MRI protocol
Axial,
sagittal and coronal T2-WI,
axial FLAIR and sagittal T1-WI of the brain ( Fig. 1 ) are usually acquired along with 3D-DRIVE (figure 2 Fig. 2 ) and Cine phase-contrast sequences (Fig. 3 Fig. 4 ; video Fig. 5 Fig. 6).
Post contrast T1-WI and T2*-WI may be obtained in selected cases.
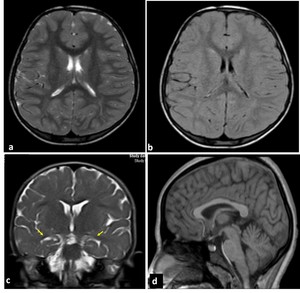
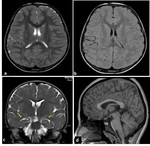
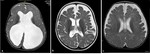
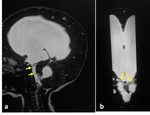
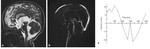
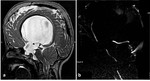
Fig. 1: MRI sequences for hydrocephalus. Normal images a) axial T2=WI, b) axial FLAIR, c) coronal T2-WI through the lateral and third ventricles, Note the morphology of the ventriclular system, especially the temporal horns (arrows), d) Midline sagittal T1-WI image shows the commissures, and fornix. The midbrain with tectal plate and aqueduct, brainstem, fourth ventricle and vermis are well seen
References: Mohammad S
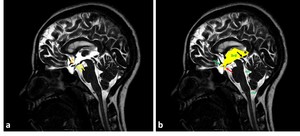
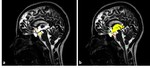
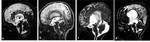
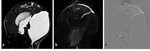
Fig. 2: Sagittal 3D-DRIVE perfectly demonstrating the anatomy of midline structures; the third ventricle (3rd), aqueduct of Sylvius (white asterisk), fourth ventricle (4th) and cisterna magna (CM). Note the concave floor of third ventricle (yellow arrows), and lamina terminalis (orange arrow heads); chiasmatic and infundibular recesses of the third ventricle (green, red arrows); pineal (black arrow), and suprapineal (asterisk) recesses of the third ventricle, anterior commissure (white arrow).
References: Mohammad S
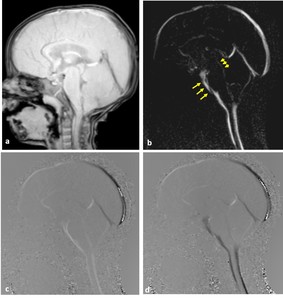
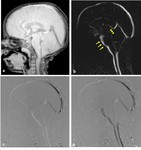
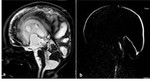
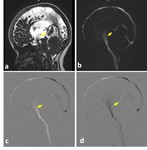
Fig. 3: Phase-contrast sequence in sagittal plane a) Re-phased image in which there is visible background; b) magnitude image in which the flow is bright with suppressed background; c,d) Phase image demonstrates the to-and-fro (pulsatile) bright and black flow of CSF throughout the cardiac cycle. Note the CSF flow in the prepontine-premedullary cisterns (long arrows) and the aqueduct of Sylvius (short arrows)
References: Mohammad S
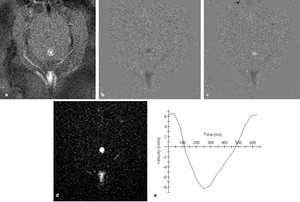
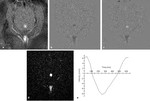
Fig. 4: phase-contrast sequence in axial plane perpendicular to the aqueduct of Sylvius (circle). a) rephrased-image; b,c ) phase images through systole and diastole; d) magnitude image; e) graph demonstrating CSF flow velocity wave.
References: Mohammad S
Image analysis
· Axial,
coronal T2-WIs and FLAIR are analysed first to document ventriculomegaly and to search for any associated structural abnormalities and white matter signal intensity alterations.
