TB is a common worldwide infection,
which has made a resurgence in recent years in non-endemic populations due to immigration from endemic areas and high prevalence of Human Immunodeficiency Virus (HIV) infection and multidrug – resistant (MDR) strains (1).
EPIDEMIOLOGY:
TB is a curable and preventable disease.
An early detection and correct treatment enable patients,
to quickly become non-infectious and eventually cured (2).
In 2015,
10.4 million people suffered TB and 1.8 million died from the disease (including 0.4 million among people with HIV).
Over 95% of TB deaths occur in low- and middle-income countries (3).
Six countries account almost 60 % of this total,
with India leading the count,
followed by Indonesia,
China,
Nigeria,
Pakistan and South Africa (3).
Tuberculosis incidence has fallen by an average of 1.5 % per year since 2000.
Moreover,
the high prevalence of tuberculosis,
in spite of the decrease in incidence last years places it as an important global health problem (3).
Among communicable diseases,
tuberculosis is the second leading cause of death worldwide after HIV.
However,
considering that is a preventable disease,
the death toll remains unacceptably high,
and efforts to combat the disease must be empowered (3) (4).
PATHOGENESIS:
TB is an airborne person to person spread disease,
caused by the bacterium Mycobacterium tuberculosis (M.
tuberculosis).
M.
tuberculosis,
M.
bovis,
M.
africanum and M.
microti conform the spectrum of M .
Tuberculosis complex.
M.
tuberculosis is a slightly curved bacillus that is a gram-negative,
strictly aerobic,
non-motile,
non-spore-forming,
rod-shaped bacterium (4).
There are various factors that determine the probability of transmission of M.
Tuberculosis depending on susceptibility,
infectiousness,
enviroment and exposure factors (2)(4)(5).
- Host suceptibility is highly related to the immune state.
- Infectiousness of the person with tuberculosis is directly related to: the amount of acid-fast bacilli in the sputum smear (is the main indicator of potential for transmission),
positive sputum culture,
lung parenchymal cavitation on imaging,
tuberculous laryngitis,
as well as high-volume and/or watery respiratory secretions,
or an inappropriate or inadequate treatment.
- Environmental factors as crowded living such as prisons,
living in an inner city,
homelessness or immigration from or travel to an endemic area,
helps the disease to spread more easily.
- Proximity,
frequency,
and duration of exposure.
The bacillum travel in airborne particles,
called droplet nuclei (1– 5 microns in diameter).
Transmission occurs when a person inhales droplet nuclei produced when persons with pulmonary or laryngeal TB cough,
sneeze,
or speak.
The distribution of inhaled droplet nuclei is determined by the ventilatory pattern and volumes of the various lobes of the lungs with the site of implantation preferentially in the middle and lower lung zones.
Transmission of disease occurs as a result of contact to a source case in more than 80% of the cases,
typically as a result of exposure to sputum smear-positive case (4)(5).
Once lodged in the alveolus,
extracellular bacilli are ingested by alveolar macrophages.
In more than a half of cases,
the bacilli are eliminated by the alveolar macrophages,
as they are capable of controlling the replication and the patient only undergoes to an exposure to the bacillum (4)(5).
But if the alveolar macrophage cannot destroy or inhibit the replication,
bacilli multiply within its intracellular environment and spread into other alveolar macrophages.
It is on this point of rapid growth,
when lymphatic dissemination occurs achieving regional lymph nodes,
leading to tuberculous primoinfection.
Secondly the tubercle bacilli could spread through the bloodstream to more distant tissues and organs (2)(4)(5).
Likewise,
in most patients,
the logarithmic phase of bacillary growth is arrested with the development of cell-mediated immunity (4). Alveolar macrophages interact with T- lymphocytes,
which directly implies an activation of the T-cell mediated immunity.
CD 4 T-lymphocytes segregate citokynes such as interferon-gamma that activates macrophages and other immune cells,
so that they kill or encapsulate most of the bacilli,
leading to the formation of a granuloma where the bacillus is enclosed and latent (2).
These patients have a latent TB infection,
and suppose about one third of the whole world population.
They are infected but they cannot spread the infection to other people.
Usually it has an asymptomatic course and is radiologically undetected (2)(4).
Latent TB infection may be diagnosed by using the tuberculin skin test (TST) or an interferon-gamma release assay (IGRA),
which has a higher diagnostic accurancy than TST (2).
The vast majority (90%) of latent TB cases,
will stay in latency stage.
In approximately 5% of infected individuals,
immunity is inadequate,
and clinically active disease will develop within 1 year of infection,
a condition known as progressive primary infection (2)(5).
Other risk factors for progression to active disease include: diabetes mellitus,
renal failure,
coexistent malignancies,
malnutrition or immunosuppressive therapies (2)(4).
In another 5% of the infected population,
endogenous reactivation of latent infection occurs remote from time of initial infection,
this concept refers to post-primary TB (2)(5).
