1.
Acute complications
- Acute Cervicitis and Endometritis
One of the most common presentations of primary PID is cervicitis or endometritis.
Infection can also occur postpartum from vaginal delivery (3%) or post-caesarean section (85%),
usually from group B streptococcus.
Imaging is not frequently needed but if there is concern of possible complications (e.g.
retained products of conception Fig. 2) or presentation is atypical with a broad differential,
US,
CT or MRI can be used to aid diagnosis.
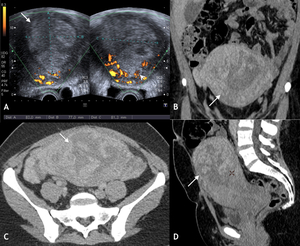
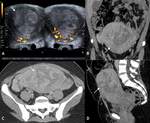
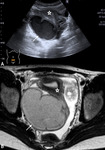
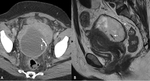
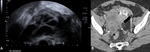
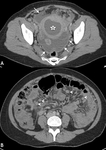
Fig. 2: Infected retained products of conception. 34 yr old lady presented 2 weeks post normal vaginal delivery with lower abdominal pain, per vaginal bleeding and fever. Duplex US (A) and portal venous phase contrast enhanced CT (Coronal B, Axial C and Sagittal D) demonstrated heterogenous material within a distended endometrial cavity (white arrow). Increased doppler signal was demonstrated in the myometrium and endometrium peripheral (white arrow head) (A). Features were in keeping with infected retained products. Microbiology samples confirmed the presence of multi-drug resistant E Coli.
Imaging features of acute cervicitis and endometritis may be subtle and are often non-specific [6,
7]:
US and CT:
- Mild enlargement of the cervix or uterus.
- Hyperaemic endocervical canal on Doppler or contrast enhanced imaging.
- Occasionally cystic changes in the endocervical lining,
but the deep cervical stroma is intact.
- Free fluid within the endometrial cavity.
- Gas locules in endometrial cavity which appear hyper-echoic on US and of low attenuation on CT.
- Parametrial fat stranding,
which is one of the most common findings seen in 65% of cases of acute PID but it is nonspecific.
- Trace of free fluid within the pelvis.
MRI has be used in some centres in the acute setting demonstrating [4]:
- Enlargement or thickening of the cervix,
myometrium,
endometrium,
utero-sacral ligaments.
- Increased T2 fat suppressed signal intensity in the cervix and myometrium compared to the normal low T2 smooth muscle signal intensity.
- Hyper-enhancement of the cervix,
myometrium and endometrium,
following intravenous contrast medium.
However,
imaging features alone are not definitive in diagnosing PID,
as other infections such as herpes simplex virus,
or post-traumatic / post-surgical changes can produce similar imaging findings.
Solid enhancing components within an abnormal cervix are suggestive of cervical malignancy rather than infectious cervicitis.
In the UK most acute cases are still diagnosed with good clinical history,
examination and swabs with appropriate antibiotics,
contact tracing and sexual health follow up.
- Salpingitis and Pyosalpinx
Salpingitis and pyosalpinx are a spectrum of PID,
which is characterised from fallopian tube thickening to distension with pus.
The fallopian tubes are not normally visualised on US,
CT or MRI imaging,
measuring between 1-4mm in diameter.
Salpingitis can be mild with subtle thickening (>5 mm) and features similar to endometritis on CT or MRI (Fig. 3) or US.
In 70-90% of cases of salpingitis,
there is coexistent endometritis [6].
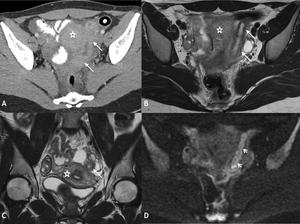
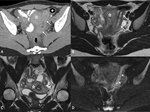
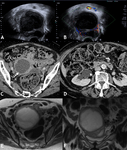
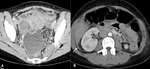
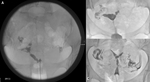
Fig. 3: Salpingitis in a 20 year old lady. Selected portal venous phase contrast enhanced axial CT with oral contrast medium (A) and T2 weighted axial (B) and coronal (C) slices of the pelvis demonstrating thickened left fallopian tube with mild enhancement on CT (white arrow) and free fluid. Mild restricted diffusion within the left fallopian tube on DWI, (ADC maps not shown) in keeping with infection (white arrow head). Diagnosis confirmed at laparoscopy where adhesions seen around the liver in keeping with Fitz-Hugh Curtis syndrome. Chlamydia infection confirmed on high vaginal swabs. Uterus = *
In pyosalpinx,
the fallopian tube is distended and therefore more easily identified as a serpiginous fluid filled structure (Fig. 4 and Fig. 5).