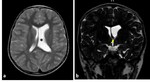
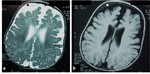
· Axial T2-WI and FLAIR demonstrate ventricular dilatation and the periventricular bright signal either secondary to defective myelination in chronic hydrocephalus or periventricular interstitial edema in acute hydrocephalus (Fig. 7, Fig. 8 ).
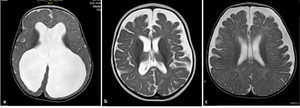
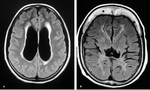
Fig. 7: Axial T2-WI in different cases a) MRI of 7-month-old girl with hydrocephalus associating septo-optic dysplasia showing ventricular dilatation with effacement of extra-axial CSF spaces ; b) 8-month-old boy with delayed development and microcephaly showing ventricular dilatation ex-vacuo, both lateral ventricles and extra-axial CSF spaces are dilated; c) MRI of 7-month-old girl complaining of macrocephaly with normal development showing benign enlargement of subarachnoid space
References: Mohammad S
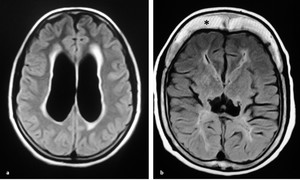
Fig. 8: Axial FLAIR MRI sequence of two different patients. a) periventricular high signal in a setting of acute hydrocephalus secondary to colloid cyst (not shown); b) periventricular high signal reflecting poor myelination due to the noxious effect of hydrocephalus. Note thick skull with prominent diploic space (asterisk) in chronic shunted patient
References: Mohammad S
· Coronal T2WI demonstrates commensurate dilatation of the temporal horn with the lateral ventricles associated with rounding of the lateral angles and medial displacement of hippocampus in cases of hydrocephalus.
Effacement of pericerebral CSF spaces above the level of obstruction is also demonstrated.
However,
in cases with ventriculomegaly ex-vacuo,
the temporal horns,
although large,
are small compared to the bodies of the lateral ventricles; they retain their normal shape and the hippocampi are not displaced medially ( Fig. 7 Fig. 9 ).
In addition,
the pericerebral spaces remain patent ( Fig. 10 ).
However,
in paediatric patients,
assessment of pericerebral CSF spaces can be misleading as both atrophy and hydrocephalus can cause enlargement of the ventricles and sulci without effacement.
Moreover,
the size of the ventricles and subarachnoid spaces have variable size over the first 2 years of life.
So,
knowledge of the infant’s head size is essential; a large head or too rapidly enlarging head suggests hydrocephalus,
whereas a small or diminishing head circumference is more compatible with atrophy [4,8].
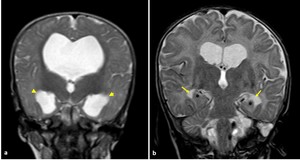
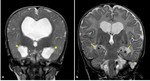
Fig. 9: Coronal T2-WI in two different patients a) MRI of 7-month-old girl with hydrocephalus showing commensurate dilatation of the temporal horns (arrowheads) with the lateral ventricles; b) MRI of 5-month-old boy with brain atrophy, the temporal horns (arrows), are small compared to the bodies of the lateral ventricles; they retain their normal shape and the hippocampi are not displaced medially (asterisk)
References: Mohammad S
· 3D-DRIVE sequence is analysed in sagittal plane.
Widening of the third ventricular recesses (chiasmatic,
sellar and pineal recesses) and the downward bulge of the floor of third ventricle were used as reliable signs for differentiating hydrocephalus from other causes of ventriculomegaly (Fig. 10 ) [4,7].
Axial and coronal reconstructions can be obtained for accurate determination of the site and the cause of obstruction.
The shape of the obstructed/ stenosed aqueduct (either tubular narrowing,
focal obstruction/ stenosis,
associating proximal funnelling) are described.
Presence of syenchiae in the ventricular outlet foramina and basal cisterns can be demonstrated ( Fig. 11 ) [8].