Tuberculous disease can occur in pulmonary and extrapulmonary sites.
TB is usually confined to respiratory system (pulmonary TB) (1).
If there is haematogenous dissemination (miliary TB) or extension to the pleura,
is considered extrapulmonary tuberculosis.
The clinical presentation of the pulmonary TB can be undiagnosed since it has an insidious course.
The symptoms of active tuberculosis are: cough,
pleuritic chest pain,
haemoptysis,
fever,
weight loss,
night sweats etc (2)(5).
Classical knowledge divides pulmonary tuberculosis into primary,
common in childhood,
and post-primary,
usually presenting in adulthood,
which were considered two different forms of the disease on the basis of time of exposure (1).
Patients who develop disease after initial exposure are considered to have primary TB,
whereas patients who develop disease as a result of reactivation of a previous focus are considered to have reactivation TB (2)(4)(6).
According to this,
upper lobe cavitary disease was believed to be the hallmark of reactivation of latent infection,
whereas lower-middle lobe affectation,
lymphadenopathy,
and effusion were believed to indicate a recent infection.
Sometimes it is difficult to differentiate the pattern,
since clinically and radiologically their features can overlap (1)(2)(6).
This paradigm has been challenged,
on the basis of DNA fingerprinting and genotyping of M.
tuberculosis (analysis about restriction fragment length polymorphism (RFLP) (2)(4)(6).
To date,
there have been publications such as a molecular epidemiology study published in 2005 by Geng et al.
(7),
whereby a correlation between clinical and radiographic features of primary and post-primary TB was made,
and which proved that radiographic features of pulmonary tuberculosis are more related to immune stage,
rather than time of acquisition of infection.
On the basis of the above,
time from acquisition of infection to the development of disease does not reliably predict the radiographic appearance of tuberculosis,
and therefore,
there is no imaging difference between primary and post-primary TB.
Consequently,
the most important and dependent factor to determine the radiological appearance is the immune state (2)(4).
We review: Primary,
post-primary and miliary presentation patterns of pulmonary tuberculosis,
response to treatment evaluation,
as well as,
the specific findings in specific scenarios as: HIV-TB co-infection and multidrug resistant tuberculosis (MDR-TB).
PRIMARY PATTERN:
This type of radiological pattern is more common in children and immunocompromised patients.
Immunocompromised patients have a higher prevalence of TB than dose the general population,
and present more frequently extrapulmonar involvement (1).
The prevalence of this primary pattern in adults is increasing (1)(4).
Normal radiographic findings may be seen in up to 15% of patients with proven tuberculosis (1)(4).
RADIOLOGICAL FINDINGS (Chest radiograph and CT):
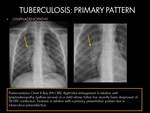
- Hilar or mediastinal (essentially paratracheal) lymphadenopathy: Usually unilateral,
right-sided and ipsilateral to parenchymal illness.
Radiographic evidence of lymphadenopathy is seen in up to 96% of children and 43% of adults.
Bilateral in about one-third of cases (1)(2)(4).
Fig. 1
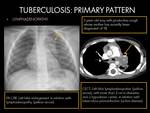
- CT is better at detecting nodal disease.
When active disease,
the nodes have central low attenuation and peripheral rim enhancement,
in relation with caseation or liquefaction necrosis and and peripheric granulation tissue with inflammatory hypervascularity.
This pattern of lymphadenopathy is very characteristic of tuberculosis,
but it is important to evaluate the features considering the clinical setting,
without forgetting similar findings may also be caused by atypical mycobacterial,
metastasis,
lymphoma or other infectious diseases (1)(2)(4).
Fig. 2
- Ghon´s primary complex regards to the primary site of infection in the lungs (2).
- In approximately two-thirds of cases,
the parenchymal focus resolves without imaging sequelae,
although this resolution can take up to 2 years In the remaining cases,
a radiologic scar persists that can calcify in up to 15% of cases (1)(4).
- The combination of calcified hilar nodes and a Ghon focus is called a Ranke complex and is suggestive of previous tuberculosis (1)(2)(4).
Fig. 23
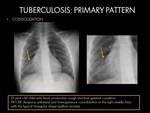
- Parenchymal consolidation area,
usually unilateral and homogeneous.
More frequent in the middle/lower lobes,
with predilection for right lung.
It could be segmental,
lobar or multifocal (1)(2)(4).
Fig. 3
- Parenchymatous involvement could also appear as patchy,
linear,
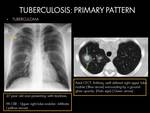
nodular or masslike parenchymal opacities (tuberculomas) (2)(4).
- Tuberculoma appears as a round-oval parenchymal granuloma with central caseous necrosis,
and a wall lined by granulomatous inflammatory tissue or encapsulated by connective tissue. It can be solitary or multiple,
range in diameter from 0.5 to 4.0 cm or greater.
