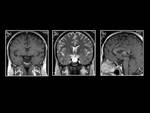
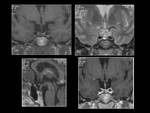
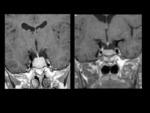
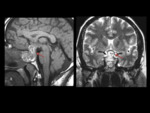
In order to analyze a sellar or parasellar mass on MRI,
the following anatomic approach could be used[4]:
1. First identify the pituitary gland and sella turcica.
2. Then determine the epicenter of the lesion and whether it is in the sella or above,
below or lateral to the sella.
3. If it is in the sella,
determine whether or not the sella is enlarged.
4. Once the location of the mass is clear,
analyze the signal intensity patterns: is the lesion cystic or solid?
5. Does it contain any abnormal vessels?
6. Are there any calcifications? And so on.
7. Finally establish a Differential Diagnosis.
Spectrum of Pathologies involving the Anatomical Structures in the Sellar and the Para-sellar region: [5]
*Pituitary Gland Lesions*
Congenital Abnormalities
· Empty sella turcica
· Pituitary Gland Hypoplasia
· Pituitary Duplication
Neoplastic lesions
· Rathke's cleft cyst.
· Microadenomas (size < 1 cm)
· Macroadenomas (size > 1 cm)
· Craniopharyngiomas
· Pituicytomas and Granular cell tumors
Inflammatory lesions
• Abscesses and Infections
Non-Infectious Inflammatory lesions
• Lymphocytic Hypophysitis.
• Granulomatous Giant cell Hypophysitis.
• Sarcoidosis
• Infundibulo-Neuro Hypophysitis.
Vascular and Ischemic Lesions
• Sheehan's Syndrome
• Pituitary Apoplexy
Metabolic disorders
· Diabetes Insipidus
· Hypermagnesemia
· Haemochromatosis
· Pituitary hyperplasia
*Pituitary Stalk Lesions*
Ø Rathke's cleft cyst
Ø Craniopharyngioma
Ø Germinoma
Ø Eosinophilic Granuloma
Ø Metastasis
Ø Lymphoma
*Optic Chiasmatic lesions*
• Glioma
• Demyelinating lesions
*Hypothalamic lesions*
• Glioma
• Hamartoma
• Eosinophilic Granuloma
• Germinoma
*Meningeal Lesions*
• Meningitis
• Meningioma
*Internal Carotid Artery Lesions*
• Aneurysm
• Ectasia
*Cavernous sinus lesions*
• Schwannoma
• Thrombophlebitis
• Carotico-Cavernous fistula
*Suprasellar lesions*
· Arachnoid cyst
· Epidermoid cyst
· Teratoma
· Lipoma
*Sphenoid sinus and skull base lesions*
· Sinusitis
· Mucocele
· Squamous cell carcinoma
· Chordomas
· Chondrosarcomas
· Metastasis
Pituitary fossa pathology
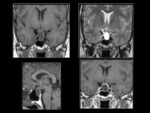
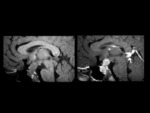
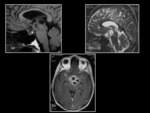
The "empty sella" appearance is due to the patulous diaphragma sella and extension of the suprasellar subarachnoid space (Figure 1). The superior margin of the pituitary is flattened or concave.
The sella turcica is often enlarged,
and there may occasionally be intrasellar herniation of the anteroinferior portion of the third ventricle or optic nerves/chiasm.
There is usually no functional impairment of the pituitary gland.
However,
a partially empty sella appearance can be seen in a premenopausal female with additional signs and symptoms of pseudo-tumor cerebri. [6] Pituitary microadenomas have also been reported to coexist with an empty sella. [7]
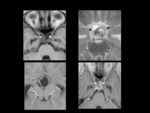
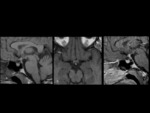
Rathke's cleft cysts are congential cystic lesions in the sellar region (25%),
suprasellar region (5%) or both (70%),
which are lined by cuboidal or columnar cells.
The MR signal intensity and CT attenuation of these lesions is variable due to the amount of proteinaceous material within them (Figure 2).
On occasion,
they can show peripheral enhancement.
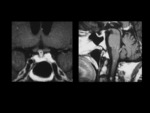
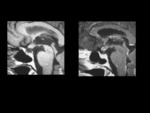
Pituitary hyperplasia can be a physiologic manifestation during pregnancy or lactation due to hypertrophy of prolactin cells.
It can also occur in the setting of hypofunction of the thyroid gland,
adrenal glands,
or gonads as a result of the lack of negative feedback [8].
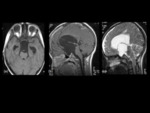
Nodular or diffuse pituitary hyperplasia can also be seen as a cause of Cushing's disease (Figure 3).
