PAVMs in patients with hereditary hemorrhagic telangiectasia
Hereditary hemorrhagic telangiectasia (HHT),
or Osler-Rendu- Weber disease,
is an autosomal dominant disorder characterized by recurrent epistaxes,
mucocutaneous telangiectasias,
and visceral involvement including arteriovenous communications that may develop in virtually any organ,
especially in the lung(3).
Clinicians may diagnose HHT using a consensus clinical diagnostic criteria known as the Curaçao Criteria or by identification of a causative mutation through genetic testing for HHT(4).
Diagnosis of HHT using the Curaçao Criteria is considered “definite” if three or more of the following criteria are present,
“possible or suspected” if two criteria are present,
and “unlikely” if 0 or 1 criterion is present(4): (a) Spontaneous and recurrent epistaxis; (b) Multiple mucocutaneous telangiectasias: characteristic sites: lips,
oral cavity,
fingers,
nose; (c)Visceral lesions: gastrointestinal telangiectasia,
pulmonary,
hepatic,
cerebral or spinal arteriovenous malformations; (d)Family history: a first-degree relative with HHT according to these criteria or genetic mutation
Superficial telangiectases attributable to HHT are the most common and frequently the only physical finding in patients with PAVM(1).
Although AVMs can be seen in virtually any organ,
lesions are especially common in the gastrointestinal tract,
lung,
liver,
and brain.
If there are missing criteria,
screening for visceral involvement is critical.
PAVMs are abnormal vessels in which there is a direct communication between the pulmonary arterial circulation and the pulmonary venous circulation without interposition of a capillary bed(1,2).
They have a wide range of histologic features,
from diffuse telangiectasia to large complex structures consisting of a bulbous aneurysmal sac between dilated feeding arteries and draining veins(6).
Direct capillary-free communications between the pulmonary and the systemic circulation have three main clinical consequences: (a) Pulmonary arterial blood passing through these right-to-left shunts cannot be oxygenated,
a situation that may lead to hypoxemia.
(b) The absence of a normal filtering capillary bed allows particulate material (air bubbles or clots) to reach the systemic circulation directly (paradoxical embolism),
with potential clinical sequelae in the cerebral circulation (transient ischemic attack,
stroke,
brain abscess).
(c) These abnormal vessels,
particularly in pregnant women,
may rupture into the bronchus (hemoptysis) or the pleural cavity (hemothorax)(12).
This way,
PAVMs becomed one of the main causes of morbidity in patients presenting with HHT disease(2).
The most frequently reported complications relate to the central nervous system and are seen in about 30% of patients.
Less common but potentially life-threatening complications include hemothorax and hemoptysis(1).
Unfortunately,
clinical signs and symptoms of PAVMs are often absent before the development of complications(8).
Such severe complications may be the presenting manifestation leading to diagnosis of the PAVM and even of HHT itself(3).
Contrary to sporadic forms,
PAVMs in HHT patients (HHT-PAVMs) are often multiple and bilateral(1,2).
Moreover,
some of these patients have a more severe and diffuse pattern of disease,
usually presenting a more severe hypoxemia and a higher rate of neurologic complications than those with localized PAVMs(2,11).
Numerous or diffuse PAVMs are more common in the setting of HHT.
The diffuse type of PAVM introduces additional challenges to PAVM management,
not only from an embolotherapy standpoint but also during follow-up(5).
Since PAVMs are for the most part encountered in HTT patients,
once a patient is identified as having HHT,
or being at risk for HHT but lacking informative genetic testing,
the principal initial goals are to identify and mitigate such risks to the patient’s health as PAVMs(5,8).
Agitated saline contrast transthoracic echocardiography (TTE) is the study of choice for screening patients with suspected HHT for PAVMs.
In patients who have abnormal bubble echocardiography,
CT with multiplanar reconstruction is the study of choice(2,4,5,9).
Untreated or occult PAVMs can increase in size over time,
with increasing shunt volumes inducing hypoxemia,
together with an increased risk of stroke and PAVM rupture(10).
