Imaging features in CT and MRI
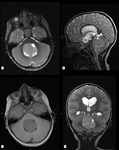
Medulloblastomas arise in the cerebellum and hence in imaging studies present as masses located in the posterior fossa (Fig.
1).
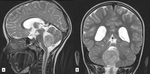
Majority of tumors lie in the midline and involve cerebellar vermis or vermis and cerebellar hemisphere (Fig.
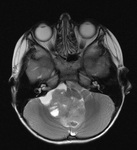
2 and 3); isolated involvement of the hemisphere is less commonly observed (Fig.
4) [2,10-12].
Besides presenting as round or polycyclic masses,
pediatric medulloblastomas rarely can also have a very nodular appearance,
which is characteristic of the MBEN histological variant (Fig.
5).
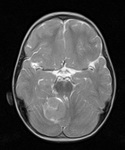
Imaging features of various degrees of obstructive hydrocephalus are present in approximately 95% of patients (Fig.
6 and 7) [2,10-13].
Typically in noncontrast CT scans medulloblastoma presents as a well-defined,
hyperattenuating mass,
in > 90% of cases surrounded by a rim of vasogenic edema (Fig.
8) [2,10,11].
In 90% of the tumors contrast enhancement is seen,
usually prominent and homogenous,
occasionally irregular [10,11,13].
Necrotic,
nonenhancing regions or cysts (either multiple and small or a single,
large cavity) are observed in 50-60% of medulloblastomas (Fig.
9),
whereas calcifications are found in around 20% of cases (Fig.
10) [2,10,11].
In CT scans performed in children,
who did not commence radiation therapy and did not have shunts placed,
falx cerebri should be carefully checked for calcifications,
as their presence may indicate that the tumor developed in a patient with nevoid basal-cell carcinoma syndrome (Gorlin syndrome) and other signs of the syndrome should be checked for [10,11,14].
Medulloblastoma,
which in those patients always belongs to the SHH subgroup,
may be the first sign of the syndrome and its diagnosis is clinically important,
as it warrants an alternative,
radiation limiting,
treatment strategy,
in order not to increase the already elevated risk of basal-cell carcinoma [2,10,11].
Imaging features of medulloblastomas in MRI are variable.
Commonly,
on T1-weighted MR images medulloblastomas are iso- to hypointense relative to normal gray matter (Fig.
11 and 12) [2,10,11].
On T2-weighted images medulloblastoma often presents as a heterogeneous mass,
with hypo- and hyperintense regions due to a presence of cysts,
necrotic regions and calcifications,
which in overall have iso- to hyperintense signal compared to normal gray matter (Fig.
11 and 12) [2,10-12].
Generally the tumor is surrounded by a visible vasogenic edema.
In a significant majority of medulloblastomas,
though not in all,
contrast enhancement is present (Figure 12 and 13).
Additionally,
the intensity of enhancement may range from weak to extensive and present different levels of heterogeneity [2,10-12].
High cellularity and high nuclear-to-cytoplasm ratio of medulloblastoma lead to a restriction of diffusion of water particles in the tumor.
This phenomenon results in a characteristically high signal of the tumor in diffusion-weighted MR images and in low apparent diffusion coefficient (ADC) values,
which results in low signal of the tumor on ADC maps (Fig.
14) [10,11].
Medulloblastomas in MR spectroscopy are characterized by high levels of choline (Cho),
taurine (Tau) and lactate,
an increased choline-to-creatine (Cho/Cr) ratio and low concentrations of creatinine and N-acetylaspartate (NAA)(Fig.
15) [10,11].
The most distinctive feature of medulloblastomas in MRS is the presence of a high taurine peak at 3.3 ppm,
which is specific for this disease among posterior fossa tumors [15].
Metastases
In all patients brain and spine should be examined in search for potential metastases,
which are diagnosed in around 30-40% of cases and their presence influences treatment protocol.
Screening for leptomeningeal spread of the disease consists of cerebrospinal fluid (CSF) cytologic analysis and MRI imaging,
including T1-weighted gadolinium-enhanced sequences.
Subarachnoidal spreading occurs more frequently along the normal CSF flow,
hence the most common location of metastases is along the posterior surface of the spinal cord (Fig.