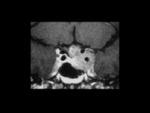
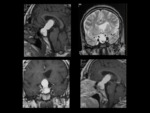
In pituitary apoplexy syndrome,
patients present with ophthalmoplegia,
headache,
and visual loss due to pituitary infarction/hemorrhage.
This may occur in the setting of massive hemorrhage or tumor necrosis.
Acute hemorrhage will appear hypointense on T2-weighted images with T1 hyperintensity as the hemorrhagic products evolve (Figure 4). [9] There is usually no hemosiderin deposition associated with pituitary hemorrhage.
In the postpartum setting,
insufficiency of the hypophyseal portal system can result in infarction of the adenophysis,
i.e.
Sheehan's syndrome.
Adenomas comprise the majority (80%) of pituitary lesions.
A large percentage of these (approximately 75%) are functioning [10] and result in endocrine abnormalities.
Hyperprolactinemia is the most common manifestation.
They can be clinically silent and detected incidentally on MRI performed for other reasons.
Patients may have symptoms due to compression of adjacent structures,
particularly the optic chiasm and optic nerves.
Microadenomas are defined as lesions ≤10 mm.
Upon immediate imaging after contrast enhancement,
they are typically hypointense on T1-weighted imaging relative to the remaining normal pituitary tissue.
As there is up to a 20% false-negative rate in the detection of microadenomas,
dynamic contrast-enhanced MRI can be performed to increase the sensitivity and specificity. [11]
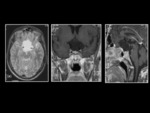
Macroadenomas are >10 mm in diameter.
These lesions are isointense to gray matter on T1- and T2-weighted imaging and demonstrate homogenous enhancement.
Approximately,
10% of adenomas can be complicated by hemorrhage,
infarction or necrosis/cystic change (Figure 5),
which alters the MRI signal intensities.
Adenomas can expand cephalad into the suprasellar cistern and compress the optic chiasm and optic nerves,
caudally to erode the sella turcia,
and laterally to compress or invade the cavernous sinuses.
A prolactin level that is >1000 ng/mL correlates with cavernous sinus invasion. [12] A reliable imaging finding of cavernous sinus involvement is abnormal tissue between the lateral wall of the cavernous sinus and the cavernous internal carotid artery (ICA). [13]
Craniopharyngiomas arise from remnants of Rathke's pouch along the path of the craniopharyngeal canal.
Most involve both the intrasellar and suprasellar compartments (70%); 10% are intrasellar and 20% are suprasellar.
They can be seen in children (peak at 5-10 years) and adults (fifth through seventh decades).
Craniopharyngiomas can cause symptoms such as visual disturbances,
endocrine abnormalities,
motor deficits and increased intracranial pressure.
The main histologic subtypes are adamantinomatous and squamous papillary.
Adamantinomatous subtypes are more often encountered in the suprasellar region in children.
They are predominantly cystic,
lobulated calcifications and they are highly likely to recur.
The squamous papillary subtype presents more frequently in adults.
These craniopharyngiomas are predominantly solid,
spherical and they occur in either the intrasellar or suprasellar regions. [14] Calcification is seen in 80% of lesions and is best delineated on CT.
Cystic areas are observed in 85% of cases and can have a variable appearance on MRI depending on the degree of methemoglobin and/or high protein.
Enhancement can be solid or nodular (Figure 6).
Meningiomas in this region originate from the tuberculum sella,
anterior clinoid processes,
diaphragma sellae,
planum sphenoidale and upper clivus.
They can cause osseous changes including hyperostosis and erosion.
As meningiomas in other locations,
they are isointense to slightly hypointense on T1-weighted imaging.
They are isointense to slightly hyperintense on T2-weighted imaging and they enhance homogenously and intensely.
The vascular supply to these lesions is from the carotid meningeal and ophthalmic arteries.
Infundibular pathology
The infundibulum has a funnel-shaped contour 3 to 3.5 mm superiorly and 2 mm inferiorly.
A variety of inflammatory (such as giant-cell granuloma and Langerhans cell histiocytosis) and neoplastic lesions can involve the infundibulum.
These lesions often involve the hypothalamus and can result in diabetes inspidus.
Langerhans cell histiocytosis (LCH) is the most common pediatric infundibular tumor [15,16] There may be meningeal involvement and choroid plexus lesions. In nearly all cases of LCH,
the normal T1 hyperintensity of the neurohypophysis is absent.
Neoplastic processes that can enlarge the stalk include lymphoma metastases,
germinoma and teratoma.
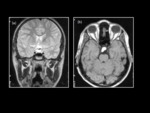
Metastases have been reported in 3% of patients with carcinoma,
particularly breast or primary bronchogenic (Figure 7). [17] The hematogenous route is likely and additional intra-axial or extra-axial lesions may be a diagnostic clue.
