This poster is published under an
open license. Please read the
disclaimer for further details.
Keywords:
Image verification, Embolism / Thrombosis, Connective tissue disorders, Technology assessment, Image manipulation / Reconstruction, CT, Lung
Authors:
I. Leineman, R. Faizenberg, T. Golimbievskaya, M. Shostak, I. Itskovich, V. Mazurov; Saint-Petersburg/RU
DOI:
10.1594/ecr2017/C-1760
Methods and materials
Study population
Between 2013 and 2015,
in North-Western State Medical University named after I.I.
Mechnikov (Saint Petersburg),
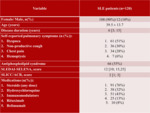
120 patients (Table 1),
aged 39.5 ± 13.7,
with active SLE (SELENA-SLEDAI 12 [10; 15.25]), but without known chronic respiratory diseases,
underwent laboratory tests,
electrocardiograms and echocardiograms,
pulmonary function and imaging tests at baseline and after 6 months of therapy.
55% of SLE patients had secondary APS.
Imaging tests
Patients were prospectively assessed using GE Lightspeed VCT 64 Slice CT Scanner.
For reconstruction of standard native CT scans we used work station,
which includes post-processing software.
Software consists of such techniques as multiplanar reformations (MPR),
maximum intensity projection (MIP),
minimum intensity projection (mIP) and color mapping (Page Phase and Hot Iron).
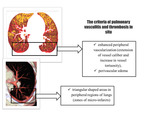
According to the reconstructed CT scans we formulated the criteria (Fig. 2): for detection of pulmonary vasculitis on color maps we used the criteria - zones of enhanced peripheral vascularization (extension of vessel caliber and increase in vessel tortuosity) and perivascular edema; for detection of thrombosis in situ – trianglar shaped zones of micro-infarctions in the peripheral regions of lungs.
Patients underwent routine CT contrast-enhanced protocol to verify the areas with pulmonary thrombosis and pulmonary perfusion scintigraphy to assess the areas with active vascular lesions and impaired perfusion.
Images of native CT scans,
post-processed CT scans,
CT-angiograms and perfusion scans were independently reviewed by three blinded physicians.
Statistical analysis was performed using Statistica StatSoft v.
10.0.
The study was approved by local ethics committees.
There are no conflicts of interest,
and the researchers acquired the patients’ informed consent before enrollment.