1) Dual Phase CT of the abdomen using pancreatic protocol with image acquisition at
- 35-40 seconds after I.V.
contrast corresponding to pancreatic parenchymal phase
- 65-70 seconds after I.V.
contrast corresponding to portal venous phase
2) Endoscopic Ultrasound (EUS) using linear echoendoscope of 5-13 Mhz under mild sedation.Endoscopic Ultrasound guided FNAC/Cyst aspiration using 22G echotip needle wherever indicated.
PANCREATIC ADENOCARCINOMA
Pancreatic ductal adenocarcinomas constitute the most common primary malignant tumor,
accounting for 85%-95% of all pancreatic malignancies and is also the fourth leading cause of cancer related death [1].
On dual phase MDCT using pancreatic protocol,
Pancreatic adenocarcinoma appears
- Hypodense,
non- enhancing mass relative to uninvolved pancreatic parenchyma.
- Infiltrates the retroperitoneum
- Encases vessels
- Involves CBD and MPD.
The aim of preoperative assessment should be to:
- Localize the pancreatic adenocarcinoma
- Stage the tumor and determine if it is locally resectable
- Advise the surgeon preoperatively of relevant anatomic vascular anatomy.
Combination of axial source data with VRT and curved MPR images has been suggested as the optimal method for assessing tumor resectability.
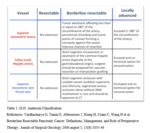
Vascular infiltration grading system (Fig 1)
M.D.
Anderson classication system (Table 1[2])
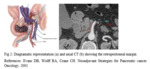
In tumors of head/uncinate process,
infiltration of retroperitoneal margin is critical in pre-operative assessment.
This corresponds to the fatty layer between right margin of proximal SMA and the left margin of head/uncinated process of pancreas (Fig 2 [3]).
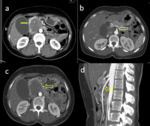
Pancreatic Adenocarcinoma- RESECTABLE (Fig 3)
Pancreatic Adenocarcinoma –BORDERLINE RESECTABLE (Fig 4[4])
Pancreatic Adenocarcinoma- UNRESECTABLE (Fig 5)
PANCREATIC ENDOCRINE TUMORS-
Pancreatic endocrine tumors (PETs) are well-differentiated pancreatic or peripancreatic tumors which demonstrate endocrine differentiation.
Pancreatic endocrine tumors are classified (based on the hormone secreted by tumor):
- Insulinomas
- Gastrinomas
- Glucagonomas
- VIPomas
- Somatostatinomas
Typically PETs (Fig 6) appear as well circumscribed hypervascular masses on cross sectional imaging,(finding attributed to their rich capillary network) and hypoechoic masses on ultrasonography.
Calcification,
cystic changes,
and necrosis are common in tumors of large size.
MASS FORMING CHRONIC PANCREATITIS-
The most important diagnostic mimicker of pancreatic adenocarcinoma is mass forming chronic pancreatitis (Fig 7,
Table 2).
PANCREATIC LYMPHOMA (Fig 8)
On CT two different patterns noted:
- Well circumscribed tumoral form
- Diffuse infiltrating type
Enhancement after I.V.
contrast is poor yet homogenous,
and presence of enlarged lymph nodes below renal veins virtually excludes the diagnosis of pancreatic adenocarcinoma.
PANCREATIC METASTASES-
Pancreatic metastases (Fig 9[5]) account for only 2% of all pancreatic malignancies.
The most common primary tumours giving rise to pancreatic metastases are:
- Lung cancer
- Breast cancer
- Renal cell carcinoma
- Malignant melanoma
- carcinoma of gastrointestinal origin
3 patterns of metastatic involvement can occur:
- Multiple nodules
- Diffuse infiltration
- Solitary mass
PANCREATOBLATOMA-
Pancreatoblastoma (Fig 10[6]) usually affects patients between the ages of 1 and 8 years.
The congenital form is associated with Beckwith-Wiedemann syndrome.
Pancreatoblastoma is a fairly well circumscribed tumor; however its origin may be difficult to determine.
It often appears to have a pancreatic or hepatic origin.
It may behave aggressively,
with evidence of local and distant spread.
The tumors are described as large,
well-defined,
multilobulated masses with enhancing septa on CT.
Pancreatoblastoma should be considered in the differential diagnosis of an upper abdominal mass in a child,
especially when the mass appears to arise from the pancreas or liver.
EUS guided FNAC may aid in reaching the histopathological diagnosis in ambiguous cases.