They may have a dumbbell morphology from invasion through the diaphragma sella.
Primary brain tumors such as medulloblastoma,
pineoblastoma and germinoma,
and lymphoma and leukemia may spread to the infundibular or suparsellar region via cerebrospinal fluid (CSF) seeding (Figure 8).
Germinomas are more commonly seen in the pediatric population,
presenting with diabetes inspidus,
hypopituitarism and optic chiasm compression (Figure 9).
There may be coexistent pineal masses,
and there may be subarachnoid spread.
Ectopic neurohypophysis can be caused by trauma or neoplastic processes,
which disrupt the transport of hormones from the hypothalamus to the neurohypophysis.
Congential ectopic neurohypophysis is associated with other midline abnormalties such as septo-optic dysplasia.
The expected posterior pituitary bright spot is frequently located in the suprasellar region (Figure 10).
This can lead to growth hormone deficiency.
In the extreme manifestation of this abnormality,
i.e.
pituitary stalk interruption syndrome,
there is also an absence or hypoplastic infundibular stalk and adenohypophysis [18].
Lymphocytic hypophysitis is an autoimmune disorder seen primarily in peripartum/postpartum females though it has also been reported in males and postmenopausal women. [19] On MRI,
there is intense enhancement of a pituitary mass extending along the infundibulum to the floor of the hypothalamus.
The posterior pituitary bright spot may be absent.
The adenohypophysis and/or the neurohypohysis can be affected. 1 Dynamic MRI studies have shown that blood supply to the neurohypophysis is often compromised.
Patients may present with visual field impairment and headache due to mass effect on adjacent structures.
As the pituitary parenchyma is destroyed by the inflammatory process,
partial or pan-hypopituitarism develops. However,
the condition is responsive to corticosteroid treatment and can spontaneously resolve.
Neurosarcoidosis is an inflammatory granulomatous process,
which has a predilection for the leptomeninges. [11]There can be involvement of the optic chiasm,
hypothalamus,
pituitary gland and infundibulum (Figure 11).
Suprasellar pathology
Pituitary abscesses can occur as sequelae of sepsis,
extension of sinusitis,
sphenoid osteomyelitis,
cavernous sinus thrombophlebitis and meningitis.
Infectious processes such as tuberculosis and fungal infection may be spread in a hematogenous route or by direct extension. [20] This can lead to brainstem infarction due to arteritis of the perforating vessels,
pseudo-aneurysm of traversing vessels or leptomeningeal dissemination.
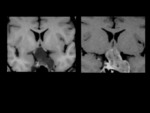
Postoperative sellar abscess is a potential complication of transphenoidal surgery for pituitary macroadenoma resection (Figure 12,
13).
Arachnoid cysts can occur in the suprasellar compartment and account for 1% of intracranial masses (Figure 14). [3]These smoothly marginated lesions follow CSF imaging characteristics on CT and MRI.
They may displace or compress the adjacent structures including the infundibulum,
pituitary gland and third ventricle.
Hamartoma of the tuber cinereum is a developmental heterotopia,
which presents in childhood as precocious puberty and seizures (i.e.
characteristic gelastic seizures).
Abnormalities of the corpus callosum and optic tracts can be seen in these patients.
These suprasellar masses are isodense on CT,
isotense on T1-weighted MRI,
and hyperintense and nonenhancing on T2-weighted MRI.
Other lesions involving the suprasellar cistern include epidermoid (Figure 15),
dermoid (Figure 16),
teratoma and lipoma.
Epidermoids can be differentiated from arachnoid cysts on fluid attenuated inversion recovery (FLAIR) and diffusion-weighted imaging as they tend to appear slightly hazy on the first sequence and bright on the latter.
Mucocele of the sphenoid sinuses can extend into the suprasellar cisterns.
Chiasmatic and hypothalamic gliomas are seen predominantly in the first decade of life and 20-50% of these patients have neurofibromatosis I. [3] These lesions may be difficult to distinguish from hypothalamic astrocytomas or gangliogliomas.
They demonstrate variable enhancement and appear T1 hypointense and T2 hyperintense; this signal intensity can extend along the optic tracts.
Vascular lesions such as aneurysms and cavernous carotid fistula and cavernous sinus thrombosis or thrombophlebitis can be seen in the cavernous sinus region.
Aneurysms can produce mass effect on the intracavernous cranial nerves.
Rupture of aneurysms of the cavernous segment of the internal carotid arteries can result in cavernous-carotid fistulas.
Paramedian internal carotid aneurysms can mimic an intrasellar aneurysm and cause mass effect on the pituitary gland.
Meningiomas in this region can narrow the adjacent internal carotid artery.
Schwannomas of the trigeminal nerve are more common than those originating from cranial nerves III,
IV and VI.
Osteocartilagenous tumors arising from the basicranium can extend into the cavernous sinus or suprasellar region (Figure 17).