1) Common imaging findings
1.1) Ultrasound:
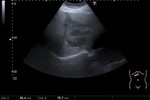
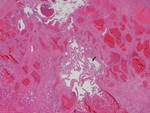
The most common sonographic pattern of hemangiomas (Fig.
2) consists of homogeneous hyperechoic liver nodule,
with discrete posterior acoustic enhancement,
devoided of Doppler signal both on colour-coded and spectral examination.
Multiple interfaces between the walls of the sinuses and the blood within them account for the typical hyperechogenicity seen at US.
Because hemangiomas may undergo various changes such as internal hemorrhage with necrosis,
thrombosis,
myxomatous change,
fibrosis,
and rarely calcification as they become larger1,4,
atypical US features are frequently seen in larger ones.
1.2) CT:
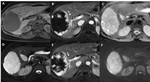
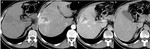
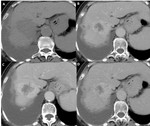
CT (Fig.3) usually shows a hypodense well-defined lesion,
with an internal density similar to vessels,
that is a characteristic feature of blood pool lesions.
Dynamic studies performed after the administration of non-specific extracellular iodine compounds exhibits early nodular peripheral enhancement (puddling),
since its feeding vessels originate from the hepatic artery.
Subsequent phases of liver enhancement reveal a progressive slow centripetal fill-in,
with iso or even slight hyperdensity on delayed phase as compared to the normal liver parenchyma.
This classical presentation is diagnostic with a high level of confidence.
Late intra-tumoural accumulation of contrast can be explained by the slow flowing blood within its vascular channels as well as absence of noticeable washout on the latter phases of the dynamic study determine iso/hyperdensity.
However,
delayed homogeneity due to complete contrast filling of the tumor should not be regularly expected,
especially for large tumors,
and,
thus,
should not be used as mandatory diagnostic criteria.
Small hemangiomas may appear to enhance to a lesser extent but this may result from partial volume averaging.
1.3) MRI:
Magnetic resonance imaging is the imaging method of choice to diagnose hemangiomas (Fig.4)5,6.
On T1-weighted (-w) images,
hemangiomas display low signal intensity and on T2-w,
they are strikingly homogeneous,
clearly demarcated from the adjacent parenchyma,
with very high signal intensity,
similar to cerebrospinal fluid (CSF),
due to the long T2 relaxation time of its blood-filled vascular channels,
a feature that has been coined as the bulb-light sign.
On diffusion MR images hemangiomas show restricted diffusion because of the long T2-relaxation time rather than the limited mobility of the water protons (T2 shine-through).
So we will find areas demonstrating substantial restriction on diffusion images and high diffusivity on the ADC map.
The enhancement in dynamic study is equivalent to that seen at CT,
with complete filling of the lesion.
2) Uncommon presentations of hemangiomas:
Although the prevalence of atypical hemangioma has not been precisely determined,
it seems that approximately 20-40% of hemangiomas are of this kind.
Less typical imaging findings can result from one of three main causes: altered morphology or structure,
unusual flow pattern or associated liver abnormalities. Although these aspects do not preclude in all instances the correct diagnosis,
they may be misleading and,
therefore,
should be known by radiologists in order to avoid other potentially more invasive diagnostic tests.
Whenever the diagnosis is doubtful,
especially in oncologic patients,
even after a multimodality imaging approach,
a percutaneous biopsy could be sought,
with considerable evidence concerning its safety.
2.1) Uncommon morphology/structure
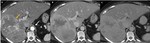
2.1.1.) Hemangioma with Echoic Border or with Central Scar:
On grey-scale US,
atypical hemangiomas can show an internal iso/hypoechoic texture with a peripheral hyperechoic border (Fig.5),
which has been associated with internal thrombosis and scarring.
Large hemangiomas are frequently heterogeneous and may display a central scar (Fig.5),
which does not enhance on the late interstitial phases of the dynamic studies.
On pathology,
the scar is formed by myxomatous degeneration,
thrombosis,
fibrosis or necrosis.
The T2-w images are frequently unable to determine its exact composition,
as seen on pathological studies,
because a central fluid-like hyper-intensity can be displayed,
even in cases of myxomatous degeneration.