For patients with only small untreated PAVMs and in patients with suspected microscopic PAVMs (for example,
detected on TTCE but not detectable on CT),
a follow-up period should be determined on a case by case basis (approximately every 1- 5 years) with multidetector CT (MDCT) of the thorax,
with consideration for limiting radiation exposure(4).
Management of PAVMs relies mainly on prophylactic administration of antibiotics before dental and surgical procedures to reduce paradoxical embolic abscess risks and percutaneous transcatheter embolization (TCE),
also known as percutaneous image-guided embolotherapy or “embolotherapy”,
which has become the first-line treatment as an alternative to surgery(2,4,8,11).
PAVMs should be treated before the complication may appear owing to the high complication(2).
TCE is the recommended treatment for all adults and symptomatic children with PAVMs(8).
After transcatheter treatment,
a follow-up examination is recommended in 6 to 12 months and every 3 years thereafter with thoracic MDCT in order to identify embolized PAVMs that have reperfused and other PAVMs that have grown large enough to be considered for embolization(4).
Contrast echocardiography is not useful for follow-up after TCE,
as it remains positive in up to 90% of patients despite successful occlusion of all angiographically visible vessels(4,8).
Particularly,
in patients presenting diffused forms of or a multiple for,
this positive result can be explained by the detection of microscopic PAVMs that cannot be detected on CT,
whose size does not require treatment,
and whose afferent artery would be too small for a selective embolization(2,9).
This fact has important long-term implications for these patients.
As noted,
measures such as the continued need to take antibiotic prophylaxis for dental work and similar procedures and IV catheter bubble precautions remain in effect throughout the patient’s life(5).
After TCE,
thoracic contrast-enhanced CT plays a key role depicting for to procedure-related complications during the early clinical follow-up,
and evaluating embolization success or insufficient treatment sings during the long- term follow-up(2,9).
CT angiography of PAVMs
Catheter-based pulmonary angiography and later the digital sustraction angiography (DSA) used to be the gold standard in the diagnosis of PAVM and it was usually necessary if resectional or obliterative therapy was considered.
Angiography had to be performed on all portions of the lungs looking for unsuspected PAVM,
and intra and extrathoracic vascular communications were searched for at the same time.
Moreover,
in order to detect PAVMs and accurately define the angioarchitecture of individual lesions amenable to embolotherapy,
supplemented hyperselective angiograms were necessary(1).
Fortunatelly,
there has been marked change in this step over the past decades,
paralleling diagnostic CT and endovascular treatments evolution. Nowadays MDCT provide not only the diagnosis but also a high anatomical resolution allowing to plan and guide therapy,
by evaluating the number of PAVMs,
their location,
the size of the afferent artery,
the aneurysmal sac,
and depicts the precise angioarchitecture of the PAVMs.
Furthermore,
this tool is useful for follow-up after percutaneous embolization(2,5,9,13).
The overall accuracy of CT in the diagnosis of pulmonary vascular disorders increases with administration of a contrast material bolus(10).
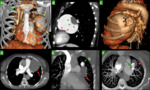
CT angiography (CTA) allows for high quality maximum intensity projections (MIP) and Three-dimensional (3-D) reconstructions,
with better assessment of vascular structures (feeding artery,
aneurysmal sac and draining vein).
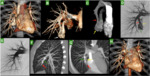
Three-dimensional images are specially helpful when evaluating large PAVMs or lesions with more than one feeding artery in planning for TCE(10) (Figures 1A,
1B,
1C,
2A,
2B and 3A).
CTA also allows a better characterization of the aneurysmal sac (the lumen can be spontaneously thrombosed before treatment),
detection of PAVMs systemic supply and gives information about subphrenic specific visceral involvement(2).
Moreover,
intravenous contrast medium injection may be neccesaty owing to the potential underestimation of catheterization difficulties on unenhanced CT scanner examination(2).
Valuable information about the upper abdomen,
such as the presence of extrapulmonary AVMs,
would not be obtained without contrast(5).
