This pictorial essay reviews the radiological MRI findings in patients scanned at our centre,
demonstrating involvement of the corpus callosum of fetuses,
children and adults.
The wide spectrum of lesions in the corpus callosum,
both congenital and acquired,
were illustrated.
1.
Developmental Abnormalities
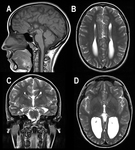
The most extreme form of developmental malformation of the corpus callosum is agenesis.
Agenesis of the corpus callosum can be complete (agenesis) (Fig. 1, Fig. 2) or partial (dysgenesis) (Fig. 5) [4].
It is an anatomically defined condition that result from disruption of the early stages of fetal callosal development.
The diagnosis is based on a finding of absent callosal fibers as visualized through neuroimaging.
Agenesis of the corpus callosum is typically accompanied by a characteristic dilatation of posterior lateral ventricles (colpocephaly) and often the presence of atypical fiber bundles (Probst Bundles) that run anterior to posterior just lateral to the interhemispheric fissure.
[1] Suspected defects of the corpus callosum should be confirmed by MRI because in 80% of cases they coexist with other CNS pathologies [3].
Radiological terminology to describe corpus callosum abnormalities used in literature is rather confusing and heterogeneous.
[4]
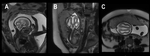
Hypoplasia consists of uniformly thin or partially underdeveloped corpus callosum in the posterior region.
There are 3 subtypes of hypoplasia: hypoplasia without dysplasia (generalized hypoplasia but intact morphology) (Fig. 3),
apple core corpus callosum abnormality (hypoplasia of posterior corpus callosum) (Fig. 4),
and anterior remnant of corpus callosum (agenesis of the mid and posterior corpus callosum with an anterior remnant),
based upon the physical appearance.
[5]
Dysplasia without hypoplasia encompasses cases in which corpus callosum is morphologically abnormal but has no evidence of hypoplasia,
and is represented by a case with hump-shaped corpus callosum.
In hypoplasia with dysplasia of corpus callosum,
there are 2 subtypes,
stripe corpus callosum (uniformly thinned corpus callosum,
with dysplasia) and kinked corpus callosum (hypoplasia and kinked).
[5]
2.
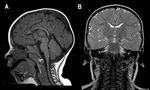
Pericallosal lipoma (corpus callosal lipoma) (Fig. 5)
Intracranial lipomas are rare callosal or pericallosal lesions associated with persistence and maldifferentiation of the meninx primitiva,
which is the embryonic precursor of the meninges.
The morphology may be tubulonodular or curvilinear.
Tubulonodular lipomas have a round or cylindrical shape,
are larger in diameter,
and are usually located in the anterior brain.
There is a high association with frontal lobe anomalies,
encephaloceles,
and callosal dysgenesis.
Curvilinear lipomas are thin lesions that curve posteriorly around the splenium.
Associated callosal malformations are rarer and less severe [6].
On MRI,
lesions are homogeneously T1 hyperintense and T2 hyperintense,
following the signal of subcutaneous fat,
and without contrast enhancement.
Loss of signal on fat-suppressed or STIR images confirms the presence of internal fat.
3.
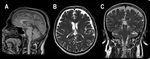
Phakomatoses (Fig. 6)
Phakomatoses belong to congenital diseases in which callosal abnormalities are observed.
Neurofibromatosis type 1 or von Recklinghausen disease is the most frequent of them,
with the estimated incidence of 1:3000.
Neurofibromatosis bright objects (UNO),
called formerly unidentified bright objects (UBO),
are T2 hyperintense and appear most often in the basal ganglia,
brainstem,
and posterior fossa.
They are also found in the corpus callosum,
mainly in the splenium.
UNO are rare before the age of 4 years; they increase in number and volume till the age of 10–12 years and tend to resolve thereafter,
so that after the age of 20 they are almost never seen.
Usually they do not undergo malignant transformation but they can,
so follow-up MRI studies are very important in NF1 patients [7].
Besides it has been shown that NF1 children have a significantly larger corpus callosum while their IQ is significantly lower than in control subjects [8].
4.
Dysmyelinating disorders - Leukodystrophy (Fig. 7 ,
Fig. 8)
Dysmyelinating disorders are a subset of white matter disorders characterised by abnormal myelination [9].
They are also known by the term leukodystrophy and are composed of a group of inherited conditions that are characterized by a defective structure and function of the myelin sheath [10].
They typically,
although not invariably,
affect children,
and may affect the corpus callosum.
Metachromatic leukodystrophy is the most common form and is caused by arylsulfatase A deficiency.
Other hereditary leukoencephalopathies include X-Linked Adrenoleukodystrophy,
Metachromatic leukodystrophy,
Alexander disease,
Krabbe disease,
Sudanophilic leukodystrophy and others.
MR imaging is highly sensitive in determining the presence and assessing the severity of underlying white matter abnormalities in patients with leukodystrophy.
