NORMAL FINDINGS AT US AND CT AFTER RENAL TRASPLANTATION
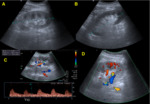
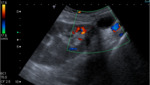
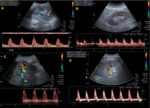
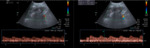
Ultrasound (Image 1)
A complete US evaluation should include B-mode,
Doppler-color and Doppler-spectral imagines:
Renal parenchyma: normal transplanted kidney demonstrates no differences in comparison with a normal native kidney.
- Length: Variations in size of the graft are indirect sings of pathology.
- Cortico-medullary differentiation: Usually,
cortico-medullary differentiation in a transplanted kidney is more pronounced,
due to its more superficial location.
- Cortical assessment: higher-frequency transducers allow better appreciation of the individual cortical segments.
Collector system: Assessment of whether or not hydronephrosis is present. Often a mild degree of pelvicalyceal dilatation can be transitorily seen in the immediate post-operative period,
due to the reflux,
oedema at site of anastomosis and autonomic denervation.
In the early-days visualization of a thin urothelium in the renal pelvis is possible.
Peri-renal space: check for perinephric collections.
Color imaging:
Color maps in renal grafts should be homogeneous,
with arterial and venous flow extending to within a few millimeters of the capsule.
Main renal artery and vein and iliac vessels should be permeable,
homogeneously filled with color along their whole trajectory.
Spectral imaging:
Normal Doppler reflects a low resistance vascular bed.
Resistive indices (RIs) are obtained from interlobar or segmental arteries in upper,
medium and lower thirds.
A cut-off point of 0,80 is considered for transplant dysfunction.
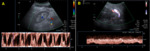
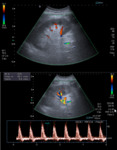
Peak systolic velocity (PSV) should be recorded at the main renal artery) and in the external iliac artery:
- PSV of the main renal artery should be less than 250 to 300 cm/s.
- Normal triphasic high-resistance waveforms should be observed in the ipsilateral external iliac artery (image 2).
- The normal PSV ratio of the renal artery to the ipsilateral external iliac artery has been described to be less than 1.8 to 3.5.
The main renal vein should have monophasic to mildly pulsatile flow,
similar to the external iliac vein (image 2).
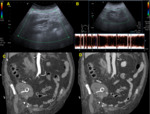
CT and CT angiography
At unenhanced CT,
the renal parenchyma demonstrates homogeneous soft-tissue attenuation.
After contrast material administration three phases can be obtained:
- Arterial-phase (cortical nephrogram-phase): A ROI is placed in the aorta and the scan starts when a determined threshold is reached.
It is used to evaluate renal graft artery and iliac arterial system.
Because of early venous graft return,
it is common to find graft vein filled with contrast.
In this phase,
renal cortex appears hyperattenuating compared to the medulla.
- Venous-phase (tubular nephrogram-phase): 80-100 sec after contrast administration injection.
Normal parenchyma is uniformly enhanced.
It is useful for demonstrating parenchymal masses.
The iliac venous system can also be assessed.
- Late excretory-phase (pyelogram-phase): 7 minutes after contrast administration injection.
Used to evaluate pyeloureteral system.
COMPLICATIONS AFTER RENAL TRASPLANTATION
We revised all renal transplantations performed in our center between January 2014 and June 2017 (total: 193).
We categorized complications using an anatomical classification and reviewed the prevalence and imaging features (US and CT) of each complication.
|
PERIRENAL COLLECTIONS
|
23%
|
|
PARENCHYMAL ABNORMALITIES
|
66%
|
|
UROLOGICAL COMPLICATIONS
|
15%
|
|
VASCULAR COMPLICATIONS
|
18%
|
|
POST-BIOPSY COMPLICATIONS
|
<0,1%
|
|
OTHERS
|
|
PERIRENAL COLLECTIONS
In our study 23% of renal transplantations showed a perinephric collection.
General approach
Appearance of fluid collections is variable.
A systematic approach can be very helpful:
- First step: collection’s homogeneity.
When a collection shows homogenous fluid content,
it will be consistent with: seroma,
lymphocele,
urinoma or abscess.
