Methods for the diagnosis:
abdominal x-ray: It is the initial request for diagnostic imaging in patients with acute abdominal pain,
but it has a limited role in the diagnosis,
especially at the beginning of the picture.
An X-ray without findings does not exclude the diagnosis of MI.
In mesenteric venous thrombosis,
dilated loops of the small intestine,
hydro-aerial levels,
and thumb impression images (due to wall edema) are observed.
In some cases,
intramural pneumatosis can be seen if it is present or gas in the mesenteric or portal vein,
strongly associated with intestinal ischemia.
Ultrasound: provides indirect data such as thickening of the wall of the intestinal loops,
in the MI by venous thrombosis.
The use of Doppler Color can sometimes provide data on the arterial flow of the wall.
Sometimes the endoluminal thrombus of the superior mesenteric artery can be seen.
It is a method that depends on the trained operator and the examination is usually difficult due to the interposition of the intestinal gas.
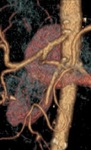
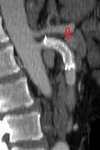
CTMS: Multislice computed tomography demonstrated high sensitivity and specificity for diagnosis,
making it the technique of choice when MI is suspected.
Study technique: Injection of 100_130 ml of water-soluble iodinated contrast intravenously,
with a flow of 2-4 ml / s. The cuts are thin and continuous of 1-2 mm.
A phase without contrast injection should be performed,
followed by an arterial phase with contrast injection at 30 seconds and a portal phase at 60 seconds.
Mesenteric vessels:
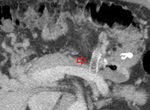
Arterial embolism is observed as a lack of filling in the lumen of the mesenteric artery.
In case of obstruction,
the cut-off sign (abrupt termination of the artery due to an embolism) or obstruction in the form of meniscus is observed.
Nonocclusive emboli are visualized as filling defects in the lumen of the vessel.
No collaterals are observed,
or very few,
if present.
Most of the time,
the plunger impacts near the origin of the middle colic artery.
In the case of thrombosis of the superior mesenteric artery,
the repletion defect is located at the origin of the artery (2 first centimeters) and is typically associated with a preexisting atherosclerotic lesion.
In these cases,
collateral circulation is usually observed.
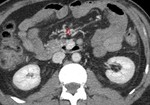
In the case of mesenteric venous thrombosis,
the vein that has the endoluminal thrombus is enlarged,
it can have poorly defined margins,
with low central attenuation.
There is engorgement of the mesenteric venous vessels.
In the case of mesenteric dissection,
the dissection flap in the artery is observed.
In the aortic dissection,
the dissection flap of the aorta can be observed,
with two lights,
a true and a false one,
and the involvement of the mesenteric artery,
partial or total,
by the myointimal flap.
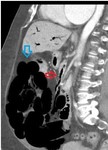
Abnormalities of the intestinal wall: Thickness of the intestinal wall:
The thickness of the normal intestinal wall is 3 to 5 mm.
Thickening of the wall is the most frequently observed finding in the CTMS of MI,
but it is not specific.
It is caused by mural edema,
hemorrhage or superinfection of the intestinal wall.
It is common in venous thrombosis,
obstruction with strangulation,
ischemic colitis and also in mesenteric arterial occlusion after reperfusion.
The greater the thickening,
the greater the severity of MI.
The thickening seen in cases of mesenteric venous thrombosis the wall measure 8-9 mm when there is ischemia,
being able to reach 15 mm.
In ischemia due to arterial occlusion,
a thinning of the intestinal wall (paper-thin Wall) is observed due to the loss of muscle tone and the volume of the parietal vessels.
Transmural necrosis has occurred.
Attenuation:
CTMS without contrast injection: the hypodense wall indicates edema of the wall,
frequent finding in MI due to mesenteric vein thrombosis.
The hyperdense wall is caused by intramural hemorrhage.
CTMS with intravenous contrast: no enhancement or decreased wall enhancement is a very specific finding of ischemia (although not sensitive).
It is associated with the non-vascularization or poor vascularization of the wall by the arteries due to occlusion.
The non-enhancement indicates intestinal infarction,
irreversible situation.
Another suggestive sign of ischemia is the halo or target sign of the wall,
which is produced by hyperemia and hyperperfusion associated with mural edema.
Intraluminal bleeding can be found in venous thrombosis.
Greater wall enhancement can be seen by increased venous capillary pressure and venous engorgement.
It represents an early sign of ischemia and indicates reversibility.
NOMI: The contrasted face shows a superior mesenteric artery of small caliber and the mesenteric veins with delayed opacification.
