Study Design:
Imaging data (a total of 50 unreviewed acute non-contrast CT scans from 9 different hospitals),
were retrospectively selected from the CENTER-TBI imaging database.
This database is a dynamic entity into which images of neurotrauma patients are continuously uploaded by different European hospitals,
participating in the study.
A standard clinical protocol is used in all hospitals and images are initially reviewed by two non-radiologist raters (L.C.,
NS1 or T.VdV.,
NS2).
The studies are then again reviewed by one of two supervising expert neuroradiologists (G.W.,
NR1 or L.vdH.,
NR2).
Patients were anonymized by using a Global Unique Patient Identifier (GUPI) and reviewers were provided with a a limited amount of patient information (sexe and age).
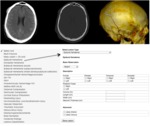
A digital structured reporting tool was developed with input from experts involved in the CENTER-TBI study (L.vdH,
P.M.P,
A.I.M) and in accordance with the most recent CDE templates (see example Fig.
1).
This tool,
embedded in the CENTER-TBI database,
allows structured and standardized reporting of essential qualitative and basic quantitative pathoanatomic features of TBI.
The two inexperienced non-radiologist raters (L.C and T.VdV.) were first trained by a neuroradiologist (L.vdH) to interpret CT images of TBI patients according to the NINDS CDEs.
A manual for CDE structured reporting was developed during a training period of around 400 CT data sets.
Experience of the supervising neuroradiologists ranged from 20 years for L.vdH to 30 years for G.W.
The two non-radiologist raters and one expert neuroradiologist (G.W,
NR1) independently reported pathoanatomic lesion types of the 50 acute non-contrast CT’s,
using the structured reporting tool.
Images were evaluated in a DICOM viewer on a large computer monitor (27”,
S27A850D).
Basic TBI features and scores on two classification scales (Marshall and Rotterdam,
see Fig.
3 and 4) were extracted from the reports,
similar to previous TBI inter-observer studies [11].
The pathoanatomic lesion types that were extracted included: skull fracture,
mass lesion (defined as a lesion over 25 cm3 in volume),
epidural hematoma,
subdural hematoma,
hemorrhagic contusion (defined as a mixture of non-hemorrhagic and hemorrhagic contused brain tissue),
intracerebral hematoma (defined as a homogeneous collection of intraparenchymal blood),
traumatic subarachnoid hemorrhage,
intraventricular hemorrhage,
traumatic and diffuse axonal injury,
midline shift (defined as a shift of over 5 mm),
cisternal compression and herniation.
All statistical analysis was performed in R (version 0.99.485).
Cohen’s kappa statistics were calculated to determine interobserver agreement of any abnormality and of the 12 individual pathoanatomic lesion types.
Weighted Cohen’s kappa statistics of agreement were calculated for scores on the Marshall and Rotterdam classifications.
Values below 0.2 indicate poor agreement,
between 0.21 and 0.4,
fair agreement,
between 0.41 and 0.6,
moderate agreement,
between 0.61 and 0.8,
good agreement and > 0.8,
almost perfect agreement [14].
Sensitivity,
specificity,
positive and negative predictive values and diagnostic accuracy were calculated to assess the diagnostic performance of the non-radiologist raters for each pathoanatomic lesion type.
The standard against which non-radiologists were compared was the evaluation of the expert neuroradiologist with the longest experience (G.W.,
NR1),
and when he was in doubt,
consensus opinion between the two expert neuroradiologists (G.W,
NR1 and L.vdH,
NR2).


![Fig. 3: The Marshall classification, reproduced from Maas et al. [12], 2007. References: Maas et al. 2007.](https://epos.myesr.org/posterimage/esr/ecr2016/134927/media/673647?maxheight=150&maxwidth=150)
![Fig. 4: The Rotterdam classification, reproduced from Maas et al. [13], 2005. The sum score, +1 is given to all patients because they have suffered a TBI. References: Maas et al. 2005.](https://epos.myesr.org/posterimage/esr/ecr2016/134927/media/673648?maxheight=150&maxwidth=150)