Most of the times hematomas can be excluded.
- Collection’s wall.
Usually,
abscesses show an irregular and enhancing wall,
while seromas,
lymphoceles and urinomas are encapsulated by a thin (sometimes not visible) and less enhancing wall.
- Hematomas may have distinctive appearance: at US hematomas demonstrate complex appearance; with hypoechogenic heterogeneous areas,
multiple septations and fluid-fluid levels.
At CT,
they are usually hyperdense in non-contrast studies,
with attenuation values greater than 30 to 40 HU.
- Gas and fat stranding: The presence of gas inside a collection is very suggestive of abscess.
Stranding of the surrounding tissues is also suggests infection.
- Location: When settled adjacent to the renal parenchyma or urinary tracts an urinoma cannot be excluded.
If the collection is detached to the urinary tract an urinoma is less probable and seromas or lymphoceles are the most likely.
- Seromas and lymphoceles appear to be indistinguishable based only on radiological features.
One key is that seromas appear immediately after surgery and decrease spontaneously,
whereas lymphoceles develop later and tend to grow.
- Contrast-enhanced CT (delayed-phase) can establish the diagnosis of urinary leakage; and thus,
also urinomas.
- Growing collection: urinary leakage (growing urinoma) should be taken into account,
hematoma with associated vascular injury (arterial or venous bleeding) or an abscess.
- Time of appearance: Urinomas,
hematomas and seromas are most likely to develop immediately after transplantation,
whereas lymphoceles generally occur 4 - 8 weeks after surgical procedure.
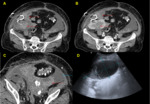
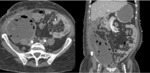
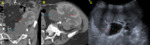
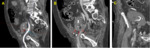
Hematomas (Image 3)
In our hospital,
hematomas have been the most common peritransplant collections (2/3 of all),
usually in the immediate postoperative period.
The radiological appearance of hematomas is time-dependent.
When showing typical appearance,
hematomas at US demonstrate complex appearance; with hypoechogenic heterogeneous areas,
multiple septations and fluid-fluid levels.
At CT,
they are usually hyperdense in non-contrast studies (attenuation values greater than 30-40 HU).
Size,
location,
and growth determine the significance of these collections.
When small,
size-stable and asymptomatic they are considered normal finding after surgery.
In hematomas,
radiologist should consider possible focuses of active bleeding,
especially when progressive enlargement of the collection is proved.
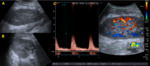
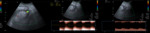
To detect active bleeding CT scan should be performed before and after contrast administration.
Non-enhanced CT helps to differentiate contrast extravasation from potential pitfalls such us surgical material (image 4).
Following trauma or transplant biopsy subcapsular hematomas can appear.
They usually show a crescentic appearance and exert mass effect on the underlying renal parenchyma,
situation known as Page Kidney.
This mass effect may result in hypertension.
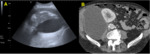
Lymphoceles (image 5)
Lymphoceles rank second in our study (22% of all collections),
although they have been described as the most common collections in literature (5-20%).
They are more likely to appear some weeks after surgery,
commonly 4 – 8 weeks.
Lymphoceles are round or oval,
well-defined,
homogeneous fluid collections.
Septations may be seen,
but are less common than in hematomas.
Seromas (image 6)
Seromas are unusual (<1%) and tend to appear in the immediate postoperative period.
Their radiological appearance consists on a rounded and sharply defined fluid collection.
Smooth and thin septations may be seen.
Radiologically lymphoceles and seromas can be very similar.
However lymphoceles tend to appear some weeks after surgery,
while seromas are present from the immediate postoperative period.
Urinomas (image 7)
Urinomas are uncommon complications (less than 1% registered at our institution).
They manifest in the early postoperative period.
Most common site of leakage is the ureterovesicle anastomosis,
due to surgical technique or distal ureteral necrosis.
Radiological features of urinomas are similar to lymphoceles and seromas.
Differentiation with lymphoceles can be done based on post-surgical time.
Urinomas and seromas are more difficult to distinguish.
If patient’s renal function allows,
contrast-enhanced CT or MR imaging with delayed images acquisition can be used.