Typically,
they have smooth or sharply defined margins.
Satellite nodules around the tuberculoma may be present in as many as 80% of cases.
Calcification is found in 20%-30% of tuberculomas and is usually nodular and diffuse (2).
Fig. 4
- Obstructive atelectasis due to bronchial compression by lymphadenopathy is seen mainly in children and it can also occur in adults with endoluminal bronchial occupation,
being able to simulate a neoplastic disease (4).
- Unilateral pleural effusion,
is not as frequent as anterior features described above.
If present,
its usually ipsilateral to pulmonary abnormalities and complications (empyema,
fistulization,
bone erosion) are rare (1)(2).
It has a self-limited course that resolved within several weeks.
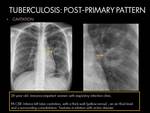
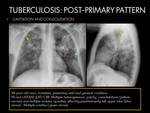
POST-PRIMARY PATTERN:
This type of radiological pattern is more common in immunocompetent adults.
RADIOLOGICAL FINDINGS (Chest radiograph and CT):
- Heterogeneous,
poorly defined consolidations or parenchymal opacities affecting multiple segments (predominantly the apical and posterior segments of the upper lobes and,
with a lower frequency,
the superior segment of the lower lobe),
with a bilateral involvement in in one-third to two-thirds of the disease (1)(4); In the early stages,
an ill-defined of density increased area often associated with nodular and/or linear component is observed (5).
Fig. 6
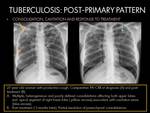
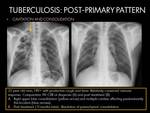
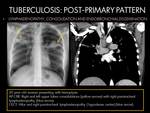
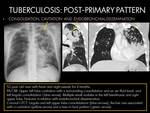
- Parenchymal consolidations are often associated with cavitation,
which is the hallmark of this pattern (1).
Cavities may be single or multiple,
thick or thin walled,
and it may contain an air-fluid level.
The features of active disease are: Wall thickening,
surrounding consolidation or occupation with soft-tissue material,
feature that could be in relation with development of an aspergilloma (4).
Fig. 5 Fig. 7 Fig. 8 Fig. 9
- Tuberculomas (described above),
may be also present in post-primary pattern (2)(4).
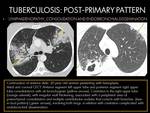
- Endobronchial spread occurs when establishing communication between an area of caseous necrosis (parenchymal or lymphatic node) and the bronchial tree (4).
Fig. 11 Fig. 12
- Endobronchial spread (CXR findings): Multiple,
ill-defined,
small nodules distributed in a segmental or lobar close or distant from the site of cavity formation,
affecting predominantly the lower lobes (4).
- Endobronchial spread (High-resolution thin-section CT): Centrilobular nodules (measuring from 2 to 4 mm in diameter) and linear branching opacities,
giving rise to a “tree-in-bud pattern”,
due to plugging of the distal airways and adjacent acini by caseous necrosis and granulomatous inflammatory products.
It is a good indicator of active disease (2)(4).
- Hilar and mediastinal lymphadenopathy are uncommon manifestations (2)(4). If present,
they are usually involving paratracheal right stage or mediastinum (4).
CT is better at detecting nodal disease.
(As explained above).
Fig. 10
- Pleural effusion: Typically small,
loculated and stable in size for years.
Usually associated with parenchymal disease.
The pleura may become thickened,
which can result in a tuberculous empyema and an associated risk of developing a bronchopleural fistula (1).
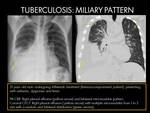
MILIARY PATTERN:
Miliary tuberculosis is secondary to haematogenous dissemination from a pulmonary or extrapulmonary focus spreading to various distant organs,
including the lungs (4).
It results in extended micronodular pattern,
which is commonly seen in the elderly,
infants,
and immunocompromised patients (1)(4).
Initially,
chest radiographs may be normal in 25% to 40% of cases (4). It is usually detectable around four weeks following the onset of symptoms,
and CXR is usually normal at the onset of symptoms. When they resolve (usually after 2-6 months of treatment) there is no residual scarring or calcification (1)(4).
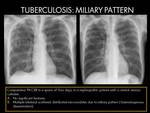
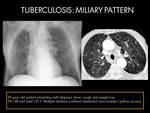
Fig. 13 Fig. 14 Fig. 15
- CXR findings: Multiple bilateral scattered distributed micronodules,
which may coalesce to form focal or diffuse consolidation (4).
- CT findings: High-resolution CT is the modality of choice because it has high sensitivity.
CT can demonstrate miliary disease before it becomes radiographically apparent (4).
- Multiple non-calcified nodules,
measuring from 1 to 3 mm in diameter,
with a random distribution throughout both lungs.
