Hepatic infarction
Hepatic infarction is defined as an area of coagulation necrosis from hepatocyte cell death caused by local ischemia,
which in turn results from the obstruction of circulation to the affected area [1].
It is an extremely rare situation because of the liver supply.
Hepatic infarction can occur when there is both hepatic arterial and portal vein flow compromise; in fact,
in most cases,
it results from either insult to the hepatic artery or portal vein thrombosis superimposed on hepatic arterial occlusion [1].
It may occur as a complication of hepatic artery stenosis or thrombosis after liver transplantation,
or it may be secondary to hypercoagulability,
vasculitis,
or infection in sepsis and shock (Fig.
1).
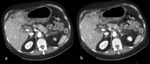
Tipically infarction presents on CT as an ill-defined wedge-shaped area of hypoattenuation,
which is mostly peripheral without mass-effect on adjacent structures in post-contrast images (Fig.
2,
Fig.
3).
Portal vein thrombosis
Decreased portal vein inflow and increased arterial inflow are two conditions that can occur in portal vein thrombosis.
Portal vein thrombosis is characterized by a thrombus developed in the main portal vein,
and/or its right or left branches,
or by the permanent obliteration that results from a prior thrombosis.
It may be seen with many clinical conditions including cirrhosis,
abdominal neoplasms,
intrabdominal inflammatory processes such as Crohn disease,
diverticulitis and appendicitis,
hypercoagulable states,
and trauma [5].
Recently,
portal vein thrombosis was found to be associated with metabolic syndrome,
especially with central abdominal obesity.
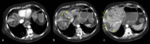
Thrombus is usually seen as a hypodense filling defect in the lumen of the portal vein,
with partial or complete occlusion on contrast-enhanced CT (Fig.
4).
Unenhanced scans have been shown to be of minimal benefit in the identification of thrombus,
except in the case of early thrombosis,
because it appears hyperdense (Fig.
5).
Occlusion of a branch of the portal vein by a thrombus can manifest as transient hepatic attenuation differences in the late arterial phase,
showing increased enhancement of the lobe or segment previously supplied by the vein due to hepatic arterial compensatory flow (Fig.
6).
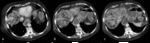
Cavernous transformation of the portal vein is a sequela of chronic portal vein thrombosis and/or occlusion,
which leads to the development of numerous periportal collaterals,
which appear as a mass of veins in the porta hepatis on contrast-enhanced CT (Fig.
7,
Fig.
8) [6].
Hereditary Hemorrhagic Telangiectasia (Osler-Weber-Rendu Syndrome)
It’s a rare hereditary autosomal dominant vascular disorder.
Hepatic involvement occurs in up to 30% of cases and consists of vascular,
parenchymal,
and biliary lesions with characteristic telangiectasias and vascular shunts.
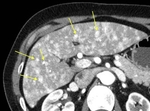
On arterial phase,
liver shows characteristically mosaic heterogeneous perfusion enhancement.
These abnormalities are best seen during arterial phases,
almost always disappearing in the hepatic phase as the hepatic parenchyma becomes homogeneous (Fig.
9,
Fig.
10).
The cause of this mosaic appearance is multiple arteriovenous shunts showing different attenuation and telangiectasias [7].
Telangiectasias are hypervascular rounded nodules with predominant peripheral location (varying from a few millimeters to 1 cm in size) in the arterial and late arterial phases,
often becoming isointense in the portal venous and delayed phases (Fig.
11) [7].
Sometimes integrated confluent vascular masses appear as larger vascular pools (25%) and these masses are characterized by early and persistent enhancement during the arterial phase (Fig.
12).
Opacification of the hepatic veins during the arterial phase was considered as an indirect sign of the presence of hepatic-arteriosystemic venous shunt (Fig.
13) ; arterial dilatation (a common hepatic artery greater than 7 mm in diameter) can be seen and it is a consequence of an increased volume flow in the hepatic artery and veins caused by intrahepatic fistulas (Fig.
9) [8].
Early and prolonged enhancement of the portal vein during the arterial phase was considered an indirect sign of the presence of arterioportal shunt.
Budd Chiari syndrome
Budd-Chiari syndrome results from the hepatic venous outflow obstruction at any level,
from the small hepatic veins to the junction of the inferior vena cava and the right atrium (Fig.
14,
Fig.
15).
Thrombosis is the most common cause of hepatic vein obstruction,
most commonly results from a hypercoagulable states.
The imaging findings of Budd-Chiari syndrome are variable and depend on the stage of the disease.
In acute Budd-Chiari syndrome,
the morphologic of the liver usually is normal,
and occlusion of the hepatic veins with ascites are the typical finding (Fig.
16).
The liver exhibits patchy,
decreased peripheral enhancement caused by portal and sinusoidal stasis and stronger enhancement of the central portion of the liver parenchyma [9].
In subacute or chronic Budd-Chiari syndrome,
the morphologic changes in the liver are the result of the type of venous involvement,
and portosystemic and intrahepatic collateral vessels are often found [9]. Contrast-enhanced CT is useful for depicting regions of hypoperfused liver parenchyma and hepatic veins thrombosis (Fig.
17).
Chronic Budd-Chiari syndrome is also characterized by the development of multiple regenerative nodules,
which can be viewed as a response to a focal loss of portal perfusion and hyperarterialization in areas with preserved hepatic venous outflow [9].
Passive hepatic congestion
Passive congestion occurs with the stasis of blood flow within liver parenchyma as a result of impaired hepatic venous drainage secondary to cardiac disease [1].
Elevated central venous pressure is directly transmitted from the right atrium to the hepatic veins [10].
On contrast-enhanced CT scans of patients with passive hepatic congestion,
an inhomogeneous,
mottled,
reticulated-mosaic pattern of parenchymal may be observed in arterial phase in association with early enhancement of hepatic veins due to contrast reflux from right atrium into inferior vena cava (Fig.
18,
Fig.
19).
In portal phase liver parenchyma become omogenous [1].
Hepatic sinusoidal obstruction syndrome
Hepatic sinusoidal obstruction syndrome (SOS) is a vascular disorder that has been described at first as a complication of oral contraceptive therapy; now it is associated with several other conditions such as pregnancy,
granulomatous disease,
neoplasm,
rheumatoid arthritis,
HIV infection,
Hodgkin's disease,
infiammatory disorders.
It is caused by toxic injury to sinusoidal endothelial cells.
Histologically,
it is the result of sinusoidal congestion and centrolobular hemorrhagic necrosis due to non-thrombotic occlusion of the central hepatic veins,
whereas large hepatic veins remain patent [12].
This disease has long been recognized as a consequence of poisoning with pyrrolizidine alkaloids-containing plants,
consumed either as contaminated flour or as traditional or herbal remedies.
Currently,
the most common cause for the disease is toxicity from various chemotherapeutic agents or regimens,
particularly,
but not exclusively,
when used for myeloablation prior to hematopoietic stem cell transplantation [11].
Today,
SOS has been associated with more than 20 drugs including conventional doses of some immunosuppressive and chemotherapeutic agents.
[11].
On contrast-enhanced CT,
hepatic veins congestion causes a mottled pattern of contrast enhancement in the hepatic arterial and early portal venous phases with decreased enhancement in the liver periphery.
The areas of decreased enhancement are due to decreased portal flow,
hepatic congestion and ischemia.
On delayed images enhancement of the liver becomes more homogeneous (Fig.
20).
On MRI,
the presence of reticular hypointensity on hepatobiliary phase images obtained using gadoxetic acid–enhanced MR imaging is highly specific for the diagnosis of SOS (Fig.
21) [12].