PANCREATIC TUBERCULOSIS-
Pancreatic tuberculosis (Fig 11) usually appears as a nonspecific focal lesion mass in HIV-seronegative patients,
whereas there is a variable spectrum of CT findings in HIV+ seropostive patients,
including:
- Focal lesion mass
- Multiple small low attenuation nodules
- Diffuse enlargement of gland
Low-attenuation peripancreatic and periportal adenopathy with peripheral rim enhancement and other ancillary findings support a diagnosis of tuberculosis.
PANCREATIC LIPOMA-
Lipomas are rare mesenchymal tumors involving the pancreas.
The characteristic features of pancreatic lipoma (Fig 12) on CT scans are:
- Homogeneity
- Low density of the lesion (ranging from -120 HU to -80 HU)
- Well defined border
- Lack of contrast enhnacement
SOLID PSEUDOPAPILLARY TUMOR (SPT)-
SPT (Fig 13) of the pancreas is a rare exocrine pancreatic tumor.
This uncommon,
typically benign tumor is found mainly in young women between the 2nd and 3rd decades of life.
CT usually demonstrates a well-encapsulated lesion with varying solid and cystic components owing to hemorrhagic degeneration.
CYSTIC LESIONS OF PANCREAS-
Cystic lesions of pancreas are being increasingly recognized due to widespread use of cross-sectional imaging.
Endoscopic ultrasound guided aspiration and biopsy aid in diagnosis of lesions that are indeterminate on cross sectional imaging.
Morphologically,
the pancreatic cysts can be classified into three major subtypes-
- Unilocular cysts
- Microcystic lesions
- Macrocystic lesions
1) Unilocular cysts
Pancreatic cysts which lack internal septa,
solid component,
or central-cyst wall calcification.
Pseudocyst is the most commonly occurring cystic lesion in this category.
Other less commonly encountered unilocular cysts include,
simple epithelial cysts (Fig 14),
unilocular variety of serous cystadenomas (Fig 15),
and lymphoepithelial cysts (Fig16).
Unilocular or macrocystic variant of serous cystadenoma is very uncommon and is seen in less than 10% of cases[7].
2) Microcystic cysts
The only cystic lesion included in this category is serous cystadenoma (Fig 17).
Majority of these benign tumors demonstrate a microcystic pattern consisting of a collection of cysts (usually more than six in number) that range from a few millimeters up to a maximum of 2cm in size [8].
A central fibrous scar with or without the characteristic stellate pattern of calcification is virtually pathognomonic for serous cystadenoma.
3) Macrocystic cysts
The cystic tumors belonging to this category include mucinous cystic neoplasms (Fig 18) and IPMNs (Fig 19).
As these lesions are considered premalignant,
surgical resection is advocated.
Mucinous cystic tumors appear as multilocular macrocystic lesions at cross-sectional imaging.
IPMNs are classified as main duct,
branch duct (side-branch),
or mixed IPMNs,
depending on the site and extent of involvement. A side-branch IPMN or a mixed IPMN (in which a side-branch tumor involves the main pancreatic duct) can have the morphologic features similar to complex pancreatic cyst,
making clear-cut distinction from a mucinous cystic neoplasm difficult.
Differntiation of serous cystadenoma and mucinous cystadenoma (Table 3)
PANCREATIC HYDATID CYST-
Pancreatic hydatid cyst (Fig 20) is a very rare entity.
It could masquerade as pseudocyst or cystic neoplasm.
Cysts in pancreatic head can present with obstructive jaundice.
Cysts show:
- Near water attenuation
- Detached membranes from pericyst ( seen as linear areas of increased attenuation)
- Daughter cysts presenting as multiloculated lesion
- Calcification in wall of cystic lesion
PANCREATIC LYMPHANGIOMA-
Pancreatic lymphangiomas (Fig 21) are multiloculated soft cystic masses,
composed of a combination of variably sized,
dilated lymphatic channels which are divided by thin septae.
They develop from lymphatic vessels which dilate progressively because of insufficient drainage,
due to atresia or inadequate efferent channels.
These tumors tend to be vary in size and are commonly seen in pancreatic body and tail.
Clinically,
these tumors may masquerade as pancreatic neoplasms.
On CT,
the tumor is:
- Well-defined
- Water attenuation
- Polycystic tumor with thin septae
Septae are better visualized on ultrasound.
The histopathological diagnosis of pancreatic lymphangioma has traditionally been established after surgery.
However,
Endoscopic Ultrasound-Guided cyst aspiration is recommended to establish a definite diagnosis pre-operatively.