THE MIDBRAIN
Anatomy
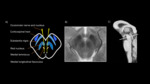
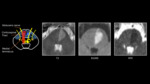
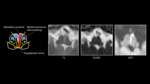
Figure 1 illustrates the anatomical structure of the midbrain on 3T T2 weighted MR imaging and through simplified pictorial representation. The corticospinal tracts,
descending white matter pathways involved in voluntary discrete movements,
lie anteriolaterally in the upper midbrain.
Within the more caudal midbrain the corticospinal tracts move medially along the ventral surface of the cerebral peduncles. The red nuclei and medial lemniscus are located medially,
whilst the medial longitudinal fasciculus lies within the paramedian midbrain[5].
The red nuclei receive afferent fibres from the motor cortex and dentate nucleus of the cerebellum and send axons to the spinal cord,
coordinating muscle tone,
body position and gait. The medial lemnisci are ascending sensory pathways,
in continuity with the dorsal columns of the spinal cord. They are responsible for touch,
vibration sense and proprioception. The medial longitudinal fasciculi connect the oculomotor,
trochlear and abducens nuclei,
thereby integrating eye movements[5].
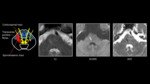
Medial midbrain infarction
Claude syndrome comprises ipsilateral oculomotor nerve palsy and contralateral hemiataxia[6],
due to occlusion of the perforating branches of the posterior cerebral artery,
usually due to underlying small vessel disease[7]. Historically,
the syndrome has been attributed to infarction of the oculomotor nucleus/fascicles and the red nucleus,
which contains dentato-thalamic fibres.
More recently there is increasing evidence that the dentato-thalamic fibres also meet at the superior cerebellar peduncle,
medial and below the level of the caudal end of the red nucleus,
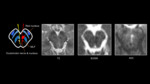
and that infarction in this region is responsible for the hemiataxia component of Claude syndrome[7-9]. Figure 2 demonstrates T2 and diffusion weighted imaging findings in a patient presenting with left sided oculomotor palsy and right sided hemiataxia.
If the infarction affects the ventral midbrain,
but not the red nucleus,
this can result in Weber syndrome with involvement of the cerebral peduncle containing the corticospinal and corticobulbar tracts and third nerve fascicle.
This results in contralateral hemiparesis and ipsilateral oculomotor nerve palsy,
without hemiataxia,
and is caused by vascular compromise of the para-median branches of the basilar artery or posterior cerebral artery[10,11].
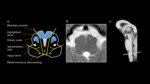
Lateral midbrain infarction
Infarction of the oculomotor nucleus and red nucleus/superior cerebellar peduncle (causing oculomotor nerve palsy and incoordination/cerebellar ataxia,
as in Claude syndrome) in combination with corticospinal tract involvement (resulting in contralateral hemiparesis) results in Benedikt syndrome[12],
with a more lateral appearance than Claude syndrome.
This is most commonly secondary to occlusion of branches of the posterior cerebral artery[12].
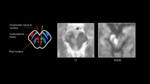
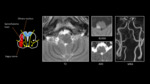
Figure 3 demonstrates a case of Benedikt syndrome,
in a patient who presented with right oculomotor nerve palsy and weakness of the left upper and lower limbs.
Involvement of the red nucleus can distinguish the lateral infarct in Benedikt syndrome from the medial infarct in Weber syndrome[13].
THE PONS
Anatomy
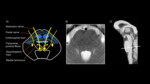
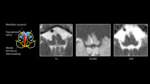
Figure 4 illustrates the anatomical structure of the pons on T2 weighted MR imaging and through simplified pictorial representation. Within the ventral pons,
the corticospinal tracts now lie medially. Transverse pontine fibres,
which lie over and lateral to the corticospinal tracts,
arise from ventral pontine nuclei which in turn receive fibres from the cerebral cortex. The fibres decussate and pass into the contralateral cerebellar hemisphere via the middle cerebellar peduncle. The spinothalamic tracts,
which carry information about crude touch,
pain and temperature from the skin to the postcentral gyrus via the thalamus,
lie laterally within the pons[5].