Hepatic,
gastrointestinal,
pancreatic,
or renal AVMs also may be seen and lend support for the diagnosis of HHT.
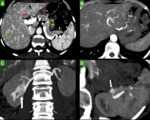
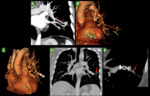
In patients presenting without PAVM,
identification of visceral involvement provides the third visceral diagnostic item and subsequently helps to early clinical diagnosis and improved management of HHT disease(2)(Figure 4).
The potential for growth of new PAVMs,
and/or late recanalization of treated PAVMs necessitates follow-up imaging using contrast-enhanced chest CT(8) Contrast CT scanner is the key follow-up examination after PAVMs treatment,
providing aditional infomation regarding embolization success or insufficient treatment sings during the long-term follow-up(2,7-9).
Although,
There remains a lack of agreement even among centers as to whether CECT is necessary or whether unenhanced studies are required,
both at the screening step and during follow- up(4,5,14,15).
CECT may provide information beyond unenhanced scanning(2,5-11).
At the authors’ institution,
we perform a thoraco-abdominal CTA examination at the time of initial screening.
Three-dimensional and MIP reconstructions of the thoracic vascular structures.are obtained.
Our CT technique is essentially a pulmonary embolus protocol.
A 20-gauge intravenous catheter is inserted into the basilic vein.
Nonionic iodinated contrast material at a dose of 0.8–1 mL/kg is infused by using an automatic injection pump at a rate of 4.0 mL/s; this injection is followed by an injection of 30 mL of saline solution.
The scanning delay after initiation of the contrast material injection is determined by using a bolus-tracking method.
The region of interest is positioned at the pulmonary trunk,
and the threshold for the MDCT angiography is set to 100 HU.
Owing to the presence of a right-to-left shunt through the PAVMs,
specific attention is directed toward excluding residual air bubbles in the syringe of the power injector and its connecting tube before administration of contrast material.
From each data set,
multiples series of images are systematically reconstructed to depict the precise angioarchitecture of PAVMs.
Three-dimensional reconstructions are generated at commercially available workstations.
PAVMs Embolotherapy
Radiology clinician should be familiar with conventional and most frequent alternatives embolization techniques in order to help the interventional radiologist in procedure planning.
PAVM treatment is challenging even in the most experienced hands(5).
The aim of transcatheter embolization is to occlude all the PAVM feeding arteries by a selective catheterization by using a coaxial system,
via a percutaneous femoral approach(2).
The prevalent technique has been embolization of the feeding artery by using a variety of mechanical agents,
mainly coils and more recently self expandable nitinol plugs (vascular plugs)(15).
The feeding pulmonary artery must be occluded as close to the aneurysmal sac as possible to prevent reperfusion(11).
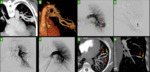
The major technical hurdle of this procedure is safely deploying an embolic device in position to occlude the lesion despite the torrentially high flow that can encourage passage into the systemic arterial circulation(8)(Figures 1D,
1E,
2C,2D,2E,2F,3A and 3B).
The technique of coil embolotherapy involves localization of the PAVM followed by selective catheterization of the feeding artery.
The catheter tip is advanced past the point of any proximal vessels that supply normal lung parenchyma and positioned as close to the neck of the PAVM as possible.
A coil advanced through the catheter and released at this point.
Additional coils are positioned if needed until blood flow to the PAVM has ceased(1).
Variables associated with recanalization include embolization > 1 cm from the aneurysm sac,
use of a single coil,
and large feeding arteries(8).
For achieving an effective occlusion of the PAVM with feeding artery embolization,
interventional radiologist should embolize close to the arteriovenous connection (preferably within 1 cm and beyond the origin of any significant supply to normal lung that may arise from this vessel) and create a dense,
cross-sectional occlusion.
When using coils,
it is important to achieve dense packing of them for a durable result(15) (Figure 3C).
