US findings:
- Simple cyst: Anechoic,
well- marginated lesion,
with imperceptible wall,
round-ovoid and with posterior acoustic enhancement.
- Hemorrhagic cyst: Cystic lesion with internal echoes (clot).
Thick calcified wall and/or multiloculated appearance due to chronic changes.
- Infected cyst: Cystic lesion with thick wall and scattered internal echoes.
It may contain debris-fluid level.
- Neoplastic cystic lesion: Cystic lesion with mural nodules and solid components with blood-flow in color Doppler ultrasound.
CT findings:
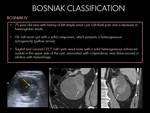
In order to classify a renal cystic lesion,
applying the Bosniak´s system,
we have to analyze different features of the lesions in CT examinations: Attenuation,
contrast material enhancement (perceived or measurable),
and the presence of calcifications and septations.
There are five categories: (I,
II,
IIF,
III,
and IV) (1) (2).
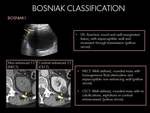
Bosniak I cysts: These are simple cysts,
and are considered to be benign,
with a likelihood of malignancy of 0%.
No further evaluation is needed.
Radiological features: Fig. 1
- Well-defined,
rounded-ovoid masses.
- Non-enhanced CT (NECT): Homogeneous fluid attenuation (0–20 HU).
- Contrast enhanced-CT (CECT): Thin or imperceptible non-enhancing wall.
Do not demonstrate contrast enhancement.
- Do not contain calcifications or septations.
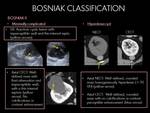
Bosniak II cysts: These are considered to be benign,
with a likelihood of malignancy of 0%.
No further evaluation is needed.
Radiological features: Fig. 2
*Minimally complicated cysts:
- NECT: Homogeneous fluid attenuation (0–20 HU).
- CECT: Thin or imperceptible non-enhancing wall,
1-3 thin (< 1 mm) septations with not measurable enhancement.
Do not demonstrate contrast enhancement. Fine calcification or a short segment of thickened calcification in the cyst`s wall or septa.
*Hyperdense cysts:
- Well-defined cystic mass.
- NECT: Homogeneous hiperdense (> 20 HU).
If they demonstrate more than 70 UH attenuation,
the cysts are considered benign.
If the attenuation is between 20 and 70 UH,
this is the range of possible solid renal neoplasm,
and contrast enhancement evaluation must be done,
in order to rule out. *Alternatively,
ultrasound or MR imaging can be used if patient cannot tolerate contrast administration,
due to allergy or renal chronic disease.
- CECT: Do not demonstrate contrast enhancement.
- Not completely intrarenal (More than one-fourth of the cyst wall is seen outside de renal contour).
- Usually ≤ 3 cm in diameter.
- Do not contain calcifications or septations.
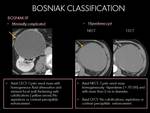
Bosniak IIF cysts: Requires CT or MR imaging follow-up to prove their benignity by showing stability over time.
It has a likelihood of malignancy of 10 to 12%.
Radiological features: Fig. 3
The difference between Bosniak II and IIF include: Cysts containing more than a few airline thin septa,
minimal enhancement of an airline thin septa or wall,
minimal thickening of septa or wall or containing nodular and thick calcification,
without enhancement after contrast administration.
IIF stands for follow-up: The length of follow-up needed for this category has not yet been clearly determined.
We can subdivide this category into:
- Minimal category IIF lesions with CT findings close to category II (abovementioned as category II minimally complicated).
One or 2 years of follow-up is recommended (3).
- Complex category IIF lesions with CT findings close to category III in complexity.
Three or 4 years (even longer),
of follow-up is recommended (3).
The introduction of the category IIF in this group of complicated cysts has helped to avoid unnecessary surgery,
and it has also decreased the percentage of benign category III masses being surgically removed (3).
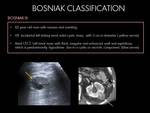
Bosniak III cysts: Complex cysts,
with a likelihood of malignancy from 50 to 60%.
Require surgery (Biopsy remains controversial).