Often associated with intralobular and interlobular septal thickening (2)(4).
- These patients could associate other radiological findings as: Consolidation (predominantly upper lobes),
cavitation,
pleural fluid,
and necrotic lymphadenopathy (4).
TUBERCULOSIS AND HUMAN IMMUNODEFICIENCY VIRUS COINFECTION:
HIV co-infection is the most important risk factor for progression to active disease in adults,
and the relation between HIV and TB has augmented the deadly potential of each disease (4).
TB is a major cause of death among people living with HIV infection or AIDS (2).
The high rate of active TB in HIV-TB co-infected patients,
has been linked to the loss of gamma-interferon,
an important cytokine produced by CD4 T cells,
depleted by HIV (6).
The imaging pattern of tuberculosis in HIV-positive patients correlated with the stage of HIV infection.
In general,
patients with lower CD4 T-lymphocyte counts,
tends to present as a primary pattern,
showing a higher prevalence of lymphadenopathy,
in addition to a higher prevalence of miliary TB,
extrapulmonary involvement and atypical manifestations.
Whereas patients with higher CD4 T-lymphocyte counts tends to present as a post-primary pattern (2)(4)(5)(6).
Fig. 8
At severe levels of immunosuppression,
10% to 20% of co-infected persons have normal radiographs.
But lung and mediastinal involvement is usually demonstrated on CT (4).
Moreover,
in general,
immunocompromissed patients are more likely to develop atypical manifestations,
as well as miliar tuberculosis and extrapulmonary involvement (4).
Highly active antiretroviral therapy (HAART) and Immune reconstitution inflammatory syndrome:
When highly active antiretroviral therapy is introduced in a VIH-TB co-infected patient,
there is a paradoxic worsening of the patient’s clinical status related to an increase in the CD4 counts and a decrease in the viral loads (2)(4). There is a restoration of cellular inflammatory response,
but it is not secondary to a progression of mycobacterial infection (4).
- CXR and CT findings: (4).
- Progression of lymphadenopathy affecting intrathoracic or cervical areas in approximately 70% of patients.
Mediastinal lymph nodes are typically affected.
- Lymph nodes exhibit central low attenuation and peripheral rim enhancement.
- Progression of lung disease and pleural effusion.
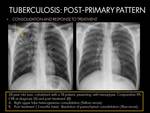
RESPONSE TO TREATMENT:
Evaluation of the response of pulmonary TB to antibiotic treatment is best assessed by sputum examinations in patients with positive bacteriology.
But when there is a negative pre-treatment sputum,
radiographic and clinical evaluation become the major indicators of response to therapy (5).
A baseline radiograph at the completion of treatment may be useful for future comparison purposes (5).
The regression of imaging abnormalities following adequate treatment is often a slow process,
and it is frequent an initial deterioration before improvement,
showing extension of parenchymal involvement and development or enlargement of lymph nodes.
- CXR and CT findings: (4) (5).
- Resolution of parenchymal abnormalities require from 6 months to 2 years on CXR and up to 15 months on CT.
Fig. 6
- Cavities may disappear after antituberculous treatment,
or it may remain as an air-filled cavity with a thin wall,
emphysematous changes or scarring.
Fig. 8
- In general,
residual areas of scarring,
focal nodularity,
and volume loss are seen as parenchymal sequels after treatment.
- Lymphadenopathy usually enlarge at the start of treatment,
and may take a long time to show signs of resolution.
The volume of lymphadenopathy is a poor indicator of response to treatment.
- After an effective therapy tuberculous pleural effusion tends to disappear.
Whereas,
persistence or increase in size of an effusion suggests either resistance to therapy or a secondary cause.
If radiographic features remain unchanged in a range from 4 to 6 months,
it generally indicates a inactive disease.
However,
because even long-term stability of radiographic findings may occasionally be associated with culture-positive disease,
such findings should be described as ‘‘radio- graphically stable’’ rather than ‘‘inactive’’ (5).
MULTIDRUG RESISTANT TB (MDR-TB):
MDR-TB is defined as a strain of M.
tuberculosis resistant to at least isoniazid and rifampin.
Extensively drug-resistant (XDR) TB is a dreaded infection caused by a strain of M.
tuberculosis that is resistant to any type of fluoroquinolone and at least 1 of the 3 following injectable drugs: amikacin,
capreomycin,
or kanamycin in addition to isoniazid and rifampin (4).
The emergence of MDR strains of M.tuberculosis in recent years has complicated the management and control of transmission of active cases.
Multidrug-resistant strains most commonly develop during the course of tuberculosis treatment,
most frequently as a result of inappropriate or inadequate adherence to treatment (4) (5).
- CXR and CT findings: There are two forms of acquisition of drug-resistance with different radiologic features.