The coils should be deposited in the afferent artery as near as possible to the aneurysmal sac so as to limit the risk of reperfusion by collateral branches(5,9).
When the pedicle is short,
rupture of the aneurysmal sac whose wall is fragile should be avoided(9). Usually multipedicular PAVMs are more difficult to treat from the technical point of view(9).
A definitive treatment with endovascular therapy requires occlusion of both the pulmonary and the systemic PAVM feeding arteries(7).
When using the traditional “packing technique”,
the firs coil is voluntarily oversized as to prevent paradoxical migration during deployment,
and the following coils are positiones at the center of the first coil deployed,
so as to obtain complete oclussion of the vascular lumen(9).
Sizing measurements to guide the choice of embolic device should be confirmed during catheter-based pulmonary angiography,
as CT evaluation alone may cause an unacceptable underestimation of the diameter of the feeding artery(8).
The vascular plug is a particularly useful tool for creating a wall in the distal feeding vessel behind which one may deploy multiple small coils.
Perhaps the greatest advantage of using this device is the ability to retract it if sizing or positioning is not optimal (Figures 1E,
2D,
2E).
After deploying the initial Amplatzer plug,
additional coils are usually placed behind the plug in the more proximal feeding artery to ensure complete embolization(8).
Venous sac embolization has been also proposed as an alternative to feeding artery embolization when the latter is problematic because of a short feeding artery or a draining vein larger than the artery,
in which case the risk of paradoxic migration may be higher(15).
This option is not feasible in patients with diffuse disease,
although this problematic group fortunately represents a small minority(15).
When performed by experienced operators as the prime treatment,
percutaneous embolization of PAVMs,
is a safe,
efficient and sustained therapy in the great majority of HHT patients (2,15).
Preprocedural evaluation
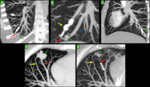
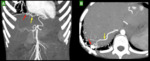
CTA allows a detailed 3D analysis of the PAVM(s) to be treated facilitating planning for the embolization procedure well in advance.
This is particularly important in large or complex PAVMs where ensuring that all of the required equipment is available before the procedure starts is critical to success(5,8) (Figures 5 and 6).
Having this information available in advance may allow the entire catheter- based procedure (any needed diagnostic arteriography and the embolization) to be performed in a single session in most patients(5).
The PAVM consists of three different anatomical components: one or more than one feeding artery(ies),
an aneurysmal sac and one or more draining veins whose diameter is usually larger than the afferent feeding artery(2,16).
Individual PAVM are typically from 1 to 5 cm in size,
although they are occasionally in excess of 10 cm(1).
They can be several in number and either unilateral or bilateral(9).
Most of them have a subpleural localization(2) and may have one of three typical appearances: (1) a large,
single sac,
(2) a plexiform mass of dilated vascular channels,
or (3) a dilated and often tortuous direct communication between artery and vein(1).
Uncommonly,
there may be mural thrombi or mural calcifications(1).
CTA with multiplanar reconstructions and detailed 3D analysis provides the diagnosis,
confirming the vascular nature of the lesion,
and allows to depict the precise angioarchitecture of the PAVMs(2,5-11,13).
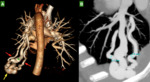
A simple PAVM can have one or several afferent arteries provided that they all stem from the same segmental artery(Figures 1,5 and 7).
In complex PAVMs,
the afferent arteries stemmed from at least two segmental arteries(9)(Figures 2,3 and 6).
Complex PAVMs constitute approximately 10% of lesions,
and a subset of these (approximately 5% of complex PAVMs) has been described as diffuse-type(5).
There is currently no consensus regarding the definition of diffuse PAVMs(11).
The diffuse pattern of PAVMs is variable defined as PAVMs involving every segmental or subsegmental artery of one lobe,
or one segment diffusely involved(2,11) (Figure 3).
A more clinically relevant classification has been proposed,
as follows: patients presenting with involvement of every subsegmental artery of at least one lobe (diffuse subsegmental form),
patients presenting with involvement of every segmental artery of at least one lobe (diffuse segmental form),
and patients presenting with a combination of subsegmental and segmental involvement of at least one lobe (diffuse mixed segmental/subsegmental form)(2,11).