2.1.2.) Hemangioma with internal septations:
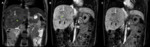
Other atypical morphological features consist of the presence of internal septations (Fig.6),
exhibiting low signal intensity on T1- and T2-w images related to the presence of a fibrotic component.
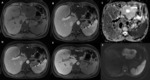
2.1.3.) Giant hemangioma:
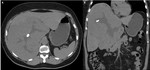
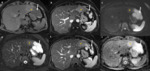
Large hemangiomas are often heterogeneous10.
They are termed giant hemangiomas when they exceed 4 cm in diameter.
Large hemangiomas may be responsible for liver enlargement and abdominal discomfort.
At US,
large hemangiomas often appear heterogeneous.
On nonenhanced CT scans,
lesions appear hypoattenuating and heterogeneous with marked central areas of low attenuation (Fig 2,
A).
After intravenous administration of contrast material,
the typical early,
peripheral,
globular enhancement is observed (Fig 2,
B).
However,
during the venous and delayed phases,
the progressive centripetal enhancement of the lesion,
although present,
does not lead to complete filling (Fig 2,
C).
At MR imaging,
T1-weighted sequences show a sharply marginated,
hypointense mass with a cleftlike area of lower intensity and sometimes with hypointense internal septa (Fig 7).
T2-weighted images show a markedly hyperintense cleftlike area and some hypointense internal septa within a hyperintense mass7.
The enhancement is equivalent to that seen at CT,
with incomplete filling of the lesion; the cleftlike area remains hypointense,
as do the internal septa12.
2.1.4.) Pedunculated hemangioma:
Pedunculated hemangiomas are rare.
They can be asymptomatic or complicated by subacute torsion and infarction.
Sometimes the liver origin of the lesion may be difficult to recognize,
as lesion can be attached to the liver by a thin pedicle,
which is nearly undetectable at imaging.
For this reason it has been described as mimicking other abdominal abnormalities,
such as hypervascular gastric or adrenal tumors.
Multiplanar reconstruction of CT scans on coronal or sagittal MR imaging can be helpful showing the origin of the lesion and signal intensity and enhancement pattern are usually consistent with the diagnosis demonstrating the typical enhancement pattern and the typical signal intensities on both T1- and T2-weighted images.
Pathologic examination either through percutaneous biopsy or surgical resection is usually not required.
Complicated pedunculated hemangiomas must be resected immediately.
However,
clear surgical indications as well as prognostic factors predicting a high risk of spontaneous bleeding were not mentioned in the reports identified in our literature review.
Further investigations about the natural course and operative indications for pedunculated hepatic hemangioma are needed.
2.1.5.) Hemangioma associated with bile duct dilatation:
Sometimes,
bile duct dilatation may occur as consequence of large hemangiomas centrally located within the liver.
Although bile duct dilatation occur more often associated with intra-hepatic cholangiocarcinoma,
all intra or extra-hepatic lesions with mass effect especially around segment IV and near the liver hilum could result in this feature.
Therefore,
bile duct dilatation does not preclude hemangioma diagnosis8.
2.1.6.) Hemangioma with calcifications:
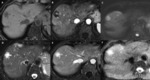
Calcifications in hemangiomas can be found with a central or peripheral location,
with multiple foci usually representing phlebolites (Fig.9).
They are better depicted on CT as tiny dense nodules or as low signal intensity foci within the hyperintense tumor on T2-w images6.
(Fig.10).
2.1.7.) Cystic and multilocular hemangioma:
Cystic and multilocular hemangiomas containing a large central cavity are extremely rare.
On MRI,
this atypical feature is represented by a single or multiple intra-tumoural cavities,
possessing long T1 and T2 relaxation times related to thrombosis and old hemorrhage.
Nevertheless,
peripheral enhancement with puddling can still occur6.
2.1.8.) Hemangioma with fluid-fluid level:
Fluid-fluid levels within hemangiomas are very rare.
A fluid–fluid level may be depicted inside the hemangioma,
which is thought to represent stagnant or slow flowing blood with sedimentation of red blood cells on the dependent portion.
The superior fluid layer consists of serum and the inferior layer contains unclotted sedimentary red blood cells.
US shows a hyperechoic or hypoechoic pattern.
This fluid-fluid level is not seen at US.
CT and especially MR imaging can easily demonstrate this feature.
The superior layer is hypoattenuating on CT scans,
isointense to muscle on T1-weighted MR images,
and markedly hyperintense on T2-weighted MR images.
