Normal 0 false false false EN-US X-NONE AR-SA
The study included 140 patients.
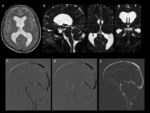
Group 1: Obstructed ventricular foramina.
(Fig.
3,4,5,6)
Nine patients were presented by headache.
Seven of them showed thin webs obstructing foramen of Monro.
While,
two patients showed fourth ventricle outlet obstruction by webs and subarachnoid synechiae at the basal CSF cisterns with obliteration of the foramina of Luschka.
If one of the fourth ventricular outlets appeared obstructed,
the hydrocephalus was categorized as non-communicating.
Identification of obstructive pathologic processes at any level through the CSF pathway is of significant importance because it can change the mode of management in patients with hydrocephalus,
avoiding shunt insertion.
Advantage of 3D-CISS has been not only demonstrating the membranes within the prepontine cistern,
but also guiding the neuroendoscopic procedure,
showing the location,
number,
and the extent of the membranes.
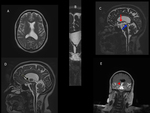
Group 2: CSF obstruction at the aqueduct of Slyvius.
(Fig.
7,8,9,10)
This group includes eighteen patients.
All patients showed dilated ventricles (lateral,
third) and foramina of Monro proximal to obstruction,
and normal fourth ventricle and foramina (Luschka,
Magendie) distal to obstruction.
Fifteen of them showed total aqueduct obstruction by web (post intracranial infection or hemorrhage or idiopathic) and by tectal plate compression by a large quadrigeminal cistern arachnoid cyst.
The other three patients showed partial aqueduct obstruction with weak CSF flow demonstrated in PC sequence.
Group 3: Communicating hydrocephalus.
(Fig.
11,12,13,14)
Fifteen patients demonstrated diffuse hydrocephalus with no obstructing point detected at either the aqueduct or ventricular foramina.
Eight of these patients had history of intracranial hemorrhage or infection and showed thin membranes traversing the basal CSF cisterns in 3D CISS images.
The membranes that obscured only the cisterns (ie,
interpeduncular,
prepontine,
and premedullary) were defined as communicating hydrocephalus with cisternal obstruction.[4]
In the other seven patients no obstructing point or pathological subarachnoid membranes could be detected.
Group 4: Arachnoid cyst (communicating vs non communicating).
(Fig.
15,16,17,18,19)
Arachnoid cysts are classified as communicating or non-communicating according to their relation to the subarachnoid space.
There has been considerable controversy regarding the indications for the surgical treatment of asymptomatic ACs.
However,
there is a consensus that patients with symptomatic cysts causing seizures,
hydrocephalus,
increased intracranial pressure,
or neurologic impairment should be managed surgically.
Two surgical approaches are encountered; the first is cyst excision or fenestration into the subarachnoid space,
basilar cisterns,
or ventricles.
The second is cystoperitoneal shunt placement.
Determination of communication between the arachnoid cyst to the CSF space is important in the preoperative evaluation.
CT cisternography (CTC) was the most widely used diagnostic method to show such communication.
But this method is considered invasive and using contrast material injected intrathecal.
Flow-sensitive cine MR imaging techniques have been increasingly used to investigate the flow characteristics of CSF and its use is also increasing as a reasonable noninvasive diagnostic method alternative to CTC for the functional evaluation of ACs.[6]
PC MRI may improve the diagnostic confidence in differentiating communicating and non-communicating arachnoid cysts and posterior fossa cystic malformations from each other.
Twelve patients had arachnoid cysts communicating with the subarachnoid space.
Ten patients had non-communicating arachnoid cysts.
Normal 0 false false false EN-US X-NONE AR-SA
Group 5: hydrocephalus in elderly with dementia.
(Fig 20,21,22,23,24,25)
Classic symptom of normal pressure hydrocephalus: triad of gait disturbance,
urinary incontinence and dementia.
Ventriculomegaly with altered CSF dynamics and normal CSF pressure,
but a slight pressure gradient persists between the ventricles and the brain parenchyma.
In properly selected patients,
ventricular shunting results in resolution of symptoms and slows progressive deterioration.
The aim of ventriculoperitoneal shunting is not to decrease mean pressure,
but to dampen the pulse pressure by providing extra capacitance to the ventricular system.
Radiological findings:
• Ventricles and Sylvian fissures symmetrically dilated out of proportion to sulcal enlargement.
• Normal hippocampus (which distinguishes NPH from atrophy).
• Upward bowing of corpus callosum.
• Increased or normal CSF flow void.
• Periventricular high signal,
primarily anterior to frontal horns or posterior to occipital horns of lateral ventricles (transependymal CSF flow).
Twenty five patients were presented by gait disturbance and clinical suspicion of iNPH. By evaluating the conventional and CSF flow imaging,
twenty patients showed radiological findings of iNPH and presented by hyperdynamic CSF flow across the aqueduct,
while five patients were matching with vascular dementia and brain atrophic changes. These patients showed significantly decreased CSF flow and low stroke volume through the aqueduct as compared with NPH patients.
Normal 0 false false false EN-US X-NONE AR-SA
Group 6: posterior fossa malformations with CSF flow disorders.
