Cross sectional imaging is rarely utilized in diagnostic purposes,
but there are certain features that can be used to differentiate benign from malignant disease.
I. Benign lesions
Cystic lesions may have an intrinsic origin,
when they arise from the sinus mucosa,
or an extrinsic origin,
mostly odontogenic cysts of the maxillary sinus.
Retention cysts - result of obstruction of small mucosal serous or mucinous glands.
They are usually small,
with a well-defined outline and are seen in approximately 10% of the population,
and do not cause any symptoms,
but may occasionally fill the sinus.
CT appearance is of a smooth,
broad-based,
clearly defined soft-tissue mass that does not cause bone erosion.
On MRI these are usually of low signal on T1WI,
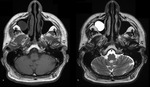
and high signal on T2WI (Fig.
1).
Mucoceles - end-stage of a chronically obstructed,
airless,
mucus-filled sinus,
most commonly affecting the frontal sinus (66%).
Sphenoidal localization is rare.
Protein content causes varying signal intensities on T1WI and T2WI. Increasing chronicity causes signal to increase on T1WI and decrease on T2WI.
Postcontrast scans show peripheral enhancement of the mucosa with no enhancement of the content.
Benign tumors may also be classified by intrinsic and extrinsic origin.
Intrinsic tumors are either epithelial (polyps,
inverted papilloma) or mesenchymal (osteoma,
angiofibroma).
Extrinsic origin may be odontogenic,
neurogenic or orbital.
Sinonasal polyposis - soft-tissue pedunculated masses of oedematous hyperplastic upper respiratory mucosa.
The most frequent location is the ethmoids,
followed by the maxillary antra and then the sphenoid sinus.
On CT scans they appear as hypodense,
but may be hyperdense due increased protein content or fungal infection (Fig 3).
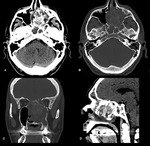
Antrochoanal polyp - unilateral polyp,
causing unilateral nasal obstruction,
that arises from the maxillary antrum,
through the ostium which it enlarges (radiological hallmark),
and then projects into the postnasal space (Fig.
2).
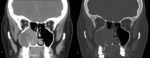
Inverted papilloma – benign lobulated epithelial tumor of the sinus mucosa,
associated with squamous cell carcinoma (10-20%).
Cerebriform pattern of enhancement is typical.
The tumor tends to remodel the bone.
Treatment is by surgical resection and recurrences occur in approximately 15% of cases (Fig.
3).
Osteomas - benign,
slowly growing tumors containing mature compact or cancellous bone,
most frequently in the frontal sinus,
usually asymptomatic.
Plain film and CT findings show a well-defined dense lesion if it consists of compact mature bone,
but a cancellous bone content appears as less ossified.
Ameloblastomas – benign but locally aggressive odontogenic tumors that usually arise from the mandible and rarely from the maxilla,
in which case they extend to the maxillary sinus.
Honeycomb,
soap bubble appearance,
or well-marked cystic areas with no matrix calcification are diagnostic (Fig.
4).
II. Malignant lesions
Paranasal sinuses neoplasias are histologically classified as epithelial or mesenchymal.
The epithelial tumors arise from the epithelial lining of the nasal and sinus cavities,
the accessory salivary tissue,
the neuroendocrine tissue or the olfactory mucosa,
whereas mesenchymal tumors arise from supporting tissues.
Carcinomas - represent 3–4% of head and neck tumors and less than 1% of all malignancies,
the most frequent type being the squamous cell carcinoma (80% of all sinus malignancies),
followed by adenocarcinomas (10%,
which includes adenoid cystic carcinomas of minor salivary gland origin,
mucoepidermoid carcinomas,
ascinic carcinomas and pleomorphic adenomas- Fig.
9-11),
most often originate from the maxillary sinuses followed by the ethmoid sinuses,
nasal vestibule and cavity.
On MRI,
they are of intermediate T1 signal intensity and slightly higher signal intensity on T2WI (Fig.10).
Large tumors may present areas of necrosis and haemorrhage altering the signal intensities within them.
Olfactory neuroblastoma - phenotype of neuroendocrine tumor arising from the olfactory epithelium.
The age distribution is bimodal: 11-20 years and 51-60 years.
Epistaxis and nasal obstruction are the most common symptoms.
These lesions are also hypointense to brain on T1WI and hypertense to brain on T2WI.
