A.Anatomic variants
Anatomic variants of the IVC are present in up to 4% of general population.
Although most of them are asymptomatic and incidentally encountered,
they should always be mentioned in the radiologic report; first,
because of their importance in the planning of vascular interventions and second,
because patients with anatomic variations are considered at higher risk of deep vein thrombosis of the common femoral and iliac veins,
which typically occurs at a younger age than those without anatomic IVC anomalies.
Anatomic variants of the IVC are consequence of alterations in the embryogenesis.
Therefore,
to understand them,
basic knowledge of the embryogenesis of this vessel is essential (Fig.3).
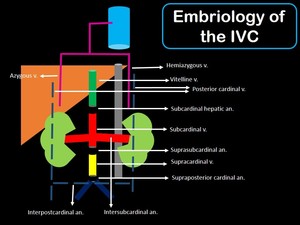
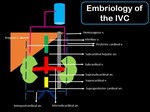
Fig. 3: Picture showing the embriology of the IVC, in which multiple venous channels regress (dashed lines), and other persist (solid lines) to form it.
Anastomotic channels (short black tubes) connect the different segments of the IVC.
Gray tube: abdominal aorta.
V: vein
An: anastomosis.
Formation of the IVC involves complex anastomoses and regression of multiple embryonic veins,
including the vitelline vein and the paired posterior cardinal,
subcardinal,
and supracardinal veins.
All of these embryologic processes,
will lead to the four segments of the mature IVC: the hepatic,
suprarenal,
renal,
and infrarenal.
-The hepatic segment of the IVC is formed by the vitelline veins.
-The suprarenal IVC is composed of a segment of the right subcardinal vein that does not regress.
-The renal segment of the IVC is formed by the anastomosis between the right subcardinal and right supracardinal veins.
-The infrarrenal segment is formed by a segment of the right supracardinal vein.
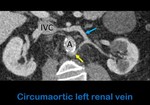
I.Circumaortic left renal vein
It is the most common IVC anomaly,
and it is present in up to 8% of people.
It is secondary to the development of two renal veins on the left from the intersubcardinal anastomosis.
One renal vein is in its normal position (anterior to the aorta),
whereas the other passes posteriorly to the aorta,
finding its way to the IVC at a lower position.
Compression of the renal vein as it passes posterior to the aorta is called the “nutcracker phenomenon”,
and can result in periureteric varices,
hypertension,
and hematuria (Fig.
4).
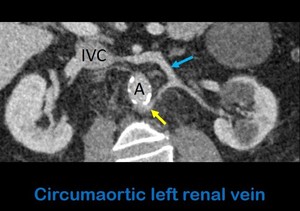
Fig. 4: Axial oblique contrast-enhanced abdominal CT shows a left renal vein with in its normal position, anterior to the aorta (blue arrow), and a renal vein with a retroaortic course (yellow arros). A: aorta. IVC: Inferior vein cava.
II.
Retrocaval ureter
In this condition,
the infrarenal IVC develops from the right posterior cardinal vein,
which lies anterior and lateral to the ureter.
As a result,
part of the right ureter becomes trapped posterior and medial to the IVC.
En route,
the ureter can undergo significant compression,
resulting in hydronephrosis or recurrent urinary tract infections.
The abnormal course of the proximal ureter as it projects over or medial to the lumbar pedicles on frontal images results in the characteristic “fish hook” or “reverse J” appearance.
III.
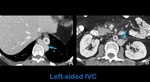
Left-sided IVC
It occurs due to persistence of the left supracardinal vein and regression of its right counterpart.
The infrarrenal IVC is at the left of the aorta up to the left renal vein,
where it crosses posterior to the aorta to join the right renal vein and form a normal right IVC above that level (Fig.
5).
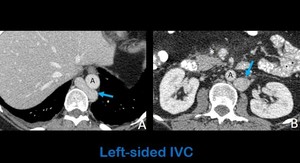
Fig. 5: Axial contrast-enhanced abdominal CT images in a patient with acute pancreatitis (asterisk in b shows the peripancreatic fat stranding) show a left-sided IVC that begins at the level of the diaphragm (arrow in a). Arrow in b shows a left-sided IVC on its suprarrenal portion. The suprahepatic veins drain directly to the atrium.