Accumulation of contrast material inside the collection confirms urinary leakage (image 6,
B and C).
Abscesses (image 8)
Peritransplant abscesses are rare.
They usually develop within the first few weeks after transplantation,
by pyelonephritis or bacterial superinfection of a lymphocele,
hematoma,
seroma or urinoma.
Patients may have few signs or symptoms of infection because of their immunosuppressed state.
Abscesses have a complex,
cystic,
nonspecific appearance.
However,
the presence of gas within a collection is specific.
An irregular enhancing wall and / or a simple collection that becomes sonographically more complex are suspicious.
PARENCHYMAL ABNORMALITIES
Diffuse parenchymal abnormalities
Acute Tubular Necrosis (ATN)
Most frequent complication after renal transplantation,
appearing in the early postoperative period.
Approximately half of renal transplantations (51% in our series) will develop ATN.
Nonspecific radiologic features have been described.
US are the most helpful modality,
as CT usually shows a non-morphologically altered graft or nonspecific perfusion abnormalities.
US may show renal enlargement,
increased cortical thickness with altered echogenicity (increased or decreased) and loss of cortico-medullary differentiation.
Duplex Doppler evaluation is mandatory.
Most cases in our hospital showed elevated resistive index (>0,
75).
Most publications consider a cut-off value of 0,8.
However,
elevated IRs is not a specific measure.
It can also be seen in other causes of graft malfunction (images 9,
10).
Final diagnosis requires biopsy.
Hyperacute and acute rejection
In hyperacute rejection,
the graft fails immediately,
usually identified in the operating room.
Acute rejection is relatively uncommon following transplantation,
up to 9% of patients in our series.
Imaging findings are non-specific and overlap with those described in ATN (image 9).
Doppler shows decreased diastolic flow causing high resistance index (>0.8).
In addition,
some authors believe that a high resistive index,
more than 0.9,
may be considered relatively specific for acute rejection.
The final diagnosis of rejection is histological.
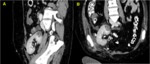
Diffuse renal infarction (image 11 and 18)
A diffuse infarction of the transplanted kidney is rare (only one in our series).
A non-enhancing,
diffusely hypodense and enlarged graft is seen on CT and on US absence of perfusion on colour-Doppler on the entire kidney.
Main renal artery and vein should be evaluated.
Contrast-enhanced ultrasound may be useful.
Diffuse pyelonephritis
Pyelonephritis,
focal and diffuse,
is not common.
Ultrasonography findings include poor cortico-medullary differentiation,
increased renal size,
and increased cortical echogenicity.
Doppler imaging shows decreased or increased perfusion,
with elevated RIs.
The presence of low-level echoes in the pyelocaliceal system in a febrile patient suggests pyonephrosis.
On CT kidney will demonstrate reduced enhancement affecting cortex and medulla and inflammatory changes on the surrounding tissues,
which may help to distinguish acute pyelonephritis and infarct.
Focal affectation of the renal parenchyma
Cyst and masses
Radiological features of cyst and masses do not defer form those in native kidneys.
As immunocompromised patients,
kidney-transplanted patients have a higher prevalence of malignant neoplasms.
Intrarenal abscess
Intrarenal abscesses manifest as nonenhancing,
hypoattenuating collections,
usually coexisting with pyelonephritis.
Fungal infection
Highly echogenic,
weakly shadowing masses within a transplanted collecting system are fairly specific for fungus balls.
Focal infarction and pyelonephritis
Features will be the same for those in diffuse infarction and pyelonephritis,
except for shape and extension.
Focal infarctions and infections adopt a wedge-shaped or patchy morphology.
Image 12 and 13.
On CT “the rim sign” may be helpful to differentiate infection and infarction.
On infarcts,
due to collateral capsular perfusion,
a thin rim of cortex continues to enhance among the infarcted territory.
On pyelonephritis the periphery of the cortex is also affected.
UROLOGICAL COMPLICATIONS
Obstructive uropathy (image 14)
Urinary obstruction occurred in approximately 5% of transplantations in our center.
Obstruction can be primary or secondary.
Primary obstruction occurs within the urinary tracts,
usually due to strictures.