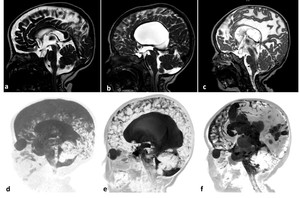
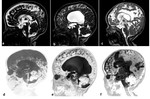
Fig. 10: Sagittal 3D-DRIVE (a,b,c), and maximum intensity projection images (MIP) (d,e,f) in three different patients. a,d) 2-year-old girl with cerebral atrophy shows concave lamina terminalis and tuber cinereum with dilated cortical sulci in the MIP image; b,e) 4-year-old boy with hydrocephalus secondary to aqueduct stenosis demonstrates dilated third and lateral ventricles (note the convex lamina terminalis and tuber cinereum and narrowed cortical sulci in MIP image; c,f) 22-month-old boy with multiple adhesions cystic spaces representing entrapped CSF cysts as a result of inflammatory adhesions. Note the narrowed cortical sulci and multiple loculations in MIP image
References: Mohammad S
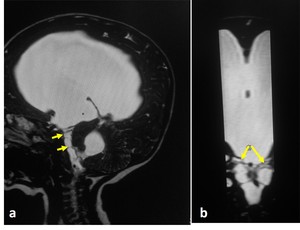
Fig. 11: 7-month-old girl with hydrocephalus. a) sagittal 3D-DRIVE demonstrates prepontine adhesions with dilated ventricular system; b) coronal reformatted image nicely demonstrates the prepontine adhesions (arrows)
References: Mohammad S
· Cine phase-contrast sequence can demonstrate CSF pulsatile flow throughout the cardiac cycle ( Fig. 3 Fig. 4; video (Fig. 5 Fig. 6). It can be acquired in sagittal section to monitor the CSF flow through the aqueduct and basal subarachnoid spaces.
Also,
It can be acquired in axial section for quantification of CSF flow [2].
· The peak CSF flow velocity in paediatric cases as measured in the aqueduct of Sylvius equals 5.77±3.18 cm/sec.
Statistically significant difference was determined between children and adolescents in peak velocity across the aqueduct of Sylvius.
It measures 5.28±2.88 in infants; 6.57±3.41 in children; and 4.12±2.07 in adolescents [9].
· Contrast injection is crucial in cases with intracranial neoplasms or in suspected inflammatory process ( Fig. 12 )
· T2*-WI may be acquired for better detection of intracranial haemorrhage as a cause of hydrocephalus
MRI findings in CSF flow disorders
Obstructive hydrocephalus
· A consensus classification of hydrocephalus has been proposed to relate most cases of hydrocephalus to obstruction to CSF pathway reflecting the surgical approach to hydrocephalus that aims to divert the CSF between the site of production and absorption [10,
11].
Obstruction can be encountered in the aqueduct of sylvius,
fourth ventricular exit foramina,
foramen of Monro ( Fig. 13 ) or secondary to synechiae at the ventricles or basal cisterns.
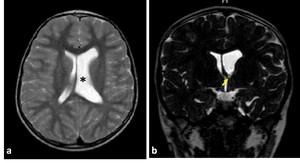
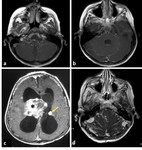
Fig. 13: MRI of six-year-old boy with foramen of Monro stenosis. a) axial T2WI showing dilated left lateral ventricle (asterisk); b) coronal 3D-DRIVE demonstrating foramen of Monro stenosis (arrow)
References: Mohammad S
· Aqueduct of Sylvius is the most prevalent site of obstruction [7,12].
This can be related to its narrow calibre. This can occur secondary to congenital stenosis,
web or post inflammatory adhesions and external compression.
The small fourth ventricle with dilated third and lateral ventricles out of proportion of cortical atrophy were described as criteria that could suggest aqueduct stenosis.
Though,
these are non-reproducible parameters [13].
3D-DRIVE can precisely demonstrate the stenosed aqueduct (Fig. 14 ).
In addition,
phase-contrast imaging support the diagnosis of CSF flow obstruction at the level of aqueduct which aids in the diagnosis of aqueduct stenosis if obstructive hydrocephalus is clinically or radiologically suspected ( Fig. 15 ).