However appearances can be similar to hydro and haematosalpinx [6,
7].
Characteristic MRI appearances of pyosalpinx have a sensitivity of 91% [7] (Table 1).
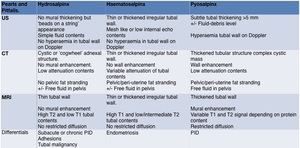
Table 1: Imaging characteristics of hydrosalpinx, haematosalpinx and pyosalpinx: Pearls and Pitfalls.
Salpingitis and pyosalpinx can be caused by primary or secondary PID.
Most common causes of secondary PID include appendicitis (Fig. 6) and diverticulitis.
In complicated cases,
with close approximation of bowel and gynaecological structures,
the primary organ of inflammation can be difficult to identify.
In a recent study,
the most discriminating finding on CT to differentiate appendicitis from primary PID,
was an appendix diameter >7 mm [8].
A dilated left fallopian tube or a tubal diameter > 10 mm was more in keeping with primary PID [6].
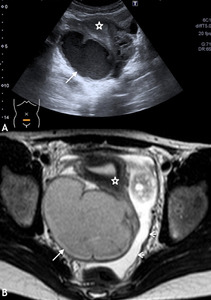
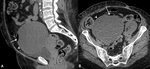
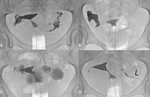
Fig. 6: Post-appendectomy: US (A) demonstrates a thick walled complex fluid collection posterior to the uterus (*) with low level internal echoes in a 15 year old girl. T2 weighted axial MRI sequences (B) demonstrates an intermediate signal intensity serpiginous structure (white arrow), in keeping with a tubo-ovarian abscess. High T2 signal intensity free fluid noted in pelvis. The collection was drained under US guidance (white arrow head).
- Tubo-ovarian abscess (TOA)
Adhesion of the tubal fimbria to the ovary allows infection to spread from the fallopian tubes to the ovary,
initially causing oophoritis but can lead to TOA.
It can occur in 15% of PID cases and is unilateral in 25-50% [6].
The ovary forms a complex cystic-solid mass with the fallopian tube and the two structures can no longer be differentiated.
On US,
a tender thick-walled,
multi-locular,
solid-cystic mass with increased vascularity may be demonstrated.
The cystic components may contain complex fluid with debris and free fluid may also be identified within the pelvis.
Similar appearances may be identified on CT and MRI (Fig. 7). On MRI,
the solid-cystic mass may have variable signal intensities on T1 and T2 weighted sequences depending on the presence of proteinaceous fluid and blood products.
Restricted diffusion may also be observed.
Uniform contrast enhancement of the cyst walls,
loss of fat planes,
thickening of the utero-sacral ligaments,
fat stranding and lymph node enlargement may also be seen on CT and MRI. Free fluid within the pouch of Douglas may be reactive,
or if complex,
may be secondary to rupture,
leading to peritonitis [4,
6,
7,
9]
Similar appearances occur with secondary PID especially with appendicitis (Fig. 6) and diverticulitis (Fig. 7 and Fig. 8) or even following procedures such as IVF ovum harvesting (Fig. 9) or perforation of the uterus by an IUD (Fig. 10).
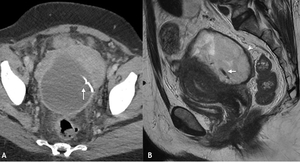
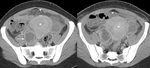
Fig. 10: A 50 year old patient presented with fevers and abdominal pain several weeks following a hysteroscopy and IUD placement for menorrhagia. Diverticulitis was suspected and a portal venous phase contrast enhanced CT was performed. A selected axial CT image through the pelvis (A) demonstrates an enhancing thick walled abscess cavity within the pouch of Douglas and surrounding fat stranding. The IUD was seen within the abscess cavity (white arrow) having perforated the posterior wall of the uterus. Sagittal T2 weighted MRI demonstrated a low T2 signal thick walled abscess cavity (white arrow head) with intermediate T2 signal fluid components and the low T2 signal IUD (white arrow). The abscess cavity was drained and IUD removed.
However,
non-gynaecological abscesses are often located further away from the adnexa and have been described as having ‘thicker walls’ and more likely to contain gas locules (Fig. 8).
Cases of inflammatory bowel disease causing secondary PID have also been reported [10].
These mainly occur in Crohn’s disease which is prone to fistulae and subsequent abscess formation.
However,
a fistula itself is not pathognomonic for Crohn’s disease as they have also been reported in chronic PID [7].
