1.
Anatomical features of pediatric chest X-ray
Systematic approach to interpreting pediatric chest radiographs is required.
Detailed understanding of normal contours of the cardiomediastinum is essential if abnormalities are to be detected,
as well as knowledge of the range of normal for pulmonary vasculature marking.
Decided majority of CXRs are taken with the newborn lying down.
In case of children in severe conditions,
examination is performed by the patient's bed; modern incubators are adapted for such endeavor.
Images are taken in anteroposterior projection,
without the use of an anti-scattering grid,
often with hard radiation [6].
Radiographic imaging of pediatric chest presents several unique challenges,
stemming from congenital variants and pