A total of 94,960 examinations with iobitridol (53.1% male,
46.9% female,
mean (±SD) age 58.8 ± 15.6 years) were carried out in 168 centres,
predominantly CT examinations (49.3%) or IV urography (48.8%).
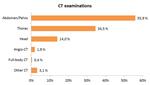
The most common CT examinations were of the abdomen/pelvis (55.9%),
thorax (34.5%) and head (14.0%) [Fig.
1].
For the CT examinations,
the mean (±SD) volume of contrast agent administered was 89.9 ± 19.9 mL with an injected volume between 50 and 120 mL in 90% of the patients [Fig.
2].
Patients who underwent IV urography were administered a mean volume of contrast agent of 83.4 ± 17.1 mL.
In 90% of cases the volume administered was between 50 and 100 mL.
At least one risk factor was documented for 29,290 of the 94,960 patients (30.8%),
most commonly hypertension (11.0%),
known allergies (9.7%),
diabetes mellitus (6.7%) or thyroid dysfunction (5.5%) [Fig.
3].
Adverse events occurred  in 469 of the 94,960 patients (0.5%).
For these 469 patients,
a total of 707 adverse events (non-serious and serious) and 103 different preferred terms (PT) were documented.
The most common adverse events were nausea (140 patients,
0.1%),
urticaria (69 patients,
<0.1%),
dizziness (42 patients,
<0.1%) and rash (38 patients,
<0.1%) [Table 1].
in 469 of the 94,960 patients (0.5%).
For these 469 patients,
a total of 707 adverse events (non-serious and serious) and 103 different preferred terms (PT) were documented.
The most common adverse events were nausea (140 patients,
0.1%),
urticaria (69 patients,
<0.1%),
dizziness (42 patients,
<0.1%) and rash (38 patients,
<0.1%) [Table 1].
Serious adverse events occurred in 24 of 94,960 patients (0.03%).
For these 24 patients,
a total of 65 serious adverse events and 41 different preferred terms were documented.
The most frequently reported were nausea and dyspnea (5 patients),
urticaria (4 patients),
feeling hot,
respiratory distress,
flushing (3 patients),
eyelid oedema,
lip oedema,
retching,
paraesthesia oral,
hypersensitivity,
erythema,
circulatory collapse (2 patients).
All other preferred terms were reported for one patient each: anaphylactic shock,
anxiety,
arrhythmia,
blister,
cardiovascular disorder,
chest pain, cold sweat,
contrast media allergy,
convulsion,
dermatitis allergic,
dizziness,
dry eye,
dysgeusia,
dysphagia,
extravasation,
hypertensive crisis,
larynx irritation,
paraesthesia,
pruritus,
rhonchi,
sneezing,
swelling face,
tenderness,
tongue oedema,
tremor,
type IV hypersensitivity reaction,
upper airway obstruction,
vomiting projectile.
Physicians assessed the causal relationship with iobitridol as highly probable in 16 of the 24 patients with serious adverse events (66.7%),
probable in 6 patients (25.0%) and unlikely in one patient (4.2%).
In one patient (4.2%),
the causal relationship could not be evaluated.
After the examination,
22 of the 24 patients with serious adverse events (91.7%) had recovered and in 2 patients (8.3%) the outcome was unknown.
The doctors were able to make a diagnosis in 99.6% of the patients (CT examination 99.9%,
IV urography 99.3%),
and the imaging quality was evaluated as being excellent or good for 96.6% of the patients (CT examination 97.9%,
IV urography 95.3%).
Table 1: Most Common Adverse Events (Preferred Terms)
|
|
Total
|
CT examination
|
IV urography
|
|
|
N
|
%[1]
|
N
|
%[1]
|
N
|
%[1]
|
|
Nausea
|
140
|
29.9
|
65
|
25.5
|
72
|
34.6
|
|
Urticaria
|
69
|
14.7
|
40
|
15.7
|
29
|
13.9
|
|
Dizziness
|
42
|
9.0
|
15
|
5.9
|
26
|
12.5
|
|
Rash
|
38
|
8.1
|
25
|
9.8
|
13
|
6.3
|
|
Erythema
|
33
|
7.0
|
20
|
7.8
|
13
|
6.3
|
|
Pruritus
|
30
|
6.4
|
12
|
4.7
|
17
|
8.2
|
|
Vomiting
|
30
|
6.4
|
22
|
8.6
|
8
|
3.8
|
|
Sneezing
|
23
|
4.9
|
17
|
6.7
|
5
|
2.4
|
|
Feeling hot
|
23
|
4.9
|
7
|
2.7
|
16
|
7.7
|
|
Flushing
|
22
|
4.7
|
9
|
3.5
|
13
|
6.3
|
|
Dyspnoea
|
18
|
3.8
|
14
|
5.5
|
4
|
1.9
|
|
Retching
|
13
|
2.8
|
6
|
2.4
|
7
|
3.4
|
|
Cough
|
12
|
2.6
|
8
|
3.1
|
4
|
1.9
|
|
Throat irritation
|
11
|
2.3
|
8
|
3.1
|
3
|
1.4
|
|
Contrast media allergy
|
9
|
1.9
|
2
|
.8
|
7
|
3.4
|
|
Injection site extravasation
|
9
|
1.9
|
8
|
3.1
|
1
|
.5
|
|
Tremor
|
8
|
1.7
|
4
|
1.6
|
4
|
1.9
|
|
Hypersensitivity
|
7
|
1.5
|
3
|
1.2
|
4
|
1.9
|
|
Paraesthesia oral
|
7
|
1.5
|
4
|
1.6
|
3
|
1.4
|
|
Rash macular
|
6
|
1.3
|
3
|
1.2
|
3
|
1.4
|
|
Throat tightness
|
6
|
1.3
|
2
|
.8
|
4
|
1.9
|
[1] Percentages are calculated on the number of patients with adverse events. Multiple adverse events could be reported for the same patient.
Total column also includes AEs reported in patients for whom the type of examination was not reported.