Other differentials for PID and TOA include endometriosis,
infected endometrioma (Fig. 11),
ovarian torsion and fallopian tube (Fig. 5) and ovarian malignancy.
Key radiological features are discussed in the PID mimics table (Table 2).
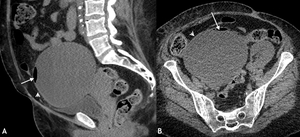
Fig. 11: Infected endometrioma. Selected sagittal (A) and axial (B) portal venous contrast enhanced CT images of the pelvis demonstrating a homogeneous right adnexal fluid attenuation lesion with a locule of gas anteriorly (white arrow). A thin wall (white arrow head) with no enhancement and minimal fat stranding were also noted. As the patient was septic with pelvic pain, she was treated with antibiotics. On subsequent MRI (not shown), signal characteristics confirmed an endometrioma but the gas locules had resolved and there were no MRI features of infection.
TOA can be successfully treated with antibiotic therapy.
Approximately 25% require drainage.
TOA that fail antibiotic therapy are often larger in size and patients have a higher mean leukocyte count and require drainage [11]. Surgery,
often resulting in salpingo-oophorectomy,
occurs more frequently in gastrointestinal-related TOA cases [12]. US guided drainage can also be performed via a trans-abdominal or trans-vaginal route (Fig. 12),
which has been shown to preserve patient fertility in 50% of patients with no increase in ectopic pregnancy rate [13].
Depending on the size of the abscess,
the TOA can be aspirated to dryness or a drain left in situ.
The drainage fluid is sent for microbiology to confirm the infective organism and guide antibiotic therapy.
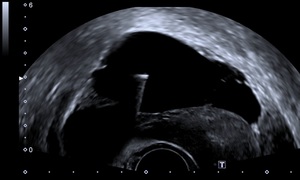
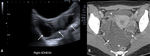
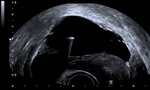
Fig. 12: Transvaginal ultasound guided drainage of pelvic collection: The transvaginal probe is positioned within the posterior fornix. The drainage needle is passed via a needle guide into the collection through the posterior fornix. On this image, the needle is seen as a linear hyper-echoic structure within the anechoic collection fluid. The uterus is anteverted and seen to the right of the image.
- Rarer Infections
- Actinomycosis
Actinomyces species are commensal gram-positive anaerobic organisms within the GI and vaginal tracts.
They have a predilection for the female genital tract and are associated with intrauterine devices (IUD).
They can cause hydrosalpinx,
pyosalpinx,
tubo-ovarian abscess or characteristically form fistula tracts by burrowing through tissue planes extending into the anterior abdominal wall,
perineum or even the upper abdomen (Fig. 13). Actinomycosis should be suspected when there is a relatively solid inflammatory pelvic mass,
which crosses fascial boundaries,
in patients with an IUD present [7] (Fig. 14).
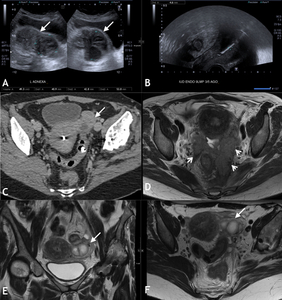
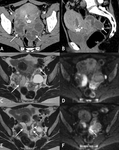
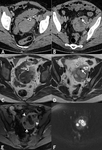
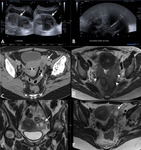
Fig. 13: 45yr old patient presented with pelvic pain. A IUD had been positioned 8 years previous for menorrhagia. Trans-abdominal (A) and trans-vaginal US imaging (B) demonstrates a complex septated left adnexal lesion with mixed echogenicity (white arrow). The hyperechoic IUD was identified within the uterine cavity (B). Selected axial portal venous phase contrast enhanced CT slice through the pelvis (C) demonstrates a peripherally enhancing left adnexal mass with a small fluid attenuation component (white arrow) and surrounding fat stranding. On MRI T2 weighted axial (D and F), and coronal (E) images of the pelvis, the cystic left adnexal lesion is seen with thick walls and central fluid component suggestive of tubo-ovarian abscess (white arrow). There was soft tissue seen around the sigmoid, crossing fascial planes in the pelvis and extending along the left uterosacral ligament (white arrow head). In addition, pelvic fat stranding and lymphadenopathy was noted. The presence of an IUD and the inflammatory soft tissue crossing fascial boundaries is highly suspicious for actinomycosis. On transvaginal aspiration of the inflammatory mass, sulphur granules were detected, confirming the diagnosis.