Foville Syndrome (medial pons)
Foville syndrome is characterised by contralateral hemiplegia,
loss of conjugate gaze and ipsilateral lower motor neurone facial weakness secondary to infarction within the medial portion of the caudal third of the pons,
disrupting the corticospinal tracts,
abducens nerve roots/paramedian pontine reticular formation and medial lemniscus respectively[14.15].
It is the result of occlusion of the para-median or short circumferential branches of the basilar artery[14]. Figure 5 demonstrates an infarct on T2 and diffusion weighted imaging in a patient presenting acutely with left arm and leg weakness,
inability to coordinate eye movements and right facial droop,
in keeping with Foville syndrome.
Marie Foix Syndrome (lateral pons)
Infarction within the lateral pons (sometime extending into the middle cerebellar peduncle) presents with contralateral hemiparesis,
ipsilateral ataxia and contralateral hemi-sensory loss to pain and temperature,
due to involvement of the corticospinal,
cerebellar (transverse pontine) and spinothalamic tracts respectively[16].
Vascular compromise of the anterior inferior cerebellar artery and branches of long circumferential branches of the basilar artery are responsible[16]. Figure 6 demonstrates the anatomical structures involved and T2 and diffusion weighted imaging findings in a case of Marie Foix syndrome,
in which there was sparing of the corticospinal tracts. The patient presented with incoordination of the right side and loss of pain and temperature sensation on the left.
THE MEDULLA
Anatomy
Figure 7 illustrates the anatomical structure of the medulla on T2 weighted MR imaging and through simplified pictorial representation. The majority of the fibres of the corticospinal tracts decussate within the pyramids on the ventral surface of the medulla. The olivary nuclei,
which are involved in the control of movement through connections with the cerebellum via the inferior cerebellar peduncle,
lie lateral to the pyramids[5].
Dejerine Syndrome (medial medulla)
Infarction of the medial medulla results in a triad of contralateral hemiparesis,
hemi-sensory loss and ipsilateral tongue weakness,
due to involvement of the medullary pyramid,
medial lemniscus and hypoglossal nerve nucleus respectively[17,18].
Vascular compromise of the vertebral,
anterior spinal or basilar artery may be responsible[18].
Figures 8 and 9 demonstrate the anatomical structures involved and T2 and diffusion weighted imaging findings in two cases of Dejerine syndrome.
Figure 8 demonstrates an infarct in a patient presenting with predominantly sensory impairment and weakness of the right side of the tongue.
Figure 9 demonstrates more extensive involvement of the pyramid,
which correlated with a clinical history of acute onset of left arm and leg weakness in combination with sensory impairment and weakness of the left side of the tongue.
Wallenberg Syndrome (lateral medulla)
Infarction within the lateral medulla results in Wallenberg syndrome[4], comprised of crossed hemi-sensory disturbance (ipsilateral face,
contralateral body),
ipsilateral Horner syndrome and ipsilateral cerebellar signs,
due to infarction of the spinothalamic tract,
vagus nerve and olivary nucleus respectively. An array of additional symptoms and signs may accompany this classic triad,
due to varying degrees of collateral flow and perfusion abnormalities within the surrounding brainstem.
Compromise of the posterior inferior cerebellar artery results in Wallenberg syndrome,
which can occur,
most notably,
in the context of vertebral artery dissection in younger patients or in the context of trauma,
but also secondary to atherosclerosis or embolic disease.
Those structures supplied by the anterior spinal artery are characteristically spared,
notably the corticospinal tracts,
hypoglossal nuclei and dorsal column medial lemniscus pathways.
Neurovascular imaging is particularly important in those patients presenting with Wallenberg syndrome,
for identification of vertebral artery dissection.
In contrast to the generally poor outcome from brainstem infarction syndromes,
with prompt diagnosis and optimal management,
Wallenberg syndrome has a favourable prognosis.
In addition,
Wallenberg syndrome accounts for at least 2.5% of all ischaemic strokes and it’s diagnosis has particular clinical significance due to a relatively high correlation with vertebral artery dissection.
Figure 10 demonstrates the anatomical structures involved and T2,
diffusion weighted imaging and MRA findings in a case of Wallenberg syndrome. The patient presented with mainly left sided,
but right facial,
sensory disturbance (spinothalamic tract involvement) and right sided cerebellar signs (olivary nucleus).