Cystic posterior cranial fossa malformation as Dandy Walker malformation,
Dandy Walker variant,
Blake's pouch cyst and mega cistern magna have different distinct CSF flow patterns which are different from that of arachnoid cyst.
[7]
Nineteen patients showed variable posterior cranial fossa cystic malformation.
Two patients had DWM,
three had DWV,
three patients had Blake's pouch cyst,
seven patients had MCM and four patients had non-communicating posterior fossa arachnoid cyst.
Normal 0 false false false EN-US X-NONE AR-SA
Posterior fossa arachnoid cyst: (Fig.
26,27)
o Could be communicating or non-communicating.
Blake pouch cyst: (Fig.
28,29,30)
o Failure of regression of the normally transient Blake's pouch which is itself secondary to imperforation of the foramen of Magendi.
o BPC communicates with the 4th ventricle and separate from the subarachnoid space (not communicating with the posterior cervical subarachnoid space).
o Normal position of tentorium.
o Normal sized posterior fossa.
o Tetra ventricular hydrocephalus.
o Displaces 4th ventricle choroid plexus into the superior cyst wall.
o Consistently associated with hydrocephalus.
DWV: (Fig.
31,32)
o Less vermian hypoplasia and cystic dilation of the 4th ventricle.
o No enlargement of the posterior fossa.
o Prominent retrocerebellar CSF space that communicates freely with a normal or dilated 4th ventricle.
o CSF flow communicating between the large retrocerebellar cistern and the posterior cervical subarachnoid space.
o CSF flow through the aqueduct is slow.
o Hyperdynamic flow along the aqueduct and 4th ventricle.
(unlike arachnoid cyst).
DWM: (Fig.
33,34)
o Large posterior fossa and superiorly displaced tentorium.
o Cystic dilated fourth ventricle.
The dilated fourth ventricle fills the posterior fossa and extends into the cisterna magna which is compressed between the dilated 4th ventricle and the dura.
o Complete or partial vermian agenesis.
o The cystic CSF collection is not communicating freely with the basal cisterns.
o No flow between the cistern magna and posterior cervical subarachnoid space.
o Hydrocephalus is present in 90% of cases.
o Posterior fossa cyst can be shunted directly.
o If aqueduct stenosis or occlusion present,
separate ventriculoperitoneal shunt is also placed.
o Preoperative MRI CSF flow is required to assess aqueduct patency.
Patent aqueduct with CSF flow from the aqueduct to the posterior fossa cyst.
Normal 0 false false false EN-US X-NONE AR-SA
Chiari 1 malformation: (Fig.
35,36)
Small sized posterior cranial fossa associated with pointed cerebellar tonsil herniated through the foramen magnum and consequently compromise or alter the normal CSF flow dynamic at the foramen magnum.
Symptomatic patients may present with headaches,
dizziness,
ataxia,
fainting with a cough,
weakness or numbness,
episodic aural fullness,
tinnitus and vertigo.
Selection criteria for surgery depends on patient symptoms and the degree of tonsillar ectopia and CSF flow obstruction.
The degree of CSF flow obstruction rather than the degree of tonsillar herniation can better select patients who are most responsive to surgery.
Improved CSF velocity profile following surgery in such patients is useful in anticipation of symptomatic improvement.[8]
Eight patients in our study had Chiari 1 malforamtion.
All patients were presented by headache mainly occipital.
Five patients had syringomyelia.
Normal 0 false false false EN-US X-NONE AR-SA
Group 7: post management of hydrocephalus
ETV:
Successful technique in treating occlusive hydrocephalus caused by primary or secondary aqueductal stenosis and space-occupying lesions of the midbrain,
the pineal region or the posterior fossa.
But it is thought to be less effective in patients with hydrocephalus caused by intraventricular or subarachnoid hemorrhage,
in patients with meningitis,
in pediatric patients with associated spinal dysraphism and in normal pressure hydrocephalus.[9,
10]
A range of image parameters have been assessed to evaluate the permeability of the ETV including:
• Ventricular size changes.
But it is not a good indicator of ventriculostomy patency.
• Flow void signal intensity.
• Stroke volume measurements by using cine phase-contrast MR.
PC CSF flow sensitive MRI imaging offers more physiologic data than structural MR images and qualitative assessment of the patency of ventriculostomy.
Sagittal Technique: For qualitative assessment of CSF flow.
Axial Technique: For quantitative assessment with an imaging plane perpendicular to the ventriculostomy.
A decrease in stroke volume during the follow-up was associated with ventriculostomy failure and clinical deterioration.[9]
In this study,
sixteen patients evaluated for ETV function:
o Twelve patients showed fair to good functioning ETV.
(Fig.
37,38)
o Four patients showed non-functioning ETV and no clinical improvement.
(Fig.
39)
Ventriculoperitoneal shunt : (Fig.
40)
Clinical assessment is the primary method for assessment of the VP shunt functioning.
PC MRI can also be used to evaluate VP shunt patency.
In shunt catheters,
because of the one-way valve mechanism,
normal flow is unidirectional and rhythmic.
No signal means no flow in PC imaging.
Because of very low CSF flow rates in shunt catheters,
minimum VENC values (2–5 cm/s) should be used for assessing VP shunts.
Eight patients were assessed for a non-functioning ventricular shunt device,
none shows CSF flow signal at the shunt tip in PC MRI,
this was clinically correlated.