This classification of patients with diffuse PAVMs seems to be clinically relevant to define long-term prognosis(11).
Characterization of the PAVMs,
particularly the size of the feeding artery,
is one of the key steps in CTA pre embolization.
Traditionally there were a size cut-off of 3 mm of diameter of the feeding artery for treatment of PAVMs,
based on an observation that the risk of cerebral infarction was low when the size of the feeding artery was <3mm,
as well as historical difficulty catheterizing arteries smaller than 3 mm in diameter(8).
However,
more recent data have shown no evidence of association between PAVM feeding artery diameter (or any other measure of PAVM severity) and risk of ischemic stroke or cerebral abscess(8).
Nowadays,
given the development and the current armamentarium of microcatheters and embolic agents,
many interventional radiologists now can treat even challenging PAVMs with feeding arteries smaller than 3 mm in diameter.
Although in practice it is uncommon to treat feeders under 1.5 mm(8).
Measure of the feeding artery beyond the segmental portion of the vessel,
and proximal to the nidus when evaluating a patient for TCE facilitates procedure planning in advance (Figure 6B).
Accurate assessment of pulmonary AVM feeding artery diameter is important for embolization device selection(2).
In cases of complex PAVMs,
the number and diameter of the feeding arteries must be depicted.
Besides,
the feeding artery may be long,
or very short,
thus making percutaneous embolization more challenging(2,15).
The aneurysmal part of the PAVM may be either a sac or a serpiginous network.
Distinction between these two different patterns is crucial to plan the appropriate percutaneous treatment as the risk of accidental embolization device migration to the left heart is higher when considering a single aneurysmal sac directly communicating with the left circulation(2)(Figures 1,
5 and 6).
Feeding artery embolization may be problematic in cases of a short feeding artery or a draining vein larger than the artery,
in which case the risk of paradoxic migration may also be higher(15).
A short pedicle may also increase the risk of rupture of the fragile wall of the aneurysmal sac(9).
The incidence of thromboembolic disease increase in HHT patients and can be associated with thrombus formation inside(17),
with the concomitant increased risk of paradoxical embolism during TCE.
Aneurimal sac lumen must be evaluated in orden to dectect the presence of thrombus formation inside.
The aneurysmal sac lumen can be spontaneously thrombosed before treatment(2,8).
CTA allows to depict for the presence of complications (e.g.
thrombosis)(2,8)(Fig 10).
Although the majority of PAVMs are fed by the pulmonary arteries,
systemic arterial supply to PAVMs via the bronchial,
inferior phrenic,
musculo-phrenic,
internal mammary,
or intercostal arteries is not uncommon,
particularly after prior treatment(7,8)(Fig.
12).
Careful evaluation of a preprocedural CTA allows for determination of several usefull parameters in the pretherapeutic evaluation (table 1).
|
TABLE 1.
PREPROCEDURAL EVALUATION
|
|
Confirm diagnosis (confirmin the vascular nature of the lesion)
|
|
PAVMs repartition (number and location of PAVMs)
|
|
Angioarchitecture characteristics of the PAVMs
· Type: simple,
complex or diffuse type
· Size of the PAVM (aneurysmal sac)
· Morphology of the aneurysmal sac (sac or serpiginous network)
· Characterization of the feeding arteries
1. Number
2. Size (diameter,
measured beyond the segmental portion of the vessel proximal to the nidus)
3. Morphology (percutaneous embolization may be more challenging in very short feeding arteries)
· Number,
morphology and size draining veins
|
|
PAVMs systemic supply (Depict for systemic arterial supply to PAVMs: bronchial,
inferior phrenic,
musculo-phrenic,
internal mammary,
intercostal arteries,
etc)
|
|
Aneurysmal sac lumen (presence of trombosis)
|
|
3-D reconstruction of larger lesions in preparation for treatment (especially helpful in malformations with more than one feeding vessel)
|
|
Subphrenic specific visceral involvement (screening for extrapulmonary AVMs,
liver and/or pancreatic involvement)
|
Of note,
the interpreting radiologist should also understand that a steal phenomenon may occur due to the PAVMs high blood flow output in a low-resistance vascular system,
and to the positive pressure gradient directed from the feeding arteries to the lesion center.