Although the findings are often nonspecific,
systematic analysis of the finer details of disease involvement may permit a narrower differential diagnosis,
which the clinician can then further refine with knowledge of patient history,
clinical testing,
and metabolic analysis.
MR imaging has also been extensively used to monitor the natural progression of various white matter disorders and the response to therapy.
[11]
5.
Demyelinating diseases
5.1.
Multiple sclerosis (MS) (Fig. 9 ,
Fig. 10)
Multiple sclerosis (MS) is a relatively common acquired chronic relapsing demyelinating disease involving the central nervous system,
and is the second most common cause of neurological impairment in young adults,
after trauma [12]
Callosal involvement is typical of MS although it has never been included in the evolving diagnostic criteria of this disease [13].
On MRI,
the disease is T2 and FLAIR hyperintense,
with contrast enhancement in the acute stage.
The presence of subcallosal striations or callosal-septal interface lesions,
which are thin bands along the undersurface of the corpus callosum,
is highly sensitive and specific (Fig.
6A).
Dawson fingers are ovoid lesions that radiate perpendicularly from the lateral ventricles in a pattern thought to reflect perivenular demyelination (Fig.
6B).
In the chronic phase,
T1-hypointense lesions (dark spots or black holes) reflect increased water content secondary to extreme demyelination and axonal loss.
Susceptibility artifact on gradient recalled-echo/susceptibility-weighted imaging sequences indicates macrophage and iron deposition in areas of permanent tissue destruction [14,
15]
5.2 Marchiafava-Bignami Disease (Fig. 11)
Marchiafava-Bignami disease refers to a disorder resulting in demyelination of the corpus callosum.
It was first described by two Italian pathologists who identified it in the autopsies of three patients who presented in status epilepticus and subsequently developed coma [16]
On MR images,
patients with Marchiafava-Bignami Disease show areas of low T1 signal intensity and high T2 and FLAIR signal intensity in the body of the corpus callosum at times extending into the genu and the adjacent white matter.
These lesions do not have mass effect and may show peripheral contrast enhancement during the acute phase.
Eventually,
the lesions cavitate and become well marginated.
[17- 20]
The lesions are difficult to visualize on CT scans,
where they appear as hypoattenuated areas.
[21]
Other lesions involving the corpus callosum that may have a similar appearance include infarctions,
shearing injuries,
and demyelination process.
[18,
22]
6.
Vascular lesions
6.1.
Infarcts (Fig. 12 ,
Fig. 13)
Because of a rich collateral blood supply,
corpus callosal infarcts are rare and associated with systemic vasculitides,
shower emboli,
major ischemic stroke,
or subfalcine herniation with mass effect.
The splenium is most commonly affected,
followed by the body and genu.
[23]
CT lacks the necessary contrast resolution to identify mild edema associated with callosal infarcts.
On MRI,
reduced diffusivity on diffusion-weighted imaging is the earliest sign,
followed by edema with T2 hyperintensity and T1 hypointensity.
Contrast enhancement is variable but more likely in the acute phase.
In the subacute to chronic stages,
edema evolves into gliosis or atrophy,
with corresponding normalization of diffusivity.
Hemorrhagic transformation may manifest as magnetic susceptibility and chronic hemosiderin on gradient recalled-echo and susceptibility-weighted imaging sequences.
6.2.
Cavernous haemangioma (Fig. 14)
Cerebral cavernous venous malformations,
commonly known as cavernous haemangioma or cavernoma,
are common cerebral vascular malformations,
usually with characteristic appearances on MRI.
It is a “mulberry-like” vascular malformation that is defined in histologic terms by blood cavities surrounded by a single layer of endothelium without muscular tissue or intervening brain parenchyma.
This lesion may occur throughout the central nervous system but is more frequently demonstrated in the cerebral hemispheres.
Callosal localization is less common.
[24]
MRI is the modality of choice,
demonstrating a characteristic “popcorn” or "berry" appearance with a rim of signal loss due to hemosiderin,
which demonstrates prominent blooming on susceptibility weighted sequences.
T1 and T2 signal is varied internally depending on the age of the blood produces and small fluid-fluid levels may be evident.
Gradient echo or T2* sequences are able to delineate these lesions better than T1 or T2 weighted images.
In patients with familial or multiple cavernous angiomas GRE T2* sequences are very important in identifying the number of lesions missed by conventional Spin echo sequences.
Susceptibility weighted imaging (SWI) may have sensitivity equal to that of GRE in detecting these capillary telangiectasias in the brain.
SWI is also highly sensitive in detecting calcification as compared to T1 and T2 images.
[25]
If a recent bleed has occurred,
then surrounding oedema may be present.
The lesions generally do not enhance,
although enhancement is possible.
[26].
6.3.
Dilated perivascular spaces (Virchow-Robin spaces) (Fig. 15 ,
Fig. 16)
Dilated perivascular spaces can occur throughout the cerebral white matter,
including the corpus callosum.