A: aorta.
IV.
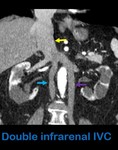
Double IVC
It occurs due to persistence of both supracardinal veins.
Both IVC usually join at the level of the renal veins,
even though variations are possible.
Double IVC should be sus- pected in cases of recurrent episodes of pulmonary embolism despite placement of an IVC filter (Fig.6).
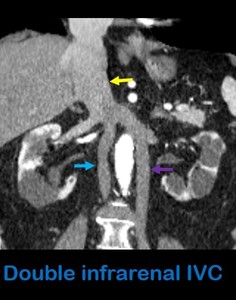
Fig. 6: Coronal contrast-enhanced abdominal CT shows a normal hepatic, suprarrenal and renal IVC (yellow arrow) and a double infrarenal IVC. Blue arrow: right-sided infrarenal IVC. Purple arrow: left-sided infrarenal IVC.
V.
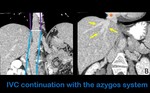
Azygos continuation of the IVC
In this anomaly,
there is failure of the subcardinal veins to anastomose with the vitelline veins.
The hepatic IVC is absent,
and blood from the renal IVC drains into the azygos system to empty in the superior vena cava.
Associated situs anomalies are present in many of these cases (Fig.
7).
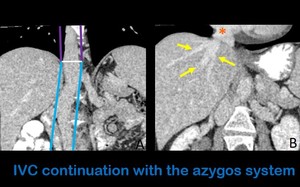
Fig. 7: Coronal contrast-enhanced images show the IVC continuation (between the blue lines) with the azygos vein (between the purple lines) (Figure A). The suprahepatic veins (yellow arrows) drain straight to the right atrium (orange asterisk) (Figure B).
VI.
Absence of IVC
Absence of the entire or part of the infrarenal IVC has an unknown incidence and unclear cause,
even though it is thought to be a sequela of intrauterine or perinatal thrombosis of the IVC and not truly embryologic in origin.
Affected patients are prone to develop deep venous thrombosis and chronic venous insufficiency.
Lower-ex- tremity venous return in these patients occurs via the ascending lumbar veins,
which drain into the azygous-hemiazygous system.
(Fig.8).
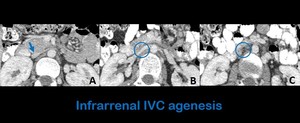
Fig. 8: Axial abdominal contrast-enhanced CT images show a normal IVC (inferior vein cava) at the level of the renal veins (figure a) and absence of this vascular structure below this level (figures b and c) in a patient with infarrenal IVC agenesis.
B- Thrombus
I.Bland thrombus
They are the most common type of thrombus to the IVC,
and the presence of a persistent filling defect within the column of contrast makes the diagnosis.
Even though they can occur isolated,
it is often that they are a consequence of the spread of deep vein thrombosis of the extremities.
Local compression of the IVC by adenopathies,
large retroperitoneal masses,
or fibrosis are known to promote local clot formation.
II.
Malignant thrombus
Tumoral thrombus to the IVC occurs as direct extension (more frequent) or compression by the main following malignancies: renal cell carcinoma (4-10% of cases),
adrenal carcinoma (30% of the right-sided cases),
hepatocellular carcinoma (5-10% cases) and by non-semynomatous tumors (by direct extension or compression by Bulky linfadenopathies).
In children,
they are most commonly secondary to Wilms tumor.
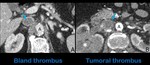
Tumor thrombus can be differentiated from bland thrombus on the basis of theenhancement of the filling defect,
which are typical features of malignant thrombus (Fig.9).