Mesentery:
Mesenteric edema and ascites appear due to the transudation of the fluid in the mesentery or peritoneal cavity caused by the elevation of the mesenteric venous pressure.
(Can be seen in venous thrombosis and intestinal oclusion) In patients with mesenteric arterial occlusion,
fat infiltration occurs when a transmural infarction occurs and is a useful finding to estimate the severity of ischemia.
Dilation of intestinal light It is produced by the interruption of normal intestinal peristalsis (paralytic ileus) and is nonspecific.
Distention of bowel loops with liquid content is produced by increased intestinal secretions,
ischemia due to venous occlusion and intestinal obstruction In ischemia due to intestinal occlusion,
the intestine rarely contains a large amount of endoluminal fluid.
Luminal air:
Intestinal pneumatosis: the air found in the wall of the intestine.
In the context of MI,
it is a late and specific sign of transmural necrosis of the wall (especially if it is associated with gas in the portal and mesenteric veins).
It is produced by the discontinuity of the wall,
with the passage of air to the outermost layers and to the intramural veins.
Middle arcuate ligament: 3D sagittal and volumetric reconstructions are the most suitable for the evaluation of the focal stenosis of the celiac trunk,
this compression site has the characteristic "hook" appearance,
located 5 mm from the aortic origin,
which helps to distinguish it from other causes of stenosis of the celiac trunk,
such as atherosclerosis.
The tomography may also show posterior dilation of the tendon and collateral circulation,
mainly through the duodenal pancreatic arch.
This information can help determine the hemodynamic importance of stenosis and the importance in surgical planning.
In many cases,
it is possible to identify the median arcuate ligament.
Occasionally,
collateral circulation is visualized.
MRI: It has a high sensitivity and specificity in the diagnosis of myocardial infarction,
but does not offer advantages with respect to CTMS,
except in patients with iodine intolerance.
Treatment:
MI treatment is based on correcting the cause and alleviating the consequences.
The type of treatment depends on the clinical presentation,
with time as the most important factor to determine the viability of the irreparable damage to the intestine.
In MI,
intestinal viability is at risk and surgical intervention is sometimes required to access bowel loops to seek infarction and revascularize the bowel,
if possible.
Endovascular procedures are more appropriate in patients with chronic mesenteric ischemia.
Follow-up with CTMS is recommended after the endovascular intervention,
in search of post-intervention complications.
Embolectomy and thrombectomy: is the treatment of choice in MI secondary to embolism of the superior mesenteric artery.
Acute complications are the presence of residual emboli,
occlusion of the distal branches and dissection.
Later,
stenosis can be observed in arterioctomy or diffuse stenosis due to intimal injury during embolectomy.
Endarterectomy is the surgical removal of a plaque from a blocked or narrowed artery due to chronic atherosclerosis.
It is performed when the atherosclerosis of the ostium of the visceral artery makes it difficult to perform the bypass.
Also in cases of intestinal perforation.
by pass: they can be anterograde (from the supra-celiac abdominal aorta) or retrograde (from the abdominal aorta,
infra celiac,
common iliac arteries),
venous or synthetic,
from a single vessel or from multiple vessels.
Bypass complications can be detected with the use of CTMS,
as early signs of intestinal ischemia and other complications such as active bleeding.
The most frequent complications are: kinking,
intimal hyperplasia and occlusion.
The retrograde are more predisposed to wriggle and the antegrade is associated with renal ischemia because of the need to hold the aorta above the renal arteries.
All bypasses are predisposed to present stenosis due to intimal hyperplasia,
especially in the sites of anastomosis.
Treatment of arcuate ligament syndrome: surgical.
It consists of removing the ligament with or without the placement of a bypass.
Endovascular therapy: They include: catheter-directed thrombolysis,
mechanical thrombectomy,
angioplasty,
stenting,
stent-graft endoluminal placement.
Stent placement: They have less recurrence of symptoms and less presence of infarcted organs,
after placement.
The follow-up must be with CTMS because the stent can make an artifact in the IMR,
which makes its inspection difficult.
At follow-up,
intimal hyperplasia and restenosis can be observed,
the latter when untreated leads to thrombosis.
Hyperplasia is visualized in the CTMS as hypodense tissue on the internal surface of the stent (in the non-contrasted phase).
Stent migration and fracture are other uncommon complications that can also be detected during follow-up.
Endovascular treatment of aortic dissection: the implantation of an aortic endoprosthesis,
the placement of a stent in the visceral branches or the fenestration of the intimal flap,
allow the reestablishment of the flow in the true lumen.