Malignant melanomas- nose location is more common and has superior prognostic,
high signal intensities may be identified on T1WI due to the presence of melanin or bleeding.
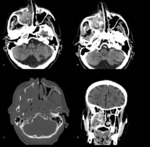
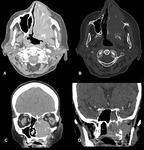
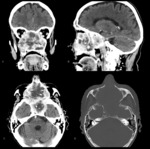
Chondrosarcoma - most common sarcoma affecting the paranasal sinuses.
At CT,
the tumor is seen as a lobulated,
heterogeneous lesion with a chondroid matrix and scattered calcifications (Fig.
5).
The chondroid matrix has high water content and therefore presents with high signal on T2WI and low signal on T1WI; calcifications of the chondroid matrix cause signal voids at all sequences.
Also the septum shows low signal at both T1WI and T2WI.
Both CT and MR imaging demonstrate a heterogeneous enhancement pattern due to septal enhancement,
but the avascular chondroid matrix is unchanged.
The most typical feature is however the extremely high signal intensity on T2WI that is much higher than the signal of cerebrospinal fluid.
Rhabomyosarcoma 5-10% of all malignant solid tumors in children <15 years of age.
MRI is imaging modality of choice.
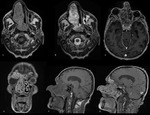
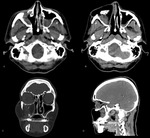
Sinonasal lymphomas- Non-Hodgkin's lymphoma may primarily occur in the nasal and paranasal regions,
with or without associated cervical or systemic nodal disease.
They are often seen as bulky masses causing expansion,
erosion or infiltration that tend to have an intermediate signal intensity on both T1- and T2WI,
however at both CT and MR imaging,
the contrast enhancement of lymphomas can be variable and not discriminated from other malignancies.
Lethal midline granuloma is a prelymphomatous state (Fig.
6).
The key role of imaging is essential in the preterapeutical staging of patients with paranasal sinuses tumors in describing the tumor extension,
nodal involvement and metastases.
Radiological results,
as an essensial basis for treatment planning,
should thoroughly describe tumoral spread in all planes and structures involved or destroyed.
To provide adequate information necessary for resectability and curability criteria,
radiologists must have detailed knowledge not only about the anatomy of this region,
but also about all the possible tumoral extension areas that will influence the surgical or irradiation treatment plan.
III. Tumors of the maxillary sinus
Anatomically,
the maxillary sinus is a pyramid-shaped cavity within the maxillary bone.
The medial border is the lateral nasal wall.
Superiorly,
the sinus adjoins the orbital floor and includes the infraorbital canal (containing the infraorbital nerve and artery).
The posterolateral wall is anterior to the infratemporal fossa (housing brances of the maxillary artery and of the mandibular nerve) and pterygopalatine fossa (containing the pterygopalatine ganglion,
the distal part of the maxillary artery and the maxillary nerve).
The anterior wall is posterior to the facial skin and soft tissue.
The inferior wall of the maxillary antrum extends below the nasal cavity floor and is in close proximity to the hard palate and maxillary tooth roots.
The maxillary is divided,
by lines drawn on a coronal view of the sinuses through the alveolar process of the maxilla and the orbital floor,
into an infrastructure,
mesostructure and suprastructure.
Tumors limited to the mesostructure and infrastructure are usually treated with a maxillectomy (partial or total),
but tumours involving the suprastructure require total maxillectomy and even orbital exenteration in some cases.
Critical extention areas include the floor of the anterior and middle cranial fossae,
the pterygopalatine fossae,
the orbits,
and the palate.
Curative surgery is not usually possible if there is central skull base obstruction,
tumor extension into the pterygopalatine fossa or the nasopharynx.
It is thus important to include coronal views in the staging examination.
Axial views are also necessary to determine posterior and anterior extension of disease.
Tumor limited to mucosa with no erosion or destruction of bone are considered to be T1 using the TNM classification system,
whereas bone erosion or destruction including extension into the hard palate or middle nasal meatus is T2.
Tumors located in the maxillary sinus may extend to the midline through the maxillary ostium,
into the middle nasal meatus,
involving the nasal septum (T2 in the TNM classification),
and even the etmoidal cells (TNM T3).
Cribriform plate involvement is considered T4a,
it is best determined by contrast enhanced MRI scans T1WI in coronal and sagittal views,
whereas the enhancement of dural envelope confirms intracranial invasion (T4b).
