procedure: injection
of local anesthetic with axial scan of plantar fascia and the medium-lateral direction (less painful approach) of the needle.
2) DRY NEEDLING: We perform with about 15-20 punctures of the tendon, a dry needling on the degenerated portion of the plantar fascia (Fig.21).
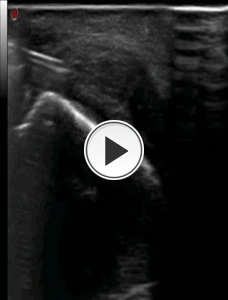
Fig. 21: Video showing the step n° 2 : dry needlind procedure on the plantar fascia with axial scan of plantar fascia and the medium-lateral direction (less painful approach) of the needle.
3) STEROID INJECTION: The needle is retracted to reach the perifascial soft tissues and a small amount of steroid (1mL of triamcinolone acetonide 40mg/mL) is injected,
avoiding the fascia.
Steroid has to be injected between the plantar fascia and the calcaneal fad pad (Fig.22).
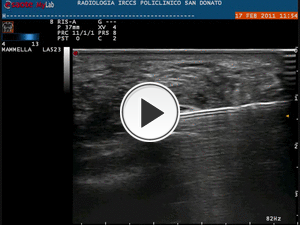
Fig. 22: Video showing the step No. 3 of the infiltrative procedure: steroid injection with axial scan of plantar fascia and the medium-lateral direction (less painful approach) of the needle.
Post procedure care
The patient is kept in observation for at least 10 minutes.
A rest period of the affected structure and associated physical therapy are suggested.