Radiological features: Fig. 4
- NECT: Homogeneous fluid attenuation (0–20 HU).
- CECT: Thick irregular walls and/or septa with measurable enhancement (> 20 UH).
Multiple thin (< 1 mm) septations.
Do not demonstrate nodular enhancement.Thick nodular calcifications.
There is a wide range of entities that involve the Bosniak III differential diagnosis,
which includes both benign and malignant conditions: Mixed epithelial and stromal tumor (MEST),
benign multiloculated cyst,
hemorrhagic cyst,
chronic infected cysts,
scarred cysts secondary to trauma,
renal abscess,
cystic nephroma and cystic renal cell carcinoma.
This is the reason why these patients usually undergo surgical resection (3).
Bosniak IV cysts: These are considered malignant lesions until proven otherwise,
with a likelihood of malignancy of 100%.
Require surgery.
Radiological features: Fig. 5
- Cystic mass with enhancing nodular soft-tissue components.
- Thick irregular walls and/or septa with measurable enhancement (> 20 UH).
- Enhancing nodule in septa/cyst wall.
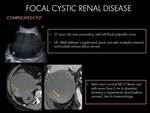
FOCAL CYSTIC RENAL DISEASE:
Focal cystic renal disease includes renal sinus cyst,
pyelocalyceal diverticulum,
simple cyst,
complicated cyst,
pyogenic renal abscess,
mixed epithelial and stromal tumor (MEST),
cystic nephroma and cystic renal cell carcinoma.
1. Renal sinus cyst: Fig. 6
1.1. Peripelvic: Asymptomatic cysts that arise from the lymphatics of the renal hilum.
They usually are multiple,
small,
and bilateral and non-communicating cysts (1).
- US findings: Anechoic lesions with posterior acoustic enhancement. Easily confused with hydronephrosis on US and NECT.
- CT findings: Hypoattenuating cystic lesions.
CECT during excretory phase helps to differentiate them from hydronephrosis,
where the high-attenuation contrast agent–filled collecting system can easily be distinguished from fluid-attenuation renal sinus cysts.
1.2. Parapelvic: These are cortical simple cysts with extension into the renal sinus fat.
They usually are solitary,
unilateral,
and spherical,
non-communicating cysts (1). Although most are asymptomatic,
parapelvic cysts may cause hydronephrosis if they compress the collecting system.
- US findings: Anechoic lesions with posterior acoustic enhancement.
No Doopler US signal is shown.
- CT findings: Hypoattenuating round cystic lesions which do not communicate with the collecting system and,
therefore,
do not fill in with contrast material during contrast-enhanced CT excretory phase.
2. Pyelocalyceal diverticulum:
Pyelocalyceal diverticulum is a urine-filled cavity within the renal cortex that communicates with the renal collecting system.
There are two types of pyelocalyceal diverticulum: type 1 (most common),
that communicates with a minor calyx,
and type 2 that connects with a major calyx or the renal pelvis (1).
- US findings: Similar to a simple cyst.
If complicated by calculi or milk of calcium it appears as an anechoic cyst with layering,
mobile and echogenic material (1).
- NECT or nephrographic phase CECT: A simple pyelocalyceal diverticulum without stones has a similar appearance to a simple cyst.
- Excretory phase CECT: There is a contrast layer of contrast material within the cystlike structure,
because of communication with the renal collecting system.
3. Complicated cyst:
A simple cyst may become complicated due to hemorrhage,
infection,
ischemia or presence of proteinaceous fluid (2).
Fig. 7 Fig. 8
- Haemorragic cyst: Cystic lesion with internal echoes (clot).
Thick calcified wall and/or multiloculated appearance due to chronic changes.
- Infected cyst: Cystic lesion with thick wall and scattered internal echoes.
It may contains debris-fluid / air-fluid level.
An increase of peripheral vascularity could be demonstrate at Doppler- US.
- CT findings: Classification and management is based on Bosniak´s classification .
- Hemorrhagic cyst: Homogeneous hyperdense content (60-90 UH),
due to acute phase.
Heterogeneous content due to clot or debris,
wall thickening and calcifications,
due to chronic changes.