They appear as well-circumscribed ovoid lesions isointense to CSF on all sequences.
These lesions have no known clinical significance but increase in size and frequency with age.
There is an association with mucopolysaccharidosis,
in which accumulation of glycosoaminoglycans dilates the Virchow-Robin spaces and produces a cribriform (état criblé) appearance in the white matter,
corpus callosum,
and basal ganglia.
[23]
7. Neoplastic Lesions
7.1.
Primary neoplastic lesions
7.1.1.
Glioma
Glioblastoma multiforme (WHO grade IV astrocytoma) (Fig. 17) is the most common primary brain malignancy,
accounting for 12%–15% of all intracranial neoplasms.
The prognosis is uniformly grim.
Glioblastoma occurs most frequently in the cerebral hemisphere of adults between 45 and 70 years of age.
It is rare in the cerebellum and spinal cord,
and fewer than 10% of cases are found in children,
in whom the brainstem is affected more commonly than in adults [27].
Callosal GBM,
in addition to the corpus callosum,
affects also both cerebral hemispheres resulting in the typical “butterfly glioma”.
[28].
Conventional gadolinium-enhanced MR imaging is the standard technique for the evaluation of glioblastoma and typically demonstrates a large,
heterogeneous mass in the cerebral hemisphere exhibiting necrosis,
hemorrhage,
and enhancement.
MR spectroscopy and perfusion MR imaging have been shown to be useful in prospective determination of tumor grade.
At spectroscopy,
elevation of choline and depression of NAA suggest tumor; metabolite ratios (choline-creatine,
choline-NAA,
NAA-creatine,
myoinositol-creatine) exhibit relationships to tumor grade.
At perfusion MR imaging,
relative cerebral blood volume is increased in higher-grade astrocytomas [29,
30]
Gliomatosis cerebri has been deleted from the 2016 CNS WHO classification as a distinct entity,
rather being considered a growth pattern found in many gliomas,
including IDH-mutant astrocytic and oligodendroglial tumors as well as IDH-wildtype glioblastomas.
[31,
32] (Fig. 18)
Thus,
widespread brain invasion involving three or more cerebral lobes,
frequent bilateral growth and regular extension to infratentorial structures is now mentioned as a special pattern of spread within the discussion of several diffuse glioma subtypes.
[33]
The tumour may be primary (de novo) or secondary,
with the latter as a result from the spreading of a more focal glioma [34].
Gliomatosis cerebri can contain areas of WHO grade II or III tumours and rarely grade IV.
[35,
36]
7.1.2.
Lymphoma (Fig. 19)
Primary CNS lymphoma accounts for approximately 16% of primary brain tumors.
They preferentially affects immunocompromised patients and can involve or extend through the corpus callosum.
Most of them are non-Hodgkin’s and represent B-cell type.
The hypercellular histology manifests with increased density on unenhanced CT.
Lesions are multifocal and nodular,
tending to show less mass effect and peritumoral edema than expected for the size.
They are most often isointense-hypointense on T1-weighted images,
hypointense on T2-weighted images.
Most lesions enhance in a solid or ring fashion,
although nonenhancing lymphoma has been reported.
DWI often shows restricted diffusion.
In patients with immunodeficiency lymphoma is more often multifocal,
irregular,
and heterogeneous in terms of signal intensity and ring enhancing.
Lymphoma is highly radiosensitive and regresses rapidly with steroid treatment (so-called “vanishing lesions”) [37].
7.2.
Metastases (Fig. 20 ,
Fig. 21)
Corpus callosum may also be affected by metastases although it is reported to be rare.
Callosal involvement is more frequent in case of infiltration by a lesion from the adjacent structures [38].
8.
Trauma - traumatic brain injury (Fig. 22 ,
Fig. 23)
Traumatic brain injury is a primary cause of neurologic deficits in patients with severe head trauma,
most commonly high-speed motor vehicle accidents.
The corpus callosum,
particularly the posterior body and splenium,
are preferentially involved because of their fixation to the overlying dura,
with resulting torque injury.
CT is not sensitive for TBI,
but shows hyperdense petechial foci of hemorrhage in 20% of cases.
MRI is a much more sensitive technique,
with microhemorrhage detected as foci of magnetic susceptibility on T2*-weighted gradient-recalled echo or susceptibility-weighted imaging sequences (SWI).
Nonhemorrhagic lesions show reduced diffusivity on diffusion-weighted imaging,
possibly with surrounding edema.
Chronic lesions are associated with hemosiderin and encephalomalacia.
Diffusion-tensor imaging with tractography also holds promise for detailed assessment of axonal injury.
[23]
9.
Iatrogenic Injuries (Fig. 24 ,
Fig. 25)
Corpus callosum may be injured as a result of shunting procedures in patients with hydrocephalus [6] as well as a result of various neurosurgical interventions.