The inferior layer is of higher attenuation on CT scans,
hyperintense to muscle on T1- weighted images,
and slightly hyperintense on T2-weighted images.
Nevertheless,
fluid-fluid levels in hepatic lesions do not indicate a specific diagnosis and can be observed in both malignant and benign conditions25.
Some authors have suggested that fluid-fluid levels that are clearly demonstrated with CT or MR imaging but not visible at US could be suggestive of hemangioma.
2.2) Uncommon enhancement patterns
2.2.1.) Rapidly Filling Hemangioma
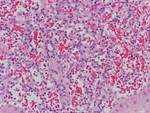
Rapidly filling hemangiomas are not very frequent (16% of all hemangiomas).
Their pathological examination corresponds to a capillary hemangioma which presents small vascular spaces and extensive connective tissue (Fig.10).
The so-called flash-filling pattern is mostly seen in small tumors under 2 cm and show fast,
intense and uniform enhancement on the arterial phase of the dynamic study,
typically,
paralleling aortic enhancement (Fig.11)10. These hemodynamic differences are probably related to the size of the vascular channels composing the hemangioma,
with hyperdynamic flow in those containing small-sized vessels.
Since these tumors may also display colour spots on Doppler examination,
differentiation from other hypervascular tumors may be problematic.
So hemangioma show immediate homogeneous enhancement at arterial-phase CT or contrast-enhanced T1-weighted MR imaging (Fig 10,
B)2,
and remain hyperattenuating or hyperintense in delayed-phase CT or MR imaging,
whereas hypervascular metastases do not.
A key point resides on the evaluation of the late interstitial phase of the dynamic study,
where flash-filling hemangiomas should not reveal contrast washout or fading.
Another important finding in diagnosis of hemangioma is an attenuation equivalent to that of the aorta during all phases of CT.
We should consider clinical context as small fibrotic scars in cirrhotic livers can also enhance in a similar pattern.
2.2.2.) Hyalinised or sclerosed hemangioma
Hyalinised or sclerosed hemangioma is rather unusual and is believed to represent the end stage of a hemangioma involution.
The replacement of the vascular spaces by hyalinised fibrotic tissue leads to marked modifications of its imaging features,
with loss of the typical globular enhancement on contrast-enhanced studies and of its high signal intensity on T2-w images on MRI (Fig.12).
Since it is virtually impossible to propose a definitive diagnosis in such a case,
pathologic proof may be necessary,
which discloses extensive fibrotic tissue and obliteration of the vascular channels.
A reasonable exception to need for pathological evaluation is when a past imaging exam displays a typical hemangioma in the same location of the present sclerosed hemangioma.
Differential diagnosis should include hypovascular tumors,
such as metastases7.
2.3) Associated liver abnormalities
2.3.1.) Hemangioma with capsular retraction:
Liver capsule retraction is not a frequent finding,
thought it should not preclude the diagnosis of hemangioma,
since it may be seen as a consequence of peripheral fibrotic changes6,
7.
A possible mechanism could be fibrous degeneration.
In this case,
other benign and malign entities should be ruled out,
such as cirrhosis,
intra-hepatic cholangiocarcinoma,
epithelioid hemangioendothelioma or,
even,
metastases with a fibrotic component.
This entity is rare and most of time is diagnosed through surgical biopsy.
2.3.2.) Hemangioma in Fatty Liver Infiltration
Diffuse fatty infiltration of the liver is a common finding and may change the typical appearances of lesions,
making them more difficult to characterize due to the decreased liver–lesion contrast,
which,
in severe steatosis,
may even be reversed in both imaging modalities,
with the hemangioma being hypoechoic and denser,
respectively,
when compared to the adjacent fatty liver.
At US,
a hemangioma may appear slightly hyperechoic,
isoechoic,
or hypoechoic relative to a fatty liver11.
Posterior acoustic enhancement is usually observed.
At nonenhanced CT,
the lesion may be hyperattenuating relative to the liver or may not be seen.
Contrast-enhanced CT shows peripheral enhancement and delayed filling,
an appearance similar to that of a hemangioma in a normal liver.
However,
at arterial-phase imaging,
the hemangioma may be isoattenuating relative to the liver.
Sometimes,
in CT studies,
a peripheral dense halo may be seen surrounding the hemangioma.