- Patients with primary drug resistance,
who develop MDR-TB without a history of anti-TB chemotherapy or a therapy history of less than 1 month,
were found to present with noncavitary consolidation,
pleural effusion,
and therefore,
as a primary pattern.
On the other hand,
patients with secondary drug resistance,
who acquired MDR-TB with a chemotherapy history of longer than 1 month often show cavitary consolidations and,
in general as a post-primary pattern (2)(4).
- Chronicity findings such as bronchiectasis,
emphysema,
bronchovascular distorsions and calcified granulomas can also be encountered (2).
In adults,
failure of improvement in the extent of lung parenchymal involvement and a positive sputum culture after 4 months of multidrug therapy,
indicate treatment faiure (4).
IMAGING TUBERCULOUS ASSESMENT:
A complete evaluation for TB must include: clinical history,
physical examination,
CXR,
TST,
IGRA and microbiological smears and cultures (2).
Chest radiograph remains the basic imaging tool for the screening,
diagnosis and follow-up of pulmonary TB.
The aim of screening chest radiographs is to identify persons with active TB,
bearing in mind that it counts with a low specificity,
and radiographs may be normal or show non-specific findings in patients with active disease.
The distinction between active and inactive disease can only be reliably made on the basis of temporal evolution (2)(5).
CT is more sensitive on diagnosis,
improves lesion detection (especially in patients in whom the chest radiograph is normal or inconclusive),
improve characterization and extension of the disease,
determination of disease activity,
treatment response evaluation,
and detection of sequels and complications (2).
CT is more useful than chest radiograph in determining disease activity,
based on the pattern of parenchymal abnormalities,
the presence of cavitation or evidence of endobronchial spread.
CT is also helpful in the evaluation and management of MDR-TB and complications,
such as empyema and bronchopleural fistula,
and may show pleural disease that is not evident on chest radiography (2).
COMPLICATIONS AND SEQUELS
Sequelae and complications can be categorized as parenchymal,
airway,
pleural,
mediastinal,
chest wall and vascular lesions (8).
A.
Parenchymal lesions: Cicatrization,
end-stage destruction of the lung,
aspergilloma and bronchogenic carcinoma.
B.
Airway lesions: Bronchiectasis,
tracheobronchial stenosis,
broncholithiasis.
C.
Pleural lesions: Chronic empyema,
fibrothorax,
bronchopleural fistula and pneumothorax.
D.
Chest Wall lesions: Empiema necessitatis.
E.
Mediastinal lesions: Lymph node calcification and extranodal extension,
tuberculous pericarditis,
myocardial tuberculosis,
esophagomediastinal or esophagobronchial fistula and fibrosing mediastinitis.
F.
Vascular lesions: Pulmonary or bronchial arteritis and thrombosis,
bronchial artery dilatation and Rasmussen’s pseudoaneurism.
A.
PARENCHYMAL LESIONS:
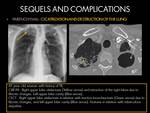
A.1: CICATRIZATION AND DESTRUCTION OF THE LUNG:
Local control and resolution of pulmonary TB is usually accompanied by some destruction of involved tissues.
Up to 40% of patients with post-primary TB have a marked fibrotic response (8).
Complete destruction of a whole lung or a major part of a lung is not uncommon in the end stages of tuberculosis with a severe fibrotic response.
Such damage results from a combination of parenchymal and air- way involvement.
It may result from a progressive primary infection or from post-primary TB with a prolonged process of cavitation,
spread to new areas,
and subsequent fibrosis (8).
- Atelectasis of the upper lobe,
retraction of the hilum,
compensatory lower lobe hyperinflation,
and mediastinal shift toward the fibrotic lung.
- Another fibrotic findings could be present,
consisting of parenchymal bands,
fibrotic nodules,
cavities and traction bronchiectasis.
A.2: ASPERGILLOMA:
It refers to Aspergillus infection of a pre-existing pulmonary cavity (from previous TB) or ectatic bronchus,
leading to the emergence of a conglomeration of fungal hyphae admixed with mucus and cellular debris.
The upper lobe is the most common location (70%) (4)(8).
Although aspergilloma may exist for years without symptoms,
hemoptysis and productive cough are the most common clinical manifestations (4)(8).
Surgical resection is indicated for patients with severe life-threatening hemoptysis,
and selective bronchial artery embolization can be performed in those with poor lung function.
The cavitary lesions tend to be complex (75%) with gross pulmonary disease in the surrounding parenchyma,
more often than simple (25%),
in which the lesion consists of an isolated thin-walled cyst (4).
Fig. 17
- CXR findings:Intracavitary,
spherical or ovoid solid nodule with surrounding gas (4)(8).
- Thickening of the walls of tuberculous cavities or of the adjacent pleura is an early sign of infection.
- Heterogeneous intracavitary mass.
The mass may fill completely or partially the cavity.
