Keywords:
Breast, Abdomen, Oncology, MR, Ultrasound, Mammography, Screening, Diagnostic procedure, Biopsy, Cancer, Radiotherapy techniques, Tissue characterisation
Authors:
J. C. Benson, P. Bolan, M. Nelson; Minneapolis, MN/US
DOI:
10.1594/ecr2018/C-0119
Aims and objectives
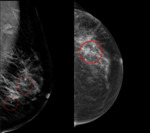
Breast biopsy markers assist in the precise localization of biopsy-proven neoplastic breast lesions,
minimizing the volume of breast tissue needed to be excised during the surgical removal of the tumor.1 Already,
markers have been shown to be useful in the delineation of the tumoral bed,
with negative margins reportedly to be as high as 90% with the use of clips.2 Furthermore,
biopsy clips are known to be extremely safe,
with allergic reactions reported only in exceptionally rare cases.1,3
In addition,
successful treatment of breast cancer with neoadjuvant chemotherapy can result in total pathologic response within a few weeks.
This can significantly increase the technical difficulty of ultrasound delineation and/or detection of residual tumor during preoperative guidewire placement.
Hence,
optimization of breast markers is of utmost import in the accurate localization of residual tumor.
To address this,
we assessed the use of a relatively new,
FDA-approved breast marker (VizMark,
Mermaid Medical,
Copenhagen,
Denmark) in patients receiving neoadjuvant chemotherapy for biopsy-proven breast cancer.
The purposes of the study were to: 1) assess the efficacy of the breast marker to successfully delineate the location of biopsy-proven tumor following treatment with neoadjuvant chemotherapy,
and 2) to note the imaging characteristics of the marker on follow-up examinations.