This corresponds to spared non-fatty parenchyma,
as a consequence of the preferential arterial inflow and/or decreased portal flow around the lesion due to compression phenomena12.
MR imaging is more helpful than CT and allows reliable detection and differentiation of hemangiomas from other hepatic masses13.
MRI may be preferable in this setting,
especially using in-phase and out-of-phase gradient-echo sequences.
The high signal intensity on T2-w images and the enhancement features are preserved,
thus,
endering the diagnosis relatively straightforward.
2.3.3.) Hemangioma in cirrhotic liver
Hemangioma in cirrhotic liver is rare,
with an incidence estimated to be about 1.7% at pathologic examination and 0.6% at CT,
which is clearly lower than the frequency in unselected autopsy series or at CT in non-cirrhotic patients11.
Despite the stiffening of the liver parenchyma,
hemangiomas generally retain their characteristic findings,
both on T2-w images and the enhancement pattern.
In rare cases,
however,
mostly in advanced cirrhosis,
they can lose their typical imaging features due to the development of fibrosis,
determining lower volume and lower signal intensity on T2-w images11.
2.3.4.) Hemangioma with Arterial–Portal Venous Shunt
Arterial–portal venous shunts are mainly associated with hepatic malignancy,
but can also be seen in benign liver masses8,9,
in particular hemangiomas.
This entity is usually asymptomatic.
An arterial–portal venous shunt can be detected with helical CT or dynamic contrast-enhanced MR imaging (Fig 15).
The findings consist of early parenchymal enhancement associated with early filling of the portal vein.
Peri-lesional enhancement is seen in 19–25% of cases and manifests itself as transient hepatic attenuation differences (THAD),
corresponding to staining areas seen on the arterial phase of liver enhancement,
fading away on the subsequent phases of the dynamic study6,7.
Pathological explanation resides in arterio-venous shunting related to their hyperdynamic status.
Although THAD is mostly seen in cases of small flash-filling hemangiomas,
larger tumors can also show the same perfusion abnormalities due to portal vein compression and compensatory arterial inflow,
determining a peri-tumoral area of parenchymal enhancement6 (Fig.14 ).
It must be stressed that peri-lesional THADs have been described in association with a variety of focal liver lesions,
such as metastases,
abscesses and hepatocellular carcinoma8.
2.3.5.) Nodular hyperplasia surrounding hemangioma
Hemangioma has been reported in approximately 20% of cases of patients having a concomitant FNH (Fig.16) ,
a prevalence that is higher than that observed in the general population.
A common physiopathological mechanism may explain this association,
which is believed to result from a focal disturbance of the liver blood supply facilitating a hyperplastic response and subsequent development of these benign tumours12.
3.
Pitfalls in US,
CT and MRI
3.1) Not all hyperechoic liver lesion on ultrasound are hemangioma.
A hyperechoic liver lesion on ultrasound is not pathognomonic of hemangioma,
it can arise from a number of entities, both benign and malignant.
Examples of benign lesions are focal hepatic steatosis,
hepatic adenoma with high fat content (Fig.17),
focal nodular hyperplasia,
focal hepatic steatosis,
and inflammatory pseudotumour of the liver.
Examples of malignant lesions are hepatic metastases (colorectal carcinoma (up to 50% of hyperechoic liver metastases),
treated breast cancer,
endocrine tumors of the pancreas,
renal cell carcinoma,
thyroid carcinoma,
melanoma,
some sarcomas,
choriocarcinoma),
hepatocellular carcinoma or cholangiocarcinoma.
3.2) In arterial phase,
hemangioma typically show discontinuous,
nodular,
peripheral enhancement.
Periphery continuous enhancement is not typical of liver hemangioma and you should consider another differential diagnosis as metastases,
abscess,
cholangiocarcinoma,
hepatocarcinoma (Fig.19) in cirrhotic liver or treated lesions.
3.3) Hemangioma is a hypervascular liver lesion that always have attenuation / signal equivalent to that of the aorta during all phases.
A hypervascular liver lesion that demonstrate fading or wash out in venous phase or delayed-phase images are not a hemangioma.
In this case we should consider nodular hyperplasia focal,
adenoma or metastases,
(Fig.20) and hepatocarcinoma as differential diagnosis.