- Mycobacterium tuberculosis
Gynaecological Mycobacterium tuberculosis (TB) involvement is rare in the UK,
but can be more prevalent in endemic areas.
Gynaecological infection is via the haematological route.
The fallopian tubes are often involved in 94% of cases [7].
Acute TB salpingitis leads to multiple small granulomas forming on the wall.
Granulomas can cause multiple strictures or beaded appearance,
occlusion at the ampulla or isthmus or a rigid ‘pipe-stem’ on HSG imaging [7].
Chronic TB infection can result in peri-tubal adhesions and a persistent classical corkscrew appearance.
Contrast medium spilling from the fimbrial end can be trapped in adhesions and appear loculated and separate from the tube,
also known as the ‘halo sign’. The lesion can then calcify over time and may mimic a dermoid cyst or calcified leiomyomas [14].
Acute TB endometritis is less common and is characterised by irregularity of the endometrial cavity and intravasation of contrast into the vascular and lymphatic system.
Pearls for diagnosing TB infection compared to primary PID include [14];
- Appropriate exposure history or extra gynaecological features of TB.
- Fallopian tube obstruction between the isthmus and ampulla.
- Multiple stricture causing beaded or salpingitis isthmica nodosa appearances.
- Endometrial adhesions or distortion without other explanation.
- Calcified pelvic lymph nodes.
2.
Chronic Complications
- Fitz Hugh Curtis syndrome (FHC) (Fig. 15)
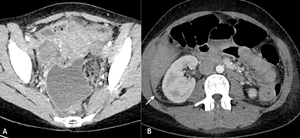
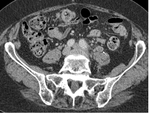
Fig. 15: Fitz-Hugh Curtis syndrome. IV portal venous phase contrast enhanced CT axial images of the pelvis (A) and abdomen (B) demonstrating free fluid and enhancing peritoneum within the pelvis (white arrow head) and trace of free fluid adjacent to the right lobe of the liver (white arrows). Chlamydia was confirmed on high vaginal swabs.
FHC presents with right upper quadrant pain secondary to peritoneal and peri-hepatic inflammation which can mimic acute hepatic or biliary pathologies.
Liver function blood tests are often normal. FHC occurs in 1-30% of cases of primary PID.
Imaging findings can be subtle on all modalities with a trace of fluid and fat stranding in the pelvis,
para-colic gutter and perihepatic region. On CT or MRI,
the anterior liver capsule may be thickened and demonstrates patchy or geographical contrast enhancement.
At surgery,
‘violin string’ adhesions can be found between the abdominal peritoneum and hepatic surface [4,
6,
7].
- Ovarian vein thrombophlebitis (Fig. 16)
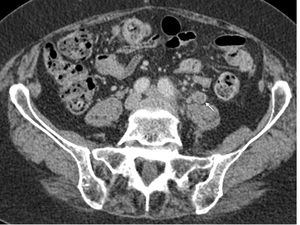
Fig. 16: Post-operative patient demonstrating a left ovarian vein thrombosis. Portal venous phase contrast enhanced CT axial image of the pelvis demonstrating a distended left ovarian vein (white arrow), with mild peripheral enhancement and fat stranding.
A rare complication of PID is ovarian vein thrombophlebitis,
which is right sided in 80-90% of cases [6].
Other causes include post-partum,
hyper-coagulable states and pelvic malignancy [7].
Patients report right-sided abdominal pain,
fever and nausea with limited examination findings depending on patient body habitus.
Occasionally septic pulmonary emboli can occur.
The condition is often diagnosed on CT,
where the ovarian vein is distended with low attenuation material and a thickened wall.
On MRI,
the thrombus within the ovarian vein will appear intermediate to hyperintense on T1 and T2 weighted imaging [7].
Treatment is still controversial but includes treating the underlying cause,
and anticoagulants may be required to prevent pulmonary emboli.
Scarring of the cervical os and intrauterine infection can lead to accumulation of pus within the endometrial cavity (Fig. 17).