As such,
the afferent feeding arteries,
particularly the PAVM accessory feeders,
may thus be overlooked on imaging studies (not opacified on CTA or DSA)(2,8).
Follow-up after embolotherapy
Contrast-enhanced CT scanner examination is the key follow-up examination in this patients(2,7-9).
Immediate complications after treatment are rare in experienced hands(2,8).
Self-limited pleurisy is the most common postprocedural complication,
occurring in up to 10% of patients and attributable to thrombosis of the feeding artery and/or pulmonary infarction.
More serious and uncommon immediate postprocedural complications are related to systemic arterial embolization of clot,
air,
or the embolic device (device migration) and stroke(2,8).
Regarding mid to long-term follow-up,
persistence or reperfusuion of the PAVM is usually reported in no more than 8% of lesions although some have found higher rates of 25%–58%(15).
Persistence or reperfusion of an apparently successfully embolized pulmonary AVM may occur by one or more of several mechanism: (1) recanalization of the embolized vessel (through or around embolization material); (2) incomplete occlusion of the embolized vessel; (3) previously unrecognized additional feeding artery(ies) or growth of a missed or previously small accessory feeding artery; (4) development of systemic perfusion of the PAVM (reperfusion by bronchial or other systemic collateral flow into the feeding pulmonary artery beyond the level of the embolization),
(5) recruitment of side pulmonary artery branches (pulmonary artery to pulmonary artery collateral flow around the embolized vessel)(2,5,6,7,8,11,15,18).
Recanalization is the most common mechanism of reperfusion(15)(Figures 8A,8B and 8C).
The risk of collateral reperfusion about the embolized site from small pulmonary branches that have grown or developed over time may be greater with young children,
whose lungs are still developing(15).
Whenever possible,
postprocedural CTA images must be compared with preprocedural CTA images (Figures 8D and 8E).
Focus should be in evaluating outcome of embolized PAVMs and in search for new or enlarged lesions.
Basessed on the appearance of embolized PAVMs CT angiograms,
several parameters can be evaluated to define the success of the procedure(2,6-9,11,14-16, 19,20)(table 2).
|
TABLE 2.
FOLLOW-UP AFTER EMBOLOTHERAPY
|
|
Search for new or enlarged PAVMs
|
|
Evaluate embolized PAVMs.
Look for:
· Size of the nidus (aneurysmal sac): enlarged,
unchanged,
decreased,
or no longer detectable (It might be considered to be substantially modified if a greater than 30% change in diameter is observed)
· Size of the draining veins (compare with pre treatment TCA).
· Perfusion: present or lack of enhancement in the aneurysmal sac,
feeding artery beyond the point of occlusion (proximal to the nidus) and adjacent draining vein
· Presence of thrombosis in the aneurysmal sac
|
|
Depict for recruitment of adjacent pulmonary arteries or systemic arterial supply to PAVM
(bronchial,
inferior phrenic,
musculo-phrenic,
internal mammary,
intercostal arteries,
etc)
|
Follow-up CT scanner examination may highlight PAVM reperfusion,
indirectly by showing incomplete PAVM retraction,
and directly by using intravenous contrast injection(2,14,15,20).
Aneurysmal sac may be enlarged,
unchanged,
decreased,
or no longer detectable after treatment.
It might be considered to be substantially modified if a greater than 30% change in diameter is observed(6).
Morphologically,
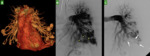
disappearance or marked reduction to a presumably thrombosed or fibrotic remnant is the desired appearance by 6–12 months (Figures 2G,
2 H,
7D,7E,
8E).