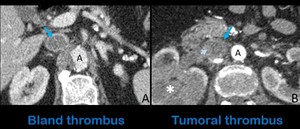
Fig. 9: Axial contrast-enhanced abdominal CT images show a bland thrombus (A) that lodges almost the whole circumference of the suprarenal IVC (blue arrow) in a patient with antithrombin III deficiency. Figure B shows a tumoral thrombus in the renal segment of the IVC (blue arrow) in a patient with renal cell carcinoma (white asterisk). Note the enhancement and expansion of the filling defect in B (light blue arrow) in comparison with the absence of enhancement in a.
A: aorta.
C.Neoplasms
I.Primary
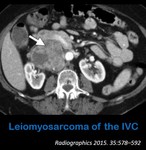
Primary tumors of the IVC are exceedingly rare,
and among them,
leiomyosarcoma is the most frequent one.
Leiomyosarcoma of the IVC is a tumor of mesenchymal origin arising from the smooth-muscle cells found in the vessel wall and is associated with a poor prognosis,
with a reported 10-year survival of 14%.
They can grow extraluminal,
appearing as large retroperitoneal masses with heterogeneous contrast enhancement,
or intraluminal,
appearing as a mass with variable contrast enhancement that focally dilates and obstructs the IVC (Fig.
10).
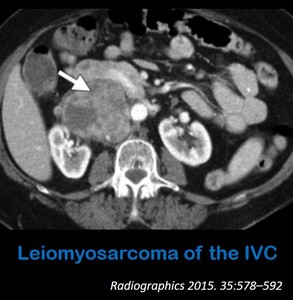
Fig. 10: Axial contrast-enhanced abdominal CT shows a mass inside the IVC (arrow) with heterogeneous enhancement and necrotic areas in keeping with a leiomyosarcoma of the IVC.
References: Smillie R. et al. (2015) Imaging Evaluation of the Inferior Vena Cava. Radiographics 35:578–592
II.
Secondary
They are usually a consequence of direct extension of abdominal neoplasms.
As previously mentioned,
renal cell,
hepatocellular,
adrenal cortical and non-semynomatous tumors are the most common to result in IVC invasion.
D.
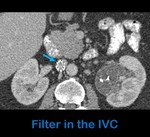
Devices and materials related to intervention
Multiple devices (such as IVC filters) and materials from procedures (i.e.
cement from vertebroplasties),
can be lodged inside the IVC.
MDCT is the most appropriate imaging technique to depict their exact location,
relationship with the renal veins and potential complications such as thrombosis migration,
and embedding in the wall of the IVC.
(Fig 11,
12).
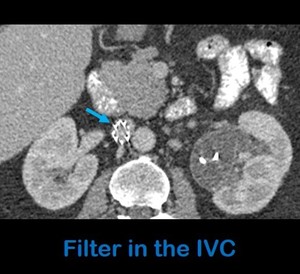
Fig. 11: Axial contrast-enhanced abdominal-CT shows a filter in the IVC (arrow).
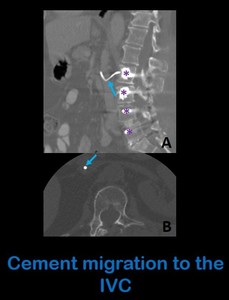
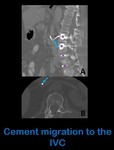
Fig. 12: Sagital (a) and axial (b) unenhanced abdominal CT images show cement (arrows) in the interior of the IVC, scaping from a lumbar arthrodesis (asterisks in figure a).
E.
Primary cardiovascular disease
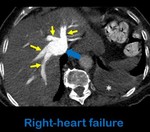
-Right heart failure: suprahepatic reflux of brightly enhanced blood from the right atrium to the IVC is associated with right-sided heart disease.
However,
this finding can also be seen at injection rates of more than 3 mL/s (Fig.
13).
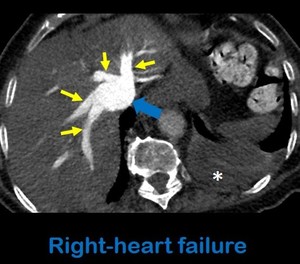
Fig. 13: Axial contrast-enhanced CT at the level of the superior abdomen shows an enlarged IVC (blue arrow) with reflux of contrast to the suprahepatic veins (yellow arrows) in a right-heart failure. Note the right pleural effusion (white asterisk).