- Infected cyst: Thick wall,
septated,
heterogeneous enhancing fluid,
debris-fluid or gas-fluid level,
calcifications may be present.
Granulation tissue in inflammatory/infected cysts may enhance.
- Ruptured cyst: Retroperitoneal or perinephric collection of fluid and/or blood.
4. Pyogenic renal abscess:
Renal abscesses are typically seen as a complication of acute pyelonephritis.
They often occur as the result of ascending urinary tract infections (80%),
and less commonly,
it can be caused by haematogenous seeding in patients with bacteremia (20%) (1).
- Anechoic-hypoechoic complex cystic mass with posterior acoustic enhancement.
Internal echoes (debris),
thick wall,
septations and loculations may also be seen.
- "Ring-down" artifacts and "dirty shadowing”,
may also be present.
- On color Doppler ultrasound there is a peripheral flow (hyperemia) with central flow absent or within septa.
- CT findings: CT and MR imaging are more sensitive modalities for detecting a renal abscess (1).
Fig. 9
- CECT demonstrates a well-defined,
hypoattenuating,
round mass,
with enhancement of abscess wall (“Ring sign”).
Gas within collection and septations may be present.
- Normal enhancement of adjacent renal tissue.
Perinephric extension of the abscess in relation with inflammatory changes,
with an altered renal contour may be seen.
5. Mixed epithelial and stromal tumor (MEST):
MEST is a rare,
typically benign condition that occurs predominantly in perimenopausal women.
Common presenting symptoms include hematuria,
flank pain,
or a flank mass,
although 25% of patients are asymptomatic.
It is usually unilateral,
and histologically is a complex solid and cystic renal tumor with stromal and epithelial elements (1) (4).
The imaging appearance is nonspecific,
and it cannot be confidently distinguished from other malignant entities.
Therefore,
most cases are treated surgically (1)(4).
- US findings: Well-marginated,
multiloculated cystic and solid mass predominantly hiperechogenic mass.
- Well-circumscribed,
multiseptate cystic and solid mass with delayed contrast material enhancement.
It may herniate into the renal pelvis.
- Internal septa demonstrated variable contrast enhancement,
and an enhancing mural nodule or mural calcifications may be seen.
- Intralesional hemorrhage may be seen (suggested by the presence of hyperattenuating components at CT).
6. Cystic Nephroma:
It has been postulated that cystic nephroma and MEST may be part of the same spectrum of stromal epithelial lesions.
Cystic nephroma is a rare benign tumor that typically occurs in infants under 4 years of age or women aged 40–60 years.
Children often present with an abdominal mass,
while adults are more likely to present with abdominal pain or hematuria but may be asymptomatic (1).
The imaging appearance is nonspecific,
and it cannot be confidently distinguished from other malignant entities.
Therefore,
most cases are treated surgically (1). Fig. 10
- US findings: Well defined,
multiloculated mass,
with multiple cysts and thin,
echogenic septa.
It is surrounded by an echogenic capsule.
- Well-circumscribed,
unilateral,
encapsulated,
multicystic mass with no connection with excretory system,
but lesions often herniate into the renal pelvis or proximal ureter.
- Thin septa with variable enhancement and calcifications may be seen.
- Adjacent normal renal parenchyma.
- Enhancement,
solid component or haemorrhagic areas,
can be depicted.
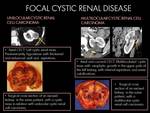
7. Cystic renal cell carcinoma:
Cystic renal cell carcinoma demonstrates identical features to those of a complicated cyst,
abovementioned.
The definitive differentiation between complicated cyst and cystic renal cell carcinoma requires histologic examination.
It can be presented as a uni or multilocular cystic renal cell carcinoma,
as a simple cyst that becomes a cystic renal cell carcinoma,
or as a solid cystic renal cell carcinoma with cystic/necrotic component.
7.1 Unilocular or multilocular cystic renal cell carcinoma: Fig. 11
- US findings: Unilocular or multilocular renal mass,
predominantly hypoechoic due to hemorrhage and necrosis.
It may contain fluid-debris levels,
thick and irregular wall or septations,
and nodules.