Lack of sac and adjacent draining vein enhancement are additional criteria when using contrast agents(15)(Figures 1F,1G,
5D,
5E,
5F).
After embolization,
the feeding artery beyond the point of occlusion as well as the nidus should shrink in size and lack enhancement(8,9,15,19).
When present,
the type of reperfusion may also be established by CTA and defined as: (1) Recanalization through or arround the coils (Figures 8A,
8B,
and 8C),
(2) recruitment of side pulmonary artery branches,
(3) or systemic artery supply (bronchial or nonbronchial systemic arteries)(11) (Figure 9B).
Of note,
enhancement of the draining vein may persist due to retrograde venous flow and should not be mistaken for reperfusion.
The pattern of draining veins is presumably related to the high flow output within the PAVM and/or to the pulmonary hypertension.
Moreover,
angiogram displays dynamic variations of the filling of draining veins according to the cardiac cycle.
In case of a high output cardiac failure with pulmonary venous hypertension,
a retrograde filling of a successfully embolized PAVM can be observed(2).
A recent study(16) suggest that a diameter of the draining vein of the embolized PAVM greater than 2.5 mm,
evaluated on the parenchymal window of unenhanced chest MDCT,
predicts reperfusion with good sensitivity and specificity and can be used to guide a decision of repeat pulmonary angiogram and embolotherapy(16).
Although,
on non-enhanced CT,
evaluation of PAVM reperfusion after embolization based on aneurysmal sac diameter reduction seems to have better diagnostic accuracy than for the PAVM feeding artery and draining vein(14).
Despite retraction of the sac is a good criterion in simple forms,
the diagnosis of reperfusion seems more difficult to advance in PAVMs with complex architecture,
particularly if there are artifacts related to the many coils necessary to occlusion(9).
Moreover reperfusion is not always related to recanalization in the center of the coils,
but sometimes to the recruitment of adjacent pulmonary arteries(9).
Besides,
there may be a wide overlap in percentage of diameter reduction (whichever structure is considered; feeding artery,
aneurysmal sac or draining vein) between occluded and reperfused PAVMs after embolization.
Any cut-off of diameter reduction may lead to an important compromise between sensitivity and specificity for the detection of PAVM reperfusion(14).
Furthermore,
although systemic supply to PAVMs can be congenital in origin,
it has also been suggested to develop as a response to surgical or percutaneous treatment(7).
When an abnormally enlarged bronchial or non-bronchial systemic artery is identified,
its connection to the PAVM should be sought.
Abnormal enlargement of one or several of the following arteries can be assessed on CHCT and may be suggestive of systemic arterial supply: right and/or left bronchial arteries,
internal mammary arteries and their terminal branches (ie,
the musculophrenic arteries),
intercostal arteries,
branches of the subclavian and axillary arteries and the inferior phrenic arteries(7).
Normal 0 false false false ES-TRAD JA X-NONE
The long-term effectiveness of embolotherapy may be classified into four categories by combining a series of criteria previously described in the literature on non-enhanced and CECT images(6,7,19): (a) success: marked retraction (more than 30%) or disappearance of the PAVM with no pulmonary or systemic perfusion; (b) partial success: reduction (less than 30%) in the size of the PAVM,
with persistent pulmonary perfusion and a patent feeding artery beyond the level of deposited coils smaller than 3 mm in diameter; (c) partial failure: substantial reuction in the size of the aneurysmal sac,
persistent pulmonary perfusion,
and a patent feeding artery (or arteries) beyond the level of the deposited coils larger than 3 mm in diameter; (d) failure: indicated by an unchanged or enlarged PAVM with persistent pulmonary perfusion.
Any case in which the development of an additional feeding artery or systemic perfusión is observed at the aneurysm level it may also considered to represent failed embolotherapy(6,7,19).
Sensitivity and specificity of contrast- enhanced CT to detect PAVM recanalization are not reported although they may be expected to be better than with nonenhanced CT because reperfusion is based on PAVM enhancement and not on diameter reduction(14).