The term non-radiographic axial spondylarthritis (nrAxSpA) is referring to patients suffering from the early phase of ankylosing spondylitis (AS).
These patients presenting with a clinical picture suspicious for AS but who haven’t yet developed radiographic sacroiliitis,
would be diagnosed today as having nrAxSpA based on magnetic resonance imaging (MRI) and a composite of clinical manifestations.[1]
Among rheumatologic diseases arthritis is the leading cause of disability in adults.
Radiologists are most familiar with diagnostic criteria, which can be defined as a set of symptoms,
signs and laboratory tests for use in routine clinical care to guide the management of individual patients.
In comparison,
classification criteria are standardized definitions primarily intended to enable clinical studies.
Unfortunately,
most rheumatologic diseases are systemic disorders with heterogeneous features that do not have a single clinical,
imaging or pathologic feature to serve as a gold standard.
As such,
the American College of Rheumatology currently provides approval only for classification criteria [1].
Classification criteria
One of the most commonly used classification criteria is the 1984 modified New York criteria for established AS,
which includes clinical and radiologic components (Table 1).
Because the modified New York criteria have an obligatory radiographic item and because it is well known that prolonged inflammation is required for structural radiographic changes,
the modified New York criteria perform poorly for the detection of early AS.
Most recently,
the Assessment of SpondyloArthritis International Society (ASAS) developed two sets of classification criteria to encompass the spectrum of spondyloarthritis,
which are divided on the basis of the predominant clinical presentation into axial spondyloarthritis and peripheral spondyloarthritis.
The 2009 ASAS criteria for axSpA are shown in Table 2 [2].
Going beyond the modified New York criteria,
which concentrate on conventional radiographs of the sacroiliac joints for the classification of AS,
the ASAS criteria add active inflammation of the SIJ as obtained by MRI and human leucocyte antigen (HLA) B27 to classify patients with chronic back pain starting at a young age as axSpA.
This should be considered as one disease that includes AS,
the radiographic form,
as well as the non-radiographic (nr-axSpA) form.
Also,
nr-axSpA may represent an early stage of AS but may also just be an abortive form of a disease which does cause much pain but which may also never lead to structural changes of the axial skeleton [3].
A study found that patients with nr-axSpA were the youngest,
and they experienced chronic low back pain for the shortest duration at slightly over 6 years compared with nearly 11 years for patients with AS.
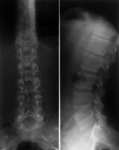
Among patients who had been diagnosed with nr-axSpA,
there was a delay of approximately 5 years between presentation of inferior back pain (IBM)(Figure 1) and diagnosis [2].
The sensitivity of conventional radiographic methods is known to be low in early stages of disease.
The demonstration of sacroiliitis in patients with a possible AS who have normal findings of the sacroiliac joints at conventional radiography is important because the diagnosis of sacroiliitis in these patients is difficult to make solely on a clinical basis [4].
Epidemiology
There are limited data on the incidence,
prevalence and proportion of patients classified or diagnosed as nr-axSpA and radiographic axSpA in the population or among patients who present to the general practitioner or the rheumatologist.
While the prevalence of IBP has been shown to be about 6% in the USA [3],
the prevalence of axSpA is a lot lower.
When including all forms of SpA,
it may well be >1%.
In a recent consecutive study,
the majority (56%) of patients presenting to the outpatient clinic with typical symptoms already had radiographic changes in the sacroiliac joints.
Overall,
the proportions of patients with nr-axSpA and radiographic axSpA seem to be largely similar,
and it can be stated that both subtypes are equally relevant for the axSpA concept.
Clinicians should be aware about the similarities and slight differences between the subtypes,
which in part may also represent different stages of the disease and also different disease courses [3].
Differences between nr-axSpA and radiographic axSpA
Despite the many similarities,
patients classified as nr-axSpA have also shown differences as compared with those classified as radiographic axSpA.
Early in disease,
cytokines mediate inflammatory changes in various musculoskeletal tissues,
including the entheses,
synovium,
and bone marrow.
Localized structural bone changes are also seen,
ranging from new bone formation (syndesmophytes,
enthesophytes,
and ankylosis) to bone destruction (erosions).
Of note,
bone formation is not a uniformly diffuse process but is localized and confined to the periosteal bone compartment.
In contrast,
trabecular bone mineral density decreases globally,
including in the axial and peripheral skeleton.
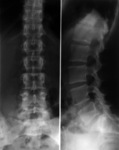
[1] Characteristic active and chronic spinal features of radiographic axSpA include spondylitis (typically manifest as a corner lesion of a vertebral body,
including anterior locations known as Romanus lesions ( Figure 2 and Figure 4),
which may be edematous,
fatty,
or sclerotic),
spondylodiscitis (manifest as a bone marrow lesion near the discovertebral junction,
also known as an Andersson lesion -Figure 3),
arthritis of the paravertebral synovial joints,
enthesitis of spinal ligaments,
syndesmophytes,
or ankylosis.
In recognition of the fact that radiographs are not sensitive for the detection of early disease,
if radiographs show normal findings,
MR imaging of the sacroiliac joints is recommended as the next diagnostic step (Figure 4 and 5). Spine involvement AS is often centered at the thoracolumbar or lumbosacral junction, and MR at these sites optimally depict subtle and early abnormalities.
Early radiographic findings are erosions at the anterior margins of the vertebral body at the discovertebral junction.
