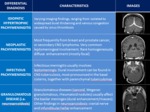
The starting point in the diagnosis of HI is an MRI study of the brain to look for the most pathognomonic signs (subdural fluid collections,
sagging of midbrain and pachymeningeal enhancement) and other associated findings that can confirm the clinical hypothesis.
More than two centuries ago,
Alexander Monro applied some of the principles of physics to the intracranial contents and hypothesized that the intracranial blood circulation was of constant volume at all times.
This hypothesis was supported by experiments by Kellie,
and underwent further modification in subsequent years.
The Monro–Kellie hypothesis,
as we know it today,
states that the sum of volumes of brain,
CSF,
and intracranial blood is constant (Fig 4).
This hypothesis has substantial theoretical implications in decreased CSF volume,
and many of the MRI abnormalities seen in intracranial hypotension or CSF volume depletion can be explained by the Monro–Kellie hypothesis.
- A decrease in CSF volume would require volume compensation
- BUT the brain volume remains nearly constant
- THEN it is the intracranial blood volume that will be affected
- Consequently,
a compensatory intracranial hyperemia occurs in intracranial hyotension.
Thus,
venous sinus engorgement,
abnormal pachymeningeal enhancement,
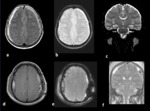
subdural effusions,
and enlargement of the pituitary gland occurring may represent compensatory changes to maintain intracranial volume in the face of CSF loss (2).
In CSF volume depletions,
subdural fluid collections participate in intracranial volume compensation,
even if the CSF leak site is far from the site of subdural collection (usually supratentorial) (Fig 5).
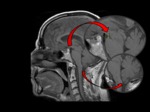
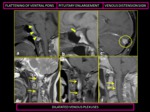
Depletion of CSF causes a reduction of the buoyant force that supports the brain,
allowing midline encephalic structures to sag and displace downwards (Fig 6,
7).
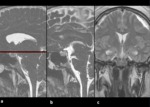
Another common finding is thickening and enhancement of pachymeninges.
It can be explained by the brain downward displacement resulting in tethering of some structures such as meninges,
venous sinuses and cranial nerves (Fig 8) (3).
Meningeal involvement,
with thickness and enhancement of meninges,
can be found in other conditions such as idiopathic hypertophic,
neoplastic and infectious pachymeningitis (4,
5,
6).
First thing to rule out other possible differential diagnosis is to determine whether it is a dural or a leptomeningeal enhancement,
because the latter is not very typical of IH and is more suggestive of an idiopathic or neoplastic or even infectious origin (Fig 9).
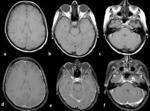
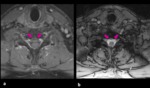
Other characteristic neuroimaging features of IH in brain MRI are engorgement of venous structures,
venous distension sign (7),
decreased mammillopontine distance,
pituitary hyperaemia,
but they can manifest in different ways.
These findings seem to be related to the same hystopathologic processes earlier described.
(Fig 10)
An interesting association has been reported in the literature between IH and superficial siderosis (SS): a dural pathology is the common denominator in both these conditions.
It seems that there might be a radiological overlap between SS and IH: classic neuroimaging manifestations of CSF hypovolemia such as pachymeningeal enhancement,
foreshortening of the suprasellar cistern,
sagging of the midbrain,
and flattening of the ventral pons have also been reported in some SS patients.
It has been suggested that these similarities are related to a common histopathology,
resulting from venous engorgement,
increased pial vascularity and microtrauma of the internal venous plexus in the epidural space by the CSF leak (8).
To sum up,
in order to study properly the main characteristic neuroimaging findings of IH,
brain MRI exam should include:
- T1 and T2-weighted sequences on the three orthogonal planes; sagittal plane is particularly necessary to demonstrate the downward displacement of midline encephalic structures.
- fluid attenuated inversion recovery (FLAIR) is useful to highlight subdural effusions.
- Venous angiography to show the engorgement of venous plexuses.
- Post-contrast T1-weighted images to detect pachymeningeal enhancement.
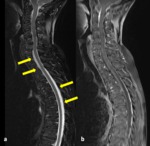
Once the diagnosis of IH has been confirmed,
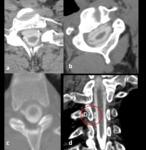
MRI study must be extended to the spine,
in order to investigate the presence of extradural fluid collections and engorgement of epidural venous plexuses (9) (Fig 11,
12).
Spine MRI should include:
- T2 fat-suppressed sequences,
both on sagittal and axial planes,
to allow an easier differentiation between extradural fluid collections and fat tissue in the epidural space.
- High resolution 3D-T2 sequences (CISS) to allow a better visualization of dilatated root sheath,
Tarlov cysts and meningeal cysts,
sometimes associated to IH (10).
- T1 post-contrast images to show distended venous plexus,
typically located in the anterior epidural space at C1-2 level and in the posterior plexus at the low lumbar and sacral level.
As previously discussed,
the detection of an exact leak site helps to guide a target-treatment,
with better outcomes than random blood patch.
Different methods are available,
such as CT myelography (MCT),
MR myelography (GdM) and radioisotope cisternography (Fig 13).
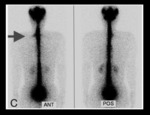
Lower sensitivity and lack of anatomical references make radioisotope technique the least used in the detection of CSF leaks (11,
12) (Fig 14).
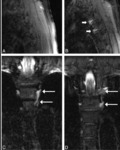
Conventional CT mielography detects the location of leak in most cases (Fig 15).
However fast-flow CSF fistulas are usually missed with conventional MCT.
When a fast CSF leak is expected,
dynamic MCT technique is more adequate: it consists of a CT mielography performed during contrast injection to obtain an immediate acquisition.
The latter is more invasive and has a higher radiation dose,
but a significant association has been reported between extra-arachnoid fluid on premyelographic spinal MR imaging and the presence of a fast CSF leak,
and some authors propose to proceed directly to dynamic study rather than conventional CTM in patients with spinal extradural CSF collections noted on MR imaging (13,
14).
MR- and CT- myelography are reported as complementary in the detection of CSF leaks.
Though intrathecal injection of gadolinium contrast is currently off-label, some authors believe it is an acceptable risk for a selected patient population.
This is particularly true in case of intermittent leakage that is missed with MCT,
or in some fast CSF leaks in high thoracic region,
where CTM is potentially degraded by artifacts from the shoulders (15,
16) (Fig 16).