It may be surrounded by a crescentic air shadow inside the cavity (air-crescent sign).
- Calcification occurs in some cases.
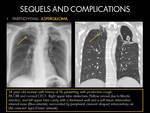
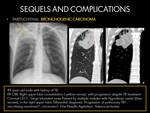
A.3: BRONCHOGENIC CARCINOMA:
Bronchogenic carcinoma can mimic or coexist with pulmonary TB.
The diagnosis of lung cancer is delayed mainly because of masking by a tuberculous lesion,
since it can be misinterpreted as progression of TB.
Moreover,
TB may promote the development of bronchogenic carcinoma by local mechanisms (scar cancer) (8)(9).
Fig. 18
- CXR findings: There are several features which,
if present,
increase the suspicion of coexisting lung carcinoma in patients with pulmonary TB (9).
- Segmental or lobar atelectasis.
- Unilateral hilar enlargement.
- Homogeneous infiltration with no air bronchogram rather than a mottled appearance with linear streaking.
- Progression of pulmonary infiltrate while the patient is on anti-tuberculous drugs.
- Asymmetrical pleural density at the apex or costophrenic angle while the patient is receiving anti-tuberculous medication,
with little improvement of symptoms despite antituberculous.
- Single pulmonary nodule with a diameter greater than 3 cm,
and an irregular nodule wall and contour.
- CT findings:CT can reduce image overlap,
since it optimizes the recognition of tumors masked by tuberculous lesions (9).
- These findings are similar to those which present in patients without TB: Large,
lobulated mass with mediastinal lymphadenopathy.
B.
AIRWAY LESIONS:
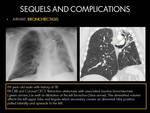
B.1: BRONCHIECTASIS:
Bronchiectasis refers to abnormal dilatation of the bronchial tree.
It may develop as a result of tuberculous involvement of the bronchial wall and subsequent destruction and fibrosis of the lung parenchyma,
causing a loss of volume,
giving rise to traction bronchiectasis (8).
Bronchiectasis in post-primary tuberculosis can be,
in a lesser extent,
a result of cicatricial bronchostenosis after local infection (8).
Typical localization of TB bronquiectasis is in the apical and posterior segments of the upper lobe (8).
Fig. 19
- CXR findings: There are non-specific findings:
- Tram-track opacities: thickened non-tapering walls of bronchi.
- Increase in bronchovascular markings.
Pulmonary vasculature appears ill-defined,
in relation with peribronchovascular fibrosis.
- CT is usefull to confirm the diagnosis and to quantify bronchiectasis,
which are seen as an increased of bronchoarterial ratio which is greater than 1,5 mm.
B.2: TRACHEOBRONCHIAL STENOSIS:
The most common cause of inflammatory stricture of the bronchus is TB. The incidence of this complication has decreased,
but still affects patients with advanced disease (2)(4).
Endobronchial TB occurs in approximately 2%– 4% of patients with pulmonary TB.
The airway is occupied by granulation tissue,
which cross the mucosa and submucosa and finally it develops a fibrotic reaction causing tracheobronchial narrowing (4)(8).
It is not usually detectable on CXR.
There are two stages of the disease: Active or fibrotic.
CT findings:(2)(4)(8).
- Active stage: Irregular bronchus wall thickening causing luminal narrowing,
associated with enhancement and enlarged adjacent mediastinal nodes.
- Fibrotic stage: Concentric narrowing of the lumen,
with uniform thickening of the wall,
with a lesser degree of enhancement,
and involvement of a long bronchial segment.
Distal trachea as well as the proximal left main bronchus are most commonly affected on this stage.
B.3: BRONCHOLITHIASIS:
Broncholithiasis is an uncommon complication of pulmonary TB and is defined as the presence of calcified material within tracheobronchial tree.
The origin of this airway complication is due to an erosion into the bronchial wall by a calcified tuberculous lymph node,
with the subsequent bronchus obstruction (8).
Presenting symptoms may include cough,
hemoptysis,
wheezing,
or recurrent pneumonia (5)(8).
- CXR findings: Change in the position or disappearance of a calcified focus on serial radiographs or development of airway obstruction,
resulting in atelectasis,
mucoid impaction,
or expiratory air trapping (8).
- CT findings: Calcified lymph node (endobronchial or peribronchial) associated with findings of bronchial obstruction (8).
C.
PLEURAL LESIONS:
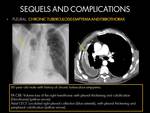
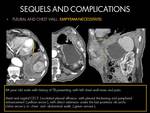
C.1: CHRONIC TUBERCULOUS EMPYEMA AND FIBROTHORAX:
Pleural infection is usually caused by rupture of a subpleural caseous focus into the pleural space; less commonly,
it is caused by hematogenous or adjacent infected lymph nodes dissemination (8).