MRI is the most useful tool to make a correct differential diagnosis.
3.4) Not all giant liver lesions with central scar are giant hemangiomas.
As principal differential diagnosis we should consider fibrolamellar HCC (Fig.21),
metastases with central necrosis,
and FHN.
3.5) Not all liver lesions with early nodular peripheral enhancement in arterial phase are hemangiomas.
We should always consider all phases in dynamic study.
In arterial phase,
liver lesion with discontinuous,
nodular enhancement could not be hemangioma,
we should always consider the clinical context.
In our department we observed some cases of liver lesions with nodular periphery enhancement that was an HCC treated by hepatic embolization,
and remain with a halo of tumoral tissue that enhances contrast.
3.6) Not all liver lesions with blood pool are hemangiomas,
we should also consider as differential diagnosis peliosis hepatic (Fig.
22,
23),
hemagioendothelioma and liver angiosarcoma.
3.7) Not all hypointensity liver lesion during late dynamic phase with hepatocyte-specific contrast agents means wash out.
Differentiation of hepatic hemangiomas and metastatic lesions is a common clinical problem and can influence treatment.
MRI is frequently used to characterize liver lesions,
and findings on T2-weighted and gadolinium-enhanced MR images have been found to play a key role in differentiation of hemangioma and metastasis.
Gadoxetate acid or its salt,
gadoxetate disodium is a hepatobiliary contrast agent and is preferentially taken up by hepatocytes.
It has extended persistence in the liver.
It has shown dose-independent renal (41.6–51.2%) and biliary (43.1– 53.2%) elimination and an enterohepatic recirculation rate of approximately 4%.
Because the effect of recirculated contrast material might be less in gadoxetate disodium–enhanced MRI than in gadopentetate dimeglumine–enhanced MRI,
the bright dot sign and the minimal enhancement observed in hemangiomas during the late dynamic phase are minimized on gadoxetate disodium–enhanced MR images.
In addition,
because the signal intensity of the liver continues to increase with time,
these delicate enhancement findings might be obscured.
Another explanation for difficulty with hemangiomas,
besides the lower gadolinium dose and effect of recirculated contrast material,
is more rapid removal of the contrast agent from the vascular space.
For these reasons,
hemangioma demonstrate avid arterial enhancement and then,
because of rapid progressive hepatic parenchymal enhancement,
become hypointense to normal liver as early as 3 minutes after injection,
simulating washout.
This recently reported phenomenon was described as “pseudo washout” 16 (Fig.24).
Because of these issues,
we do not recommend the use of gadoxetic acid for the routine imaging of known or suspected hemangiomas.
4.
Differential Diagnosis
During previous discussion we reviewed the most common differential diagnosis.
A summary table of imaging features of principal differential diagnosis follows.

Fig. 25: Differential Diagnosis.
The (+) sign indicates the frequency of the findings: (+++) very common finding, (++) less common finding, (+) rare finding; (a) extremely rare finding; (b) lesion > 2cm in a cirrhotic liver; HF= homogeneous with fading pattern; HP = homogeneous and persisting pattern; HW = homogeneous with "washout" pattern; HYPO = hypoenhancing lesion on hepatobiliary phase; ISO/HYPER = iso/hyperenhancing lesion on hepatobiliary phase; MOSAIC = mosaic pattern; PUD = "puddling" pattern; RF = fading ring pattern; RW = ring with "peripheral washout" pattern; SCAR = central scar.
5. Hepatic hemangiomas diagnosis - How to do it right?
In order to achieve the correct diagnosis,
there are some points that you should always consider:
1.
Medical history : Age and gender of patient,
risk factor for acute/ chronic hepatopathy,
history of previous neoplasm or chemoembolization,
use of oral contraceptive or anabolic steroids.
2. Clinical and laboratory abnormalties: anemia,
fever,
weight loss.
3. Dynamic studies including all phases.
4. Do not use gadoxetic acid for the routine imaging of known or suspected hemangiomas.
5. Typical and atypical features of hemangioma,
so if you do not have certain diagnosis you should call it as an indeterminate lesion,
and never diagnose it as an atypical hemangioma.
6. The most common pitfalls and knowledge on how to avoid them.
7.
The principal differential diagnosis.
8.
Review previous exams regardless of timeline.
9. If you are in doubt about diagnosis,
you must recommend pathological examination.