- Cystic renal lesion with Bosniak IV features.
- Multilocular: Variably septated and sized cysts separated from the kidney by a fibrous capsule.
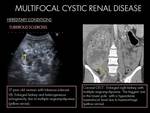
MULTIFOCAL CYSTIC RENAL DISEASE: ACQUIRED,
DEVELOPMENT AND HEREDITARY CONDITIONS
Multifocal cystic renal disease is categorized as acquired,
developmental and hereditary disorders.
1. ACQUIRED CONDITIONS
Include: Acquired cystic kidney disease and lithium-induced nephrotoxicity.
1.1 Acquired cystic kidney disease:
Acquired cystic kidney disease is a condition that develops in end-stage renal disease patients,
with no heritable cystic renal disease associated.
The incidence increases with the length of time on dialysis.
It is equally common with haemodialysis as with peritoneal dialysis.
Renal malignancy will develop in 3%–7% of patients with acquired cystic kidney disease.
Risk factors include male sex and an increased length of time on dialysis (1).
Patients are usually asymptomatic.
Diagnostic criteria (US,
CT,
MR): Require the presence of at least three cysts on each kidney (0.5–3 cm in diameter),
that vary in size and complexity (1).
- US findings: Kidneys usually decreased in size. Atrophic echogenic kidneys with cysts of varying size and complexity.
- CT and MR findings: Kidneys usually decreased in size.Cystic lesions that vary in size and complexity.
Most of them arise from renal cortex,
although they may be seen in medulla as well.
Fig. 12
1.2 Lithium-induced nephrotoxicity:
Nephrotoxicity due to long-term lithium therapy,
may develop chronic focal interstitial nephritis that results in progressive,
nonreversible,
chronic renal insufficiency (1).
- US findings: Multiple punctate echogenic foci which may be cortical or medullary in location,
in relation with multiple uniformally distributed microcysts. Normal-sized kidneys.
Fig. 12
- CT findings: Multiple milimetrical hypoattenuating lesions in the cortex and medulla,
in relation with microcysts.
Typically are 1 to 2 mm in diameter. Normal-sized kidneys.
- MR findings: Technique of election. Multiple small 1 to 2 mm round lesions distributed in the renal medulla and cortex,
which are hyperintense on T2-weighted MR.
2. DEVELOPMENT CONDITIONS:
Development conditions include: Glomerulocystic kidney disease (GCKD),
medullary sponge kidney (Cacchi-Ricci disease),
multicystic dysplastic kidney and localized cystic renal disease.
2.1 Glomerulocystic kidney disease (GCKD):
GCKD is a rare form of cystic renal disease characterized histologically by uniform cystic dilatation of the Bowman´s capsule.
Usually bilateral.
It usually affects neonates and young children,
and it may present as mild renal failure with concomitant congenital malformations (1).
- US findings: It can appear as a cortical hyperechogenicity,
but it is difficult to depict small cysts.
- CT findings: Because patients typically present with renal failure symptoms. NECT is typically performed although it has not an adequate diagnostic profitability.
- MR findings: Non-enhanced T2-weighted MR,
is the preferred imaging modality for diagnosis (1). Small renal cysts with cortical subcapsular distribution hyperintense on non-enhanced T2-weighted MR. The cortical distribution helps to distinguish this entity from other cystic renal diseases. Kidneys may be normal in size or hypoplastic.
2.2 Multicystic dysplastic kidney (MDK):
MDK is a non-heritable developmental paediatric cystic renal disease,
in which the kidney is parcial or completely replaced by non-functioning and non-communicating cysts.
It is usually unilateral.
MDK develops in utero,
when it can be diagnosed; Or in childhood which is most commonly assessed with ultrasound (1) (5).
In most cases,
it regresses by adulthood.
- The affected kidney parenchyma is replaced by multiple cysts of varying sizes and shapes,
showing a lobulated renal contour with a large cluster of peripheral non-communicating cysts.
- Fibrous renal parenchyma is shown as echogenic areas with absent or small hilar vessels.
- An absence or severe atrophy of the ipsilateral ureter,
renal collecting system,
and renal vasculature could be present.
- Real time ultrasound imaging is useful to exclude any communication with the ureter.
- There may be compensatory hypertrophy of the contralateral kidney.
- In children it is important to assess the contralateral renal collecting system to exclude vesico-ureteral reflux.
- MR findings: Multiple renal cysts replacing renal parenchyma hyperintense on non-enhanced T2-weighted MR.
2.3 Medullary sponge kidney (Cacchi-Ricci disease):
Medullary sponge kidney is a congenital developmental abnormality characterized by ectasia and cystic dilatation of the intrapapillary portions of the renal medullary collecting ducts.
It is characterized by small cysts (less than 1 cm in diameter) in the renal medulla.
Both kidneys are usually affected (75%),
but unilateral (25%),
or segmental involvement also may be seen.
Most patients with medullary sponge kidney are asymptomatic (5).
Complications such as nephrolithiasis,
renal calculi,
and urinary tract infection may be seen.
- US findings: Non-specific features. Medullary nephrocalcinosis: Discrete hiperechogenic foci with acoustic shadowing within medullary pyramids.
Fig. 14
- NECT: Medullary nephrocalcinosis and urolithiasis.
- CECT:"Paintbrush" appearance: Retention of contrast within dilated tubules in pyramids tubules.
Striated or beaded appearance of the medullary collecting ducts. Hydronephrosis and hydroureter,
due to obstruction. Severe disease: Extracalyceal contrast accumulation within papillae or abscess.
2.4 Localized cystic renal disease
Localized cystic renal disease is a rare,
benign,
non-heritable form of cystic renal disease,
in which a cluster of tightly spaced,
variably sized cysts that replace a portion or the entire renal parenchyma.
It is a non-progressive affection with unilateral involvement.
There are no other organs with cystic involvement.
It could be asymptomatic.
Common presenting symptoms include abdominal pain,
flank mass,
hematuria,
and hypertension (1).
The differential diagnosis should be established with autosomal dominant polycystic kidney disease,
multilocular cystic nephroma or cystic neoplasm.
The absence of a capsule surrounding the cysts helps to distinguish localized cystic renal disease from cystic nephroma and multiloculated cystic renal cell carcinoma (1).
- Multiple small simple cysts replacing parcial or completely the renal parenchyma.
- No Doppler signal inside the cysts.
- Adjacent normal renal parenchyma.
- Contralateral kidney is unaffected.
- Multiple small simple cysts partially or completely replacing the renal parenchyma.
- Unencapsulated mass.
- Normal atrophic parenchyma separating cysts.
- Normal excretory phase on contrast enhanced CT.
- No involvement of contralateral kidney.
- MR findings: Multiple cysts of high signal intensity on non-enhanced T2-weighted MR,
and normal intervening parenchyma. No enhancing nodularity on enhanced T1-weighted MR.
3. HEREDITARY CONDITIONS:
Hereditary conditions include autosomal dominant polycystic kidney disease,
medullary cystic kidney disease,
Von-Hippel- Lindau disease and tuberous sclerosis complex.
3.1 Autosomal dominant polycystic kidney disease (ADPKD):
ADPKD disease is a heritable cystic renal disease that causes cysts along any segment of the nephron.
It is the most common hereditary cause of end stage renal failure (1) (5).
The kidneys are normal at birth,
and with time,
they develop multiple cysts.
At the age of 30 years,
approximately 68% of patients will have visible cysts by ultrasound.
About 50% of individuals with ADPKD will develop end-stage renal disease by 6th decade.
Unlike in some other congenital cystic kidney diseases,
there is no increased risk for renal cell carcinoma unless the patient is undergoing prolonged dialysis (1) (5).
Clinical presentation is variable and includes: Flank pain,
flank mass,
haematuria,
hypertension ( which usually develops at the same time as renal failure),
renal functional impairment to renal failure.
- US findings: Enlarged kidneys with multiple cysts: Simple or more complex renal cysts (1). Additionally ultrasound is also able to detect cysts in other abdominal organs. Most commonly include hepatic cysts.
Cysts are less commonly seen in the pancreas,
spleen,
ovaries,
and testes.