Secondary obstruction is caused by extrinsic compression.
More than 90% of ureteral stenoses occur within the distal third of the ureter,
especially in the implantation site.
US are preferred to confirm hydronephrosis.
Doppler duplex imaging may demonstrate a high-resistance waveform with an elevated RIs (>0.75).
A normal RI is a strong indicator against obstruction.
Infection (urinary tract infection,
pyonephrosis,
fungal infection)
Urinary tract infection (UTI) proved to be the most frequent form of infection,
affecting 9% of all transplanted kidneys and it is responsible for almost 85% of all types of infections in our patient population.
VASCULAR COMPLICATIONS
Renal artery stenosis
Renal artery stenosis is one of the most common vascular complications (rate of 3% in our centre,
although in literature rates up to 10% have been reported).
It presents as a late complication,
most frequently at the anastomotic site.
Typically presents between 3 months and 2 years following the transplant.
Aetiology is mostly related to surgical technique (faulty suture technique,
post-surgical bendings,
twisting,
kinking,
or compression…).
Doppler-duplex imaging will allow evaluation of indirect sings.
The turbulent flow in the stenotic area will result in aliasing effect at color-Doppler (image 15) and elevation of the peak systolic velocity at spectral-Doppler.
Some authors have suggested PSV threshold of at least 250 m/s for significant stenosis.
Acceleration time greater than or equal to 0.08-0.1 second is also suggestive.
In post-stenotic segments of the artery delayed systolic acceleration or parvus - tardus waveforms also suggest stenosis (image 16).
CT angiography depicts the iliac and transplant vessels,
allowing detection of strictures (image 15).
At CTA or MRA,
hemodynamically significant stenosis is defined as luminal narrowing greater than 50%.
Focal arterial dissection (image 15)
In our series,
focal dissections represented 12,5% of all causes of arterial stenosis.
Diagnosis can be done using US,
but CT angiography is more accurate.
Renal artery thrombosis
It is considered a rare complication.
In our review 1.5% of the patients presented it.
They usually occur in the early postoperative period.
Most of our cases consisted in nonocclusive partial thrombus.
Prompt diagnosis is decisive.
Renal artery thrombosis is diagnosed when a clot is visualized inside the renal artery with decreased (partial occlusion) or absent (complete occlusion) intrarenal flow.
Confirmation must be done (CT angiography) (image 18).
Renal vein thrombosis
Renal vein thrombosis is a rare complication (2%).
It is a early post-surgical complication.
On B mode US evaluation an enlarged and hypoechoic kidney is the most meaningful feature.
Identification of echogenic material inside the renal vein suggests clot.
Spectral-Doppler may show a reversal diastolic flow in main renal artery,
which is considered very suggestive of vein thrombosis (image 17).
However,
it is not specific.
On CT,
diagnosis relies on direct visualization of thrombus in the vein (image 18).
Renal vein stenosis
Renal vein stenosis usually results from adjacent extrarenal compression.
The high velocities caused by stenosis are seen on colour Doppler US as areas of colour aliasing.
Features can also be assessed by CT.
POST-BIOPSY COMPLICATIONS
In our study 1 case of AVF and 1 case of pseudoaneurysm were diagnosed.
Arteriovenous Fistulas
Arteriovenous fistulas may form when an artery and vein are lacerated creating a connection between a feeding artery and a vein.
At US they might be seen as anechoic or cystic structures with disorganized color flow (aliasing or “soft tissue bruit") at color-doppler US (image 19).
Feeding artery will show a high-velocity,
low-resistance waveform and vein pulsatile “arterialized” flow.
On CT a round abnormality in the renal parenchyma that enhances similar to the aorta indicates the vascular nature of the lesion.
When symptomatic or large,
arteriovenous fistulas may be effectively treated with embolization (image 20).
Pseudoaneurysms
Pseudoaneurysms are usually presented as cysts with highly vascular flow within them.
Typical pseudoaneurysms will show a neck containing alternatives jets of forward and reverse flow (“to-and-fro” waveform).
The colour filling of the aneurysm sac is commonly described as the “yin-yang sign”.
On CT a round or saccular outpouching from the artery can be demonstrated with contrast-enhanced sequences.