IMAGING DIAGNOSIS
CCF is a rare and difficult to diagnosis disease.
A few techniques may be used to diagnose this condition such as ultrasonography (US),
computed tomography (CT),
magnetic resonance imaging (MRI) and digital substraction angiography (DSA).
COMPUTED TOMOGRAPHY (CT)
Represents a non-invasive cross-sectional image modality that can be an useful tool in the early diagnosis of these arteriovenous fistulas.(9) It can detect dilatation or thrombosis of the superior ophthalmic vein (SOV),
edema of the periorbital fat and thickening of the extraocular muscles.
Postcontrast phase CT imaging may visualize an ectatic SOV,
which can be one of the most important signs of a cavernous sinus arteriovenous fistula.
(13) Another CT imaging sign to be considered is arterial phase postcontrast enhancement of the cavernous sinus.
(9)
Indirect CCFs (low-flow fistula) and direct CCFs (high-flow fistula) may be distinguished by the clinical history and the contrast enhancement.
Besides cavernous sinus dilation,
dural CCFs will have slower contrast uptake,
while direct CCFs will have an increased and exuberant uptake.
CT is one of the most useful techniques in detecting the complications of CCFs,
such as intracranial hemorrhage that may be caused by bleeding of ectatic veins.
(13)
4D-CTA (4D-COMPUTED TOMOGRAPHY ANGIOGRAPHY) has a high accuracy in the diagnosis of CCFs.
The advantage in using 4D-CTA is that it can evaluate small vascular malformations,
it gives temporal information and low-flow fistulas can be more accurately described using this technique than with conventional CTA,
or MRI.
(8)
Fig. 2 Fig. 3
MAGNETIC RESONANCE IMAGING (MRI)
Represents a non-invasive technique that can be used to more effectively evaluate the intra- and periorbital soft tissue than with CT imaging.
(9)
The most common changes in a CCF that are detected with MRI are ectatic SOV,
evidenced with T2-weighted sequences and post-contrast sequences.
In direct CCFs the cavernous sinus appears dilated and presents faster contrast enhancement while in indirect CCFs the cavernous sinus has a slow contrast enhancement.
The cavernous sinus presents early contrast enhancement even in low-flow fistulas that makes MRI investigation superior to CT imaging,
in which low-flow arteriovenous shunts are not usually detected.
When the cavernous sinus arteriovenous fistula drains posteriorly into the inferior petrosal sinus (IPS),
the diagnosis may be delayed because the patient may present with headache and not the typical ocular symptoms such as those found usually in CCFs which have a predominant drainage in the SOV.
MRI can provide detection even in these posterior drainage patterns.
(13)
Comparative to normal cavernous sinuses,
in CCFs the cavernous sinus has decreased signal intensity in spin-echo sequences that is caused by the rapid flow due to the arteriovenous communication.
The differential diagnosis of the loss of signal in the cavernous sinus should include besides CCFs,
aneurysms of the ICA in the cavernous segment and other arteriovenous malformations.
(10)
3D Time-of-flight (TOF) MRI is very important in detection of CCFs by visualization of direct flow related hyperintensity with a sensitivity of 83% and 100% specificity,
thus being superior to standard MRI sequences.
(14)
MRI,
as other non-invasive techniques,
is unable to accurately describe CCF arterial sources or cortical venous drainage,
but is a valuable tool in pre-angiographic diagnostic decisions.
(15)
DIGITAL SUBSTRACTION CEREBRAL ANGIOGRAPHY (DSA)
Represents an essential tool for the diagnostic confirmation of a CCF.
This technique describes the anatomy,
the location and the classification of the fistula.
DSA detects the fistula,
identifies the patterns of drainage and the flow of the cavernous sinus and other venous sinuses.
The venous drainage into SOV or IPS,
reflux into cortical veins or complications such as possible thrombosis are identified with this method.
ICA angiography ipsilateral to the lesion is necessary for detection of the arteriovenous fistula and the presence or absence of vascular steal,
while also describing drainage venous pathways.
The treatment of a CCF is based principally on the information given by DSA.
(13,
3)
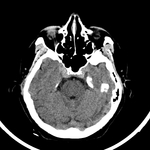
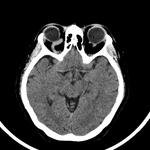
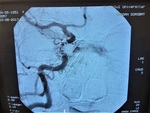
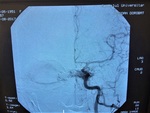
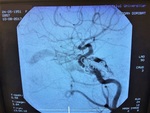
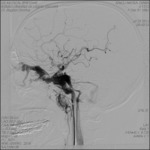
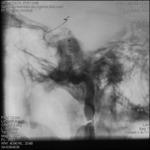
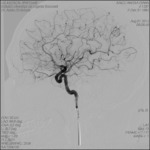
Case 2: A 29 year-old female,
with recent history of head trauma,
who progressively developed ptosis,
diplopia,
headache and tinnitus.
The cerebral angiography showed the presence of high-flow CCF on the left side.
Fig. 4 Fig. 5 Fig. 6 Fig. 7 Fig. 8 Fig. 9
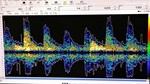
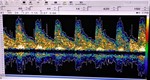
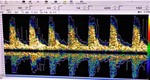
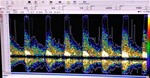
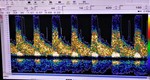
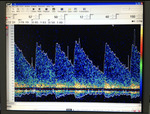
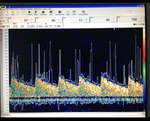
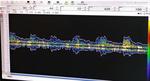
ULTRASONOGRAPHY
With the development of cerebrovascular ultrasonography a non-invasive diagnostic method was made possible for intracranial vascular diseases which could have been confidently revealed only with the use of cerebral angiography.