Tuberculous pleurisy progresses to become chronic tuberculous empyema,
which may be defined as persistent focal fluid collection due to M.tuberculosis infection,
which can develop into fibrothorax.
Fibrothorax is defined as fibrosis within the pleural space,
and occurs secondary to the inflammatory response which finally gives rise to calcification (8).
Fig. 20
- Chronic tuberculous empyema: Loculated pleural fluid collection.
- Fibrothorax: Smooth pleural thickening that may be calcified.
It is usually associated with marked volume loss in the affected hemithorax.
- Chronic tuberculous empyema: Encapsulated and infected fluid collection associated with pleural thickening,
with peripheral enhancement and/or calcification,
which can be associated with extrapleural fat proliferation.
It is ussually associated with volume loss in the affected hemithorax.
- Fibrothorax with diffuse pleural thickening but without effusion on CT scans suggests inactivity.
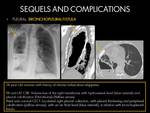
C.2: BRONCHOPLEURAL FISTULA:
Bronchopleural fistula consists on an abnormal communication between the pleural space and the bronchial tree,
that being in a tuberculous clinical setting,
is usually produced as a result from a trauma or surgical procedure,
but can also occur due to an open pathway between bronchus and pleura of the necrotizing lung infection.
Is a rare complication with a high morbidity and mortality and has a variable clinical and imaging manifestation depending on whether it is an acute,
subacute,
or chronic form as well as the underlying condition (4)(8).
The diagnosis is based on an increasing amount of sputum production,
air in the pleural space,
a changing air-fluid level,
and contralateral spread of pneumonic infiltration (8).
The abnormal communication between the pleural space and airways increases the risk for aspiration pneumonia and adult respiratory distress syndrome,
which are common causes of death in affected patients (4).
- CXR and CT findings: (4)(8).
Fig. 21
- Tension pneumothorax,
hydropneumothorax,
lung abscess,
cavitary lung lesion,
peripheral bronchiectasis and subcutaneous emphysema.
- CT with multiplanar reconstruction is the imaging modality of choice to demonstrate the location,
number,
and size of the abnormal communication between the bronchial tree and the pleural space.
C.3 PNEUMOTHORAX: (8).
Pneumothorax secondary to tuberculosis is secondary to pleural caseous infiltrates that undergo liquefaction,
resulting in pleural necrosis and rupture.
It occurs in approximately 5% of patients with post-primary tuberculosis,
usually with extense cavitations.
If any apical abnormality is seen after re-expansion of a spontaneous pneumothorax,
active tuberculosis should be considered.
In some cases,
the lung will seal and reexpand over a long period in response to chemotherapy.
D.
CHEST WALL LESIONS:
Chest wall TB is an uncommon manifestation of the disease,
which is caused by direct extension from a pleural or parenchymal focus,
and less frequently,
by haematogenous spread from a distant focus (4).
This entails an involvement of the sternum,
sternoclavicular joint,
and/or ribs leading to osseous destruction and leading to located abscess formation with chest wall soft tissue affection giving rise to myositis and cellulitis.
It may eventually rupture through the skin. It can also spread to involve other sites such as the esophageal,
breast,
retroperitoneal, peritoneal,
pericardial and paravertebral regions (4)(8).
Empyema necessitatis is a rare complication of pleural infection in which spontaneous discharge of empyema from the pleural space through the parietal pleura into the soft tissues of the chest wall forms a subcutaneous abscess (8).
It may either occur due the virulence of the organism or may be facilitated by previous thoracic surgery or trauma.
- CXR findings: There are nonspecific features.
CXR can be normal,
or show an increase of and at times can even be normal soft tissue density adjacent to the chest wall.
- Bone or costal cartilage destruction and soft- tissue masses that may demonstrate calcification or rim enhancement with or without evidence of underlying lung or pleural disease.
- Chest wall fluid collection with peripheral enhancement,
thick and occasionally calcified pleura as sequelae of chronic infection,
and rib thickening from chronic periosteal reaction due to adjacent loculated pleural fluid collection.
- The most common location is between the second and sixth intercostal spaces,
but transdiaphragmatic extension to the abdomen or extension into the mediastinum may also occur.
E.
MEDIASTINAL LESIONS:
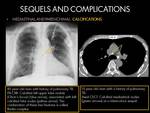
E.1 LYMPH NODE CALCIFICATIONS AND EXTRANODAL EXTENSION:
Tuberculous mediastinal lymphadenitis is a frequent manifestation of primary pulmonary tuberculosis (8).
Pubertal and young adult women,
the elderly,
and patients with acquired immunodefi- ciency syndrome are the most commonly affected among adult patients (8).
With treatment,
nodes change in appearance,
first becoming homogeneous (which can be demonstrate in CT) and finally disappearing or resulting in a residual mass composed of fibrotic tissue and calcifications (which can be demonstrate in CXR and CT) without low-attenuation areas in CT (8).