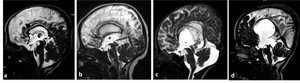
Fig. 14: Midsagittal 3D-DRIVE (a–d) exhibits variability of the third ventricle anatomy in cerebral atrophy and hydrocephalus secondary to aqueduct stenosis. a) A normal third ventricle of a patient with cerebral atrophy, the lamina terminalis and tuber cinereum are concave centrally which may suggest that the mean pressure in the third ventricle may be lower than in the cisterns; b) the appearance of the third ventricle in mild hydrocephalus secondary to multifocal aqueduct stenosis; c) Another case with more dilatation of the third ventricle secondary to congenital aqueduct stenosis (note the proximal funnelling); d) sever hydrocephalus with effacement of the chiasmatic and suprasellar recesses of the third ventricle, note dilatation of the pineal recesses
References: Mohammad S
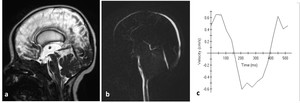
Fig. 15: 3-month-old boy with hydrocephalus. a) sagittal 3D-DRIVE demonstrates aqueduct stenosis; b)phase-contrast MRI shows absence of detectable flow at the region of aqueduct with markedly diminished velocity (c).
References: Mohammad S
Communicating hydrocephalus
· Communicating hydrocephalus implies cases with no obstruction in the ventricles or the cisterns.
Aqueductal CSF flow void is increased in communicating hydrocephalus plausibly because of decreased intracranial compliance ( Fig. 16 ) [14].
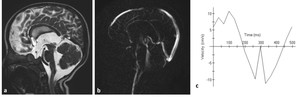
Fig. 16: MRI in communicating hydrocephalus reveals hyperdynamic CSF flow through thee aqueduct of Sylvius. a) sagittal 3D-DRIVE demonstrates flow-void through the aqueduct of Sylvius. b) phase-contrast MRI of another patient demonstrates prominent CSF flow through the aqueduct of Sylvius with accelerated CSF flow velocity (c)
References: Mohammad S
· In children,
apparently non-obstructive hydrocephalus has been attributed to an imbalance of CSF secretion and absorption.
Two patterns have been identified: the benign idiopathic external hydrocephalus (which is more common) and the choroid plexus papilloma-associated hydrocephalus (which is much rarer) [14].
· Benign idiopathic external hydrocephalus is also known as benign enlargement of subarachnoid spaces in infancy.
It is characterized by macrocephaly with dilatation of the anterior subarachnoid spaces and ventricular system to a lesser extent.
The extra-axial CSF spaces should have CSF signal in all pulse sequences and be traversed by the bridging veins (Fig. 7).
These features should be fulfilled in order to be differentiated from prominent extra-axial spaces secondary to haemorrhage especially in patients who have macrocephaly associating glutaric aciduria type 1 ( Fig. 17 ) [15].
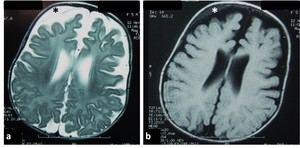
Fig. 17: Axial T2-WI (a) and FLAIR (b) of 5-month-old girl with glutaric aciduria type I and macrocephaly showing ventriculomegaly with subdural collection (asterisk)
References: Mohammad S
CSF disorders associating posterior fossa malformations
- Dandy-Walker spectrum of malformations
Dandy-Walker complex represents a continuum spectrum of posterior fossa malformations that is characterised by a combined posterior fossa cyst associated with variable degrees of cerebellar vermis abnormal development.
Classic Dandy-Walker malformation,
Blake’s pouch cyst,
posterior fossa arachnoid cyst and mega cisterna and vermian hypoplasia represent steps on this continuum.
Phase contrast sequence has value in studying CSF flow between CSF spaces in the posterior fossa and the posterior cervical subarachnoid space [16].
The typical Dandy-Walker malformation constitute large posterior fossa cyst communicating with the fourth ventricle in association with partial or complete agenesis of the cerebellar vermis.
Phase-contrast study demonstrates no flow between the posterior fossa cystic space and the posterior cervical subarachnoid space (Fig. 18 ). Hydrocephalus is associated with typical Dandy-Walker malformation in about 90% of patients.
In such conditions,
the posterior fossa cyst can be shunted directly.