If the tumour involves the fovea ethmoidalis or cribriform plate,
then a craniofacial resection is required
Inferior extension of tumors may reach the alveolar ridge and the hard palate (T2 TNM).
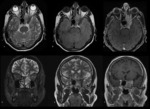
Superior extension may comprise the floor or medial wall of the orbit (T3),
the anterior orbital contents (T4a),
the orbital apex,
the dura up to cerebral involvement (T4b-Fig.
9).
It is important to assess whether the periorbital fat is involved,
since orbital exenteration is necessary in such cases.
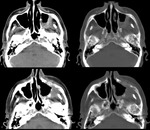
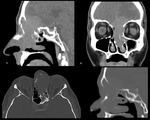
Anterior extension occurs when a tumor erodes the anterior wall of the maxilar sinus (involving the infraorbital foramen) up to the subcutaneous tissues (T3 TNM)-Fig.
8,
and even up to the skin of the genian region (T4a TNM) - Fig.7.
Posterolateral extension to the infratemporal fossa is classified as T4a while posterior extension to the pterygoid fossa (via the pterygoid fissure) T3 and the erosion of the pterygoid plate also T4a -Fig.
6.
In TNM T1 and T2 staged tumors,
partial maxillectomy is recommended.
In T3,
a total maxillectomy is required.
TNM stage T4a is considered to be a moderately advanced local disease,
when reaching the regions mentioned above,
the frontal or the sphenoidal sinus.
In T4a,
a radical maxillectomy is required including the removal of the entire maxilla and ethmoid sinus.
Craniofacial resection may be required if the roof of the ethmoid and of the orbit is involved.
TNM stage T4b represents a very advanced local disease,
with cerebral involvement,
central skull base or cranial nerves (other than the maxillary division of the infraorbital region) and contraindicates surgery.
Orbital exenteration is indicated when a tumor has spread through the periorbita and invaded the orbital fat.
If the tumor does not pass through the orbital periosteum involving the orbital contents,
then exenteration is not necessary (Fig.
7).
IV. Ethmoidal tumors
Most ethmoid sinus tumors present with advanced disease.
The diagnostic delay is caused by the low incidence of these tumors,
and due to the fact that early symptoms and signs mimic those of inflammatory disease,
or usually result from secondary obstructive sinusitis.
Anatomically,
the ethmoid sinuses are thin-walled air cells from the ethmoid bone,
located between the medial walls of the orbits by the lamina papyracea,
and the lateral wall of the nasal cavity.
The ethmoid bone consists of two lateral masses,
a sagittal midline plate and a horizontal plate,
called the cribriform plate perforated by microscopic foramina for the passage of the olfactory nerve fibers.
The labyrinths consist of three groups of ethmoidal air cells - anterior,
middle and posterior - ranging from 3 to 18 cells.
The anterior and middle air cells drainage into the middle meatus of the nasal cavity,
the posterior cells empty into the superior nasal meatus.
Special ethmoid sinus cells include the Agger nasi cells,
which are anterior-inferior air cells,
and the infraorbital Haller's cells,
along the medial foor of the orbit.
Ethmoidal tumors restricted to one ethmoidal cell are classified using the TNM staging system as T1.
Invasion within the nasoethmoidal complex is considered a T2 stage.
If a tumor extends to the medial wall or floor of the orbit,
maxillary sinus,
palate or cribriform plate it is classified as T3.
T4a classified tumors invade the anterior orbital contents,
the skin of the nose or genian region,
minimal extension to anterior cranial fossa,
pterygoid plates,
sphenoid or frontal sinuses (Fig.
9),
whereas T4b tumors orbital apex,
dura,
brain (Fig.
8),
middle cranial fossa,
cranial nerves,
nasopharynx or clivus.
The preferred treatment for ethmoidal tumors is usually surgery followed by postoperative radiotherapy.
Surgically,
an ethmoidectomy is performed,
which includes the removal of the entire lateral ethmoidal wall,
the labyrinth,
lamina papyracea,
and middle and inferior turbinates.
Endoscopic procedures are sometimes considered for T1 and T2 classified tumors.
If the tumor involves the fovea ethmoidalis or cribriform plate (T3),
craniofacial resection is required,
that may include the cavernous sinus or the sphenoid sinus,
but the outcome is poor. If the patient is not a surgical candidate (T4b),
then radiotherapy alone is recommended.