Fig. 23
Extranodal extension may occur into adjacent structures such as the bronchus,
pericardium,
and esophagus (8).
E.2 FIBROSING MEDIASTINITIS:
Fibrosing mediastinitis is an uncommon fibrotic and chronic inflammatory process. It has a silent clinic course with mild symptoms,
including cough and low-grade fever,
and symptoms due to compression of the superior vena cava,
esophagus,
and tracheobronchial tree (8).
The mediastinal granulomatous lymph nodes coalesce,
and the development of multiple tuberculous granulomas creates both reactive fibrous changes and acute inflammatory changes in the mediastinum.
The granulomas evolve into fibrosing mediastinitis when reactive changes predominate (8).
- CXR findings: Mediastinal widening or a localized mass (8).
- CT findings: Mediastinal calcific mass which obliterates different mediastinal structures (SVC,
airway and pulmonary vessels).
It may result in superior vena cava obstruction ,tracheo-bronchial narrowing,
obstructive pneumonia or atelectasis.
pulmonary vessel encasement or pulmonary infarction (8).
E.3 ESOPHAGOMEDIASTINAL OR ESOPHAGOBRONCHIAL FISTULA:
The most common origin of esophagomediastinal or esophagobronchial fististula is the erosion of the esophageal or bronchial wall by tuberculous lymph nodes,
giving rise to esophagomediastinal or esophagobronchial fistula (8).
Esophageal involvement results in strictures due to granulomatous inflammatory tissue in active disease and scar tissue after healing,
tracheobronchial or mediastinal fistulas,
and traction diverticula (8).
Presenting symptoms may include fever,
cough,
weight loss,
dysphagia,
chest discomfort,
or back pain (8).
It is not usually detectable on CXR.
- CT findings: Localized fluid-gas collection within the mediastinum,
with thickening wall esophagi,
pneumomediastino could be present.
- Sometimes the fistulous tract could be evident at CT.
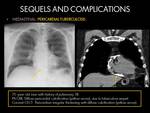
E.4 TUBERCULOUS PERICARDITIS:
Tuberculous pericarditis,
is the most common cardiovascular complication of TB,
which is found in 1% to 2% patients with pulmonary infection. Myocardial involvement by M tuberculosis is less common than pericardial disease (4)(8).
It may result from lymphatic spread from mediastinal lymph nodes or by hematogenous dissemination to the pericardium (4)(8).
Tuberculous pericarditis may present clinically as pericardial effusion,
constrictive pericarditis,
or as a combination,
with effusive-constrictive pericarditis.
In patients with active disease,
pericardial effusion should be suspected if CXR reveals an enlarged cardiac silhouette.
Tuberculous pericardial effusion,
may manifest with cardiac tamponade (10%) (4)(8).
CT findings: (1)(4)(8).
- Pericardium irregular thickening (> 3 mm) and frequently with associated mediastinal lymphadenopathy.
- Bilateral pleural effusion may be present,
tipically associated with mediastinal lymphadenopathy.
- Increase on the IVC diameter (> 3 cm).
- Less than 20% pericardial effusion.
- Pericardial calcification,
due to chronic changes.
Fig. 24
- Constrictive pericarditis: The combination of pericardial inflammation,
fibrosis,
thickening,
and calcification contributes to diastolic dysfunction with lower diastolic volume and stroke volume.
In addition to the abnormal appearance of the pericardium,
here we can also find compression of ventricles,
left desviation of the interventricular septum,
atrial enlargement and dilated inferior vena cava,
superior vena cava,
hepatic veins,
and coronary sinus.
- Effusive-constrictive pericarditis: in addition to the constrictive changes,
a variable amount of concomitant pericardial effusion coexists that may be associated with myocardial thinning.
F.
VASCULAR LESIONS:
Pulmonary arteries and veins in an area of active tuberculous infection may demonstrate vasculitis and thrombosis.
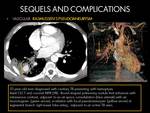
Rasmussen aneurysm is a pulmonary artery pseudoaneurysm produced by erosion into the artery wall by the adjacent TB focus,
which causes a thinning and subsequent rupture (4)(8).
It usually manifests with haemoptysis,
which may be life-threatening when it is massive (4).
- CXR findings: This entity is not recognizable at CXR.
- Round shaped structures localized near the tuberculous cavity.
- It usually presents as small (< 1 cm),
round-shaped,
single lesions,
with contrast-enhancing surrounded by a parenchymal consolidation,
or located adjacent to the wall of a lung cavity; typically in the upper lobes or superior segment of the lower lobes.
- Signs of pulmonary arterial origin of hemoptysis: Focal contrast-enhancing dilation or outpouching arising from the pulmonary artery (75%),
or the presence of a pulmonary artery in the inner wall of a pulmonary cavity (25%).