In addition,
aqueduct stenosis is frequently encountered in association with Dandy-Walker malformation (Fig. 18 ) and separate ventriculoperitoneal shunt should be placed [16].
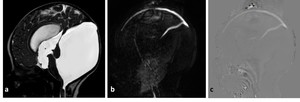
Fig. 18: MRI of 3-month-old boy with Dandy-Walker malformation and hydrocephalus, a) 3D-DRIVE showing large posterior fossa, cerebellar hypoplasia and stenosis of the aqueuduct of Sylvius hydrocephalus, (b,c) phase-contrast sequence showing absence of flow through the aqueduct of Sylvius with no communication between the posterior fossa cyst and the posterior cervical subarachnoid space
References: Mohammad S
It appears as Infra or retrocerbellar arachnoid cyst associated with tetraventricular hydrocephalus.
Phase-contrast study reveals lack of communication between the cyst and the posterior cervical subarachnoid space (Fig. 19).
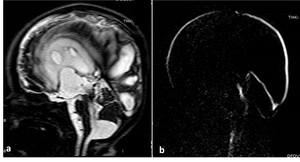
Fig. 19: 12-month-old girl with Blake’s pouch cyst. 3D-DRIVE showing infracerebellar cyst associated with dilated third and fourth ventricles. B)phase-contrast MRI reveals absence of communication between the cyst and the posterior cervical subarachnoid space
References: Mohammad S
It represents caudal displacement of the cerebellar tonsil through the foramen magnum causing plugging of CSF pathway (figure 20).
Patients who are most responsive to surgery are better selected according to the degree of CSF flow obstruction,
demonstrated by phase contrast study,
rather than the degree of cerebellar tonsil ectopia [2,
17].
Follow up after third ventriculostomy
Phase-contrast and 3D-heavy T2 (3D-DRIVE) sequences have high confidence in the assessment of the flow through the floor of the third ventricle with proper identification of ventriculostomy malfunction (Fig. 21 ,
video Fig. 22)[18].
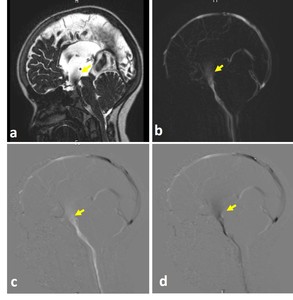
Fig. 21: 9-year-old girl underwent ventriculostomy. a) 3D-DRIVE demonstrates flow-void through a defect in the third ventricular floor (arrow). b) phase-contrast MRI reveals flow jet through the ventriculostomy (arrow). c,d) phase images through systole and diastole demonstrates the pulsatile flow throughout the cardiac cycle (arrow).
References: Mohammad S
Follow up after ventriculoperitoneal shunt
CT of the head and clinical symptoms,
including headache,
vomiting,
altered sensorium,
feeding difficulty,
and fever,
remain the main stay for the diagnosis of shunt malfunction [19].
However,
clinical symptoms are often vague with equivocal radiological findings,
in addition to the burden of frequent radiation exposure.
Ventricular enlargement compared to prior examinations is the primary sign for shunt malfunction.
However,
the shunt failure can occur without definite ventriculomegaly.
Secondary signs of acute shunt malfunction include transependymal permeation of CSF and pericatheter edema.
Subgaleal fluid collections can be encountered (Fig. 23 ) [20]. Rapid brain MRI protocols have been utilized based on single shot T2-WI [21].
Recently,
it was documented that phase-contrast sequence can be useful in the assessment of functional status of the VP-shunt through velocity measurements across the shunt tube [22].
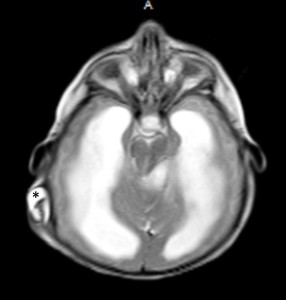
Fig. 23: Axial T2-WI of 6-month-old girl with hydrocephalus and non-functioning ventriculoperitoneal shunt demonstrates fluid surrounding the shunt tube (asterisk).
References: Mohammad S