These focal areas of osteitis become increasingly sclerotic,
a finding termed the “shiny corner sign” (Figure 2)
Definite subchondral or periarticular bone marrow edema is mandatory for the definition of active sacroiliitis at MR imaging for classification purposes.
The bone marrow edema can be strongly hyperintense or more subtle.
When the lesion is singular,
bone marrow edema should be seen on more than one section; and when subtle,
changing the brightness and contrast of the image and comparing with the center of the sacrum as an internal control are helpful to improve confidence.
When comparing patients with axSpA to nr-AxSpA patients,
in addition to the expected more advanced structural disease,
the sacroiliac joints have significantly more inflammatory lesions in the radiographic axSpA patients,
irrespective and independent of CRP levels.
In addition,
patients with radiographic AxSpA have a more advanced spinal disease compared to the nrAxSpA patients.[5]
The combined features BME and/or erosion were observed in 50% and in 16% of nr-axSpA patients in a study [6],
supporting a previous finding that structural damage may start early in the SI joint,
long before the damage is detectable by pelvic radiography.
Ankylosing spondylitis–associated sacroiliitis at MR imaging is typically bilateral but is less often symmetric on MR images,
compared with radiographs (MR imaging symmetry in 60% of cases vs radiographic symmetry in 86%),
and may even be unilateral in 9% of cases.
[5]
Therapy
This diagnostic improvement has considerable implications for patient treatment because it allows earlier diagnosis and access to therapy.
It may be years before changes of sacroiliitis are evident on radiographs,
during which time the symptom burden and impaired quality of life can be similar to those experienced by patients with established axial SpA.
Furthermore,
patients with early axSpA respond to treatment with TNFi,
and therapy at this stage may be more valuable than that in patients with recognized disease.
Another benefit of the improved diagnostic certainty before MR imaging is the exclusion of axSpA as the cause of back pain in some patients.
This is important because TNFi may be associated with serious complications and unnecessary use of it should be avoided.[7]
MR imaging can show rapid improvements after therapy,
with decreased bone marrow edema as early as 6 weeks after the start of nonsteroidal anti-inflammatory drug therapy and decreased erosions and increased “backfill” (as defined by increased signal intensity on T1-weighted MR images that is clearly demarcated from adjacent normal bone marrow by an irregular area of hypointensity reflecting sclerosis at the border of the eroded bone) at 12 weeks after initiation of therapy with a biological agent[1].
Differential Diagnosis and Pitfalls.
The anatomic structure of the sacroiliac joint is extremely complex,
with a number of variants that may appear irregular at imaging.
Among the most common variants encountered at imaging are patchy bone marrow fat,
an accessory sacroiliac joint,
and transitional vertebrae.
Bone marrow fat infiltration often accompanies other abnormalities in axSpA,
but because such infiltration can be seen in a surprisingly high percentage of normal and asymptomatic volunteers (as many as 33%–53%),
it has limited diagnostic utility when seen in isolation.
[1]
An accessory sacroiliac joint is the most common variant.
It is located at the posterosuperior portion of the joint and may develop degenerative changes,
thereby causing low back pain.
An iliosacral complex is formed by an iliac projection inserted into a complementary sacral recess and is usually located at the transitional zone between the synovial and ligamentous portions of the joint.
A bipartite iliac bone plate is located at the posteroinferior portion of the joint.
Other anatomic variants such as a crescentic iliac articular surface (usually at the posterosuperior portion of the joint),
semicircular defect in the articular surfaces,
and small ossification centers of the sacral wings have also been described [8]
Transitional lumbosacral vertebrae are also commonly encountered (estimated prevalence,
nearly 20%),
and approximately half of these will demonstrate dysplastic enlarged transverse processes that pseudoarticulate with the sacrum.
Degeneration of both the accessory sacroiliac joint and transitional vertebrae pseudoarticulations may result in back pain and can mimic sacroiliitis.
In cases of isolated bone marrow edema,
lesions that are greater than 1 cm and located away from the proximal or distal margins of the sacroiliac joint being more meaningful for the diagnosis of active sacroiliitis [1].
Many conditions,
such as osteoarthritis,
septic sacroiliitis,
insufficiency sacral fractures,
osteitis condensans ilii,
and bone tumors,
may mimic the inflammatory lesions seen in AS.
However,
sacroiliac joint inflammation in AS is usually limited to the bone and sacroiliac joint space and does not cross anatomic borders.
With osteoarthritis,
it is not uncommon to see anterior osteophytes surrounding the sacroiliac joints.
Osteitis condensans ilii is typically seen in middle-aged women,
in whom it manifests as sclerotic areas,
mainly in the iliac bone,
with relatively normal joint spaces.
In addition,
ligaments surrounded by blood vessels may appear to be,
and may erroneously be interpreted as being,
actively inflamed on STIR images (12),
and clefts,
ligaments,
and cysts can also be confused with structural lesions .
Frthermore,
inadequate fat suppression may cause normal anatomic structures to appear hyperintense,
especially in the posterior part of the sacrum.
In addition,
the so-called coil effect may result in brighter signal closer to the coil-body interface.
Similar effects on the adjacent soft tissue help distinguish these conditions from real alterations.[8]
However,
a negative MRI does not exclude nrAxSpA.
Sometimes there is MRI aspect of sacroiliitis but the patient hasn’t enough criteria for AxSpA and if it’s unilateral,
the radiologist should think about psoriatic arthritis (Figure 6).