- CT findings: Enlarged kidneys replaced by innumerable cysts: Simple or complicated (hemorrhagic or infected) cysts. Progressive increase in cyst and renal volume.
Fig. 15
3.2 Medullary cystic kidney disease:
Medullary cystic kidney disease is an autosomal dominant paediatric cystic renal disease,
that eventually leads to end-stage renal disease.
It is characterized by multiple medullary cysts and tubule-interstitial nephritis.
The diagnosis of medullary cystic kidney disease is mainly based on clinical features and a thorough review of family history (5).
Clinically,
patients with medullary cystic kidney disease present with polyuria.
The presence of medullary cysts supports the diagnosis but is not essential.
The differential diagnosis includes medullary sponge kidney,
multicystic dysplastic kidney,
and lithium-induced renal disease.
Characteristic clinical findings and the typical location of cysts in the renal medulla help differentiate medullary cystic disease from other renal cystic diseases.
- US and CT findings: Ultrasonography is the modality of choice for the initial imaging evaluation (5).
- Bilateral normal to small-sized kidneys.
- Multiple cysts (of size less than 3 cm) at the cortico-medullary junction and in the renal medulla may be seen.
3.3 Von-Hippel-Lindau disease (VHL):
VHL disease is a hereditary,
autosomal dominant disease characterized by the development of cysts,
cystic neoplasms,
and hypervascular tumors in the visceral organs (1).
- US and CT findings: (1) (5).
- Bilateral normal-sized kidneys,
because most of the cysts and renal cell carcinomas are small.
- Renal cysts (presented in 59 to 63% of patients): Usually bilateral,
multiple,
and of various size.
Simple or complex cysts (1).
- Renal cell carcinoma (presented in 25 to 45% of patients): Cystic and solid renal cell carcinoma.
Frequently multiple and bilateral (1).
Fig. 15
Although some renal cell carcinomas in patients with VHL arise as de novo solid lesions,
they many arise in preexisting cysts.
Moreover,
cysts that appear simple at imaging often contain small foci of renal cell carcinoma.Therefore,
in a patient with VHL,
no cyst can be confidently dismissed at imaging as free of tumor cells (1).
Current recommendations for the treatment of renal lesions in VHL disease include follow-up imaging at 6- to 12-month intervals for tumors with a maximal diameter of less than 3 cm and treatment of those measuring more than 3 cm (1) (5).
3.4 Tuberous sclerosis complex:
Tuberous sclerosis is an autosomal dominant neurocutaneous syndrome that causes hamartomas in the kidneys,
lungs,
liver,
brain,
heart,
eyes,
and skin.
The most common renal manifestation of tuberous sclerosis is angiomyolipoma,
which occurs in 55%–75% of patients.
Angiomyolipomas are benign tumors containing angiomatous,
myomatous,
and lipomatous tissue.
The next most commonly seen masses associated with tuberous sclerosis are simple renal cysts (1).
The overall incidence of renal cell carcinoma is similar to that in the general population.
However,
renal cell carcinoma manifests at an earlier age in patients with tuberous sclerosis.
- US and CT findings: (1) (5).
At imaging,
multiple renal cysts intermingled with angiomyolipomas in both kidneys are char- acteristic features of tuberous sclerosis complex.
Both angiomyolipomas and renal cysts may manifest during childhood and tend to increase in size and number with increasing age.
Fig. 16
- US angiomyolipoma appearance: Markedly hyperechoic mass relative to normal adjacent renal tissue. If muscle,
vascular elements,
or hemorrhage predominate,
lesion may be hypoechoic. May look similar to small renal cell carcinoma,
which also may be hyperechoic.
- CT angiomyolipoma appearance:
- NECT: Renal solid mass with intramural fat.
- CECT: Lesions may significantly enhance with contrast,
depending on extent of vascular component.
Aneurysmal renal vessels may be seen.
* At imaging,
differentiation of lipid-poor angiomyolipoma from renal cell carcinoma is difficult.
Rapid growth and the presence of calcification are two imaging findings that suggest renal cell carcinoma.
However,
a biopsy may be necessary for differentiation.