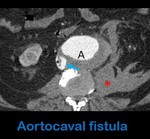
-Arteriovenous fistula: they are a rare but often catastrophic complication of abdominal trauma or abdominal aortic aneurysm.
Imaging findings include early enhancement of an enlarged IVC with absent fat plane of separation with the abdominal aorta (Fig.
14).
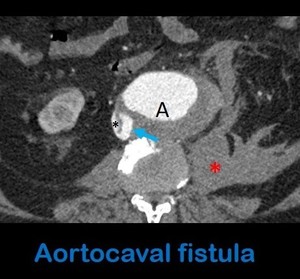
Fig. 14: Axial contrast-enhanced abdominal CT in arterial phase shows a fistulous communication (arrow) between the aorta (A) and the IVC (black asterisk), in a patient with an aortic aneurysm rupture (note the retroperitoneal hematoma (red asterisk)), consistent with an aortocaval fistula.
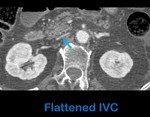
-Hypovolemic shock: a flattened or stil-like IVC (defined as a transverse-to-anteroposterior diameter ratio of 3:1 or more) can be a manifestation of hypovolemic shock.
In this setting,
other findings such as a surrounding hypodense halo and small caliber of the abdominal aorta,
which has a decreased caliber,
marked diffuse bowel distention,
moderate to extensive ascites,
hyperenhancement of the bowel wall,
kidneys,
suprarenal and pancreas ,
as well as hypoenhancement of the spleen constitutes the “hypoperfusion complex” and confirms the diagnosis.
A flattened IVC in an otherwise healthy patient with no other signs of hypovolemia is considered a normal variant,
which occurs above all in elderly women (Fig.
15).
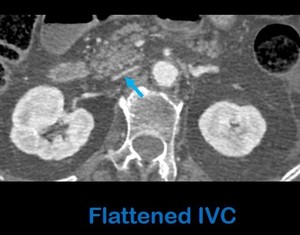
Fig. 15: Axial contrast-enhanced abdominal CT scan in portal phase in a patient with blunt abdominal trauma shows a flat inferior vena cava (arrow) indicating hypovolemia.
F.
Pitfalls
-Pseudolipoma: in patients with cirrosis,
prominent pericaval fat protruding the lumen may resemble lipoma inside the IVC.
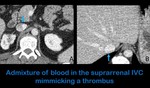
- Evaluation of the infrarenal IVC and its tributaries is more challenging because of the admixture of less opacified blood from the lower extremities,
which may resemble thrombosis (Fig.
16).
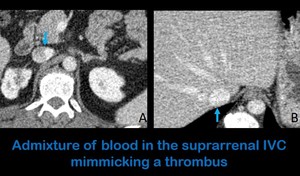
Fig. 16: Axial abdominal CT images in a portal phase (70 seconds of delay after the administration of intravenous contrast) show heterogeniety of the suprarrenal segment of the IVC (Figure A) due to the admixture of highly enhanced abdominal blood and the less opacificed blood from the lower extremities. This is a normal finding in the studies performed with 70 seconds of delay after the administration of intravenous contrast that should not be mistaken for a trhombus. Note the homogeneous enhancement of the hepatic portion of the IVC in the same patient (figure B).
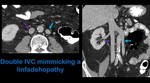
- Anomalies in the anatomy of the IVC,
above all those in which caliper is increased may be mistaken for linfadenopathies (Fig.
17).
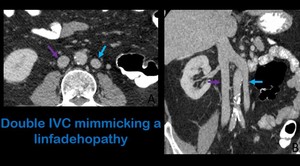
Fig. 17: Axial contrast-enhanced CT (A) shows a rounded and enhanced structure to the left of the aorta (blue arrow) that resembles an adenopathy. Coronal contrast-enhanced CT (B) proves that the rounded imaging in A corresponds to a left IVC in a patient with of double renal and infrarenal IVC. Purple arrow: right IVC.