16) [10,11,13].
They can present as a smooth enhancement of the surface of the spinal cord,
an extramedullar,
nodular enhancement or as intramedullar enhancing foci.
Intracranial metastases present as small,
nodular lesions,
identification of which may be difficult on T2-weighted images – they can be more easily assessed using additionally FLAIR,
proton-density and DWI sequences (Fig.
17) [10,11,13].
Due to implementation of chemotherapy,
hematogenous dissemination is rarely observed [11,16].
Metastases in bone,
the most common extraneural site,
appear on plain radiography and CT as radiopaque lesions and on T1-weighted MR images as hypointense foci,
which enhance after contrast administration [10,11].
Differential diagnosis
Differential diagnosis of medulloblastoma includes other tumors that may develop in posterior fossa,
above all ependymomas and pilocytic astrocytomas [10-12].
In ependymomas calcifications are more often observed,
the tumor frequently extends from the fourth ventricle,
through the foramen of Luschka into the cerebellopontine cistern (Fig.
18) and contrast enhancement in CT is very heterogeneous [10-12].
Pilocytic astocytomas,
more frequently than medulloblastomas,
arise in cerebellar hemispheres and in contrast to medulloblastomas tend to be hypoattenuating on CT images [10-11].
The tumors can be differentiated using ADC values,
which are correlated with local cellularity and nucleus-to-cytoplasm ratios [10,11,17].
A study conducted by Rumboldt et al.
indicates that in children ADC cutoff values of > 1.4x103 mm2/s for pilocytic astrocytoma and < 0.9x103 mm2/s for medulloblastoma and atypical teratoid/rhabdoid tumor (AT/RT) are 100% specific,
which leads to,
accordingly,
hyperintensity and hypointensity on ADC maps (Fig.
19) [17].
ADC values of ependymomas range between 1.00 and 1.30x103 mm2/s [17].
Association between MR imaging features and molecular subtypes
In recently conducted studies tumor location was established as a potentially most indicative imaging feature of the disease’s molecular subgroup.
Cerebellar peduncle or cerebellopontine angle cistern location was found by Perreault et al.
to be unique to the WNT subgroup of medulloblastoma [18].
It was also established as the most common site of WNT tumors,
with 75% of tumors of this subtype being in that location (Fig.
20) [18].
Lateral,
hemispheric location of medulloblastoma was recognized as indicative of the SHH molecular subgroup of the disease (Fig.
4 and 21) [18-20].
To date,
no WNT tumors were identified in this location and only 3 out of 157 analyzed non-WNT/SHH tumors were found in the cerebellar hemisphere [20].
However,
SHH tumors are not limited to this site and may,
potentially comparably as often,
be found in the midline location [18,20].
Group 3 and 4 tumors are mostly located in the midline,
occupying the fourth ventricle (Fig.
22) [18,20].
More than a half of Group 3 tumors was discovered by Perreault et al.
to have ill-defined margins and the feature was characteristic of this molecular subgroup [18].
Minimal or no enhancement of a tumor located in the midline was found to be a feature characteristic of group 4 and differentiating it from group 3 tumors [18,20].
Additionally,
contrast enhancement patterns were found to be associated with survival rates in patients >3 years old with non-WNT/SHH medulloblastomas in the midline location.
Łastowska et al.
established that patients from this population with no or weak enhancement had significantly better overall survival and event-free survival,
whereas extensive enhancement was associated with worse outcomes compared to weak or even heterogeneous enhancement (Fig.
23) [20].
Monitoring for disease recurrence
Monitoring for disease recurrence,
which happens often,
consists of regular MR imaging,
performed every 3-6 months during the period of 5 years after the initial treatment [10,11,13].
Directly after surgery a contrast enhancing rim around the resection cavity may be visible,
which begins to diminish after about 5 weeks and completely disappears a year after surgery.
Hence,
in surveillance imaging any new contrast enhancing areas > 5 weeks after surgical treatment should be considered suspicious and may possibly indicate disease recurrence [10,11,13].
Usually it presents as leptomeningeal enhancement or nodular,
enhancing foci in the central nervous system (Fig.
16 and 17) [10,11,13].