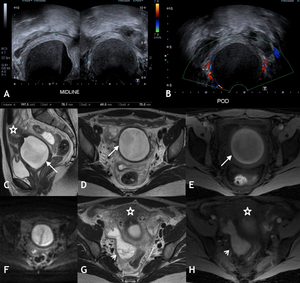
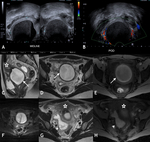
Fig. 17: Pyometra. 30 yr old patient presented with cyclical lower abdominal pain which became persistent and associated with fever. She had undergone a cervical procedure 1 year previously. Hypoechoic area with low internal echoes and fluid-fluid level seen within the pelvis, which appeared inferior and posterior to the uterus on US (A). Peripheral but no internal vascularity demonstrated on Doppler imaging (B). Initial diagnosis of endometrioma was made. Subsequent MRI demonstrated intermediate to high T2 weighted signal intensity (C sagittal and D axial) and high T1 weighted fat saturated image (E) within the cervical canal in keeping with blood products (white arrow). Restricted diffusion seen with high DWI (F) and low signal on ADC map (not shown) was identified in the cervical canal, which is difficult to interpret in presence of blood products. However given clinical findings of infection, the diagnosis of infected haematometra/pyometra was made, which was confirmed at surgery. No sexually transmitted infections or TB was detected. An associated haematosalpix was seen with high T2 signal with layered low T2 signal components (G) and high signal intensity on T1 weighted fat saturated imaging (H) (white arrow head). Uterus = *
Pyometra occurs in 0.01-0.5% of gynaecological patients but can be a high as 13.6% in post-menopausal groups [6].
50% of patients present with lower abdominal pain,
fever and purulent discharge but the remainder can be asymptomatic.
On imaging,
the endometrial cavity will be distended with complex fluid which can have a variable T1 and T2 signal intensity depending on proteinaceous contents.
Gas bubbles may be seen.
Enhancement of the endometrial cavity with para-uterine fat stranding,
free fluid and lymph node enlargement can occur.
In some cases,
scarring and intrauterine adhesions can form.
In 4% of cases,
this is secondary to infections by Mycobacterium tuberculosis [15],
which can result in pyometra (Fig. 18).
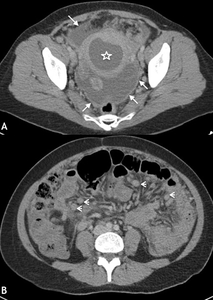
Fig. 18: TB pyometra in 40 year old patient. The patient presented with abdominal pain, 4 weeks of pyrexia but no gastrointestinal or genitourinary symptoms. On selected portal venous phase contrast enhanced CT axial images of the pelvis (A) and abdomen (B) reveal a distended fluid-filled endometrial cavity (*). Ascites with peritoneal enhancement (white arrow) seen throughout the abdominal cavity and there were multiple enlarged lymph nodes (white arrow heads). TB was suspected, which was confirmed on microbiological samples taken at hysteroscopy.
Chronic PID can cause intra-uterine adhesions.
Adhesions can cause infertility or sub-fertility,
amenorrhoea or chronic pain.
Hysteroscopy and hysterosalpingogram (HSG) are commonly used to identify adhesions (Fig. 19),
which are seen as filling defects within the uterine cavity [15].
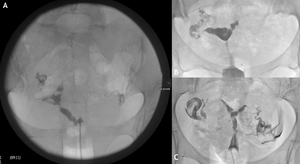
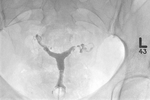
Fig. 19: Hysterosalpingogram (HSG). (A) HSG demonstrates filling defects with the endometrial cavity in keeping with Asherman’s syndrome. (B) HSG demonstrates left cornu irregular filling defect and occlusion of the left fallopian tube. (C) HSG demonstrates bilateral cornu and endometrial cavity defects, but patent fallopian tubes again in keeping endometrial adhesions.
Infertility is a common complication of chronic or recurrent PID. On HSG,
hydrosalpinx is demonstrated as a dilated tube,
with loculated or abrupt cut off contrast media transit at the level of the ampulla and no peritoneal spill [7] (Fig. 20).
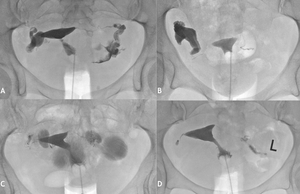
Fig. 20: Hysterosalpingogram (HSG). (A and B) demonstrate right and (C) bilateral distal tube dilation in keeping with a hydrosalpinx. (D) demonstrates a right tubal occlusion and normal left tube.
Salpingitis isthmica nodosa appears as mucosal irregularity and small diverticula on HSG (Fig. 21) The exact aetiology is unknown [14,
16].
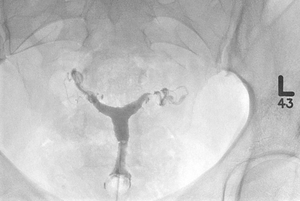
Fig. 21: Hysterosalpingogram (HSG) demonstrating multiple small diverticular and stenosis throughout the fallopian tubes in keeping with salpingitis isthmica nodosa.
As well as infertility,
tubal damage also increases the risk of ectopic pregnancy.