Techniques such as Color Doppler Imaging (CDI) and Transcranial Color Doppler Imaging (TCDI) have increased the role and confidence in ultrasonography in the diagnosis of these pathologies.
(16)
Color Doppler Imaging represent an ultrasonographic method that evaluates qualitative (direction of flow) and quantitative (resistance indexes,
velocities) the blood flow by combining traditional B-mode ultrasound imaging with simultaneous real-time spectral Doppler analysis.(4,
5)
CDI represent a non-invasive method that can describe the hemodynamic changes that appear in the orbital vasculature that may be predictive for a CCF.
The orbital vasculature can be topographically described and visualized in dynamic real-time and measured with great accuracy with the help of ultrasonography.
(4)
Ultrasonography has same advantages over other ways of investigation of a suspected CCF such as computed tomography,
magnetic resonance imaging or digital substraction angiography by being a quick,
cost-effective and dynamic method and may be repeated as frequently as needed,
also does not necessitate contrast material for diagnosis and eliminates the exposure of the patient to the risks carried by these methods.
(4,6,7).
These other modalities CT,
MRI give important topographic descriptions but may be insensitive in the diagnosis of low-flow fistulas as they cannot evaluate active blood flow or the direction of flow.
(4)
CDI is an important tool in the diagnosis and classification of a CCF.
The anatomic classification made by Barrow et al.
based on angiographic features is also used for classification in ultrasonography.
(16)
There are some important signs that may be observed with the use of ultrasonography and may be predictive for a CCF; they can be classified in direct and indirect ultrasonographic sings.
The direct sign evidenced with transcranial color-coded duplex sonography (TCCD) is the appearance of a mosaic flash.
(16) The indirect signs are: the dilatation of the superior ophthalmic vein with “arterialized” blood flow,
increase of blood flow velocities in these veins with a low resistance index.(3,
4,
5)
When TCCD detects only indirect ultrasonographic signs of CCF in a patient,
this can be predictive for an indirect type of CCF.
A direct type of CCF may be presumed to exist when TCCD detects direct ultrasonographic signs such as when the feeding artery has flow volume markedly increased and arterial indirect signs present in the ICA.
A mixed type of fistula may be considered when direct signs of a CCF and arterial indirect signs in the ICA and ECA are evidenced.
Both direct and indirect signs on TCCD are found when direct shunting between the carotid artery and the cavernous sinus is present.
(16)
In direct,
high-flow CCFs,
B-mode scans can reveal a dilated superior ophthalmic vein,
but in low-flow fistulas,
the diameter of this vein may be under the resolution of B-mode scan,
therefore without color information,
duplex scanning may not be able to detect vascular changes.(5)
Ultrasonographic techniques may detect a carotid aneurysm superimposed on CCF,
which is a rare combination,
but very dangerous because of the catastrophic hemorrhage that may occur in its rupture.
The detection of an aneurysm with TCCD varies,
depending on the dimension,
the location and if the aneurysm is thrombosed or not.
The presence of a 2-colored oval mass separated by a dark zone may differentiate a direct CCF and direct CCF superimposed with an aneurysm.
(16)
CDI has a very important role in the differential diagnosis between a CCF and a superior ophthalmic vein thrombosis,
in the latter the vein appears dilated but blood flow can’t be detected.
(5)
The pathological features evidenced by TCCD are most frequently seen using the transorbital window,
followed by the transtemporal window and the transforaminal window.
(16)
The transorbital window may reveal engorgement of the superior ophthalmic vein and other draining veins with reversal of flow direction and sometimes high velocity,
turbulent flow with a low resistance pattern.
The transtemporal window may show color mosaic flashes with the cavernous sinus at a depth of approximately 7 cm located above the carotid artery.
Within the flashes,
unidirectional or bidirectional turbulent and low-resistance blood flow may be shown.
The transforaminal window may describe dilated vessels leaving the cranium.
(16)
CDI and TCCD may be very useful in the planning of treatment of each type of CCF,
in the follow-up of patients which present with symptom change or new onset symptoms,
in the evaluation of patients suspected of a CCF and for selection of patients for additional investigations.
(16)
Ultrasonography may confirm the diagnosis of a CCF,
especially a low-flow fistula (as angiography may carry more risks than benefit),
or for patients who are conservatively treated,
or when we expect a spontaneous closure of a CCF.
(5) If the fistula closes either spontaneously or after embolization,
all hemodynamic changes may disappear.
The clinical findings may completely regress after the effective treatment of the CCF.(5)
After embolization,
CDI may be a safe and fast technique that may show if the treatment was efficient,
thus reestablishing a normal laminar flow in the superior ophthalmic vein.
(5,6)
Another important quality of CDI is that it can be used in the diagnosis of a CCF when the patient has contraindications to angiography,
for instance in atopic patients allergic to contrast agents.
(4)
As every technique,
ultrasonography has limitations; it is technology dependent and when alterations in hemodynamic parameters are not prominent,
CDI can’t accurately differentiate between the types of indirect CCFs.
(16)
Fig. 10 Fig. 11 Fig. 12 Fig. 13 Fig. 14 Fig. 15 Fig. 16 Fig. 17
Case 3: A 66 year-old female diagnosed with right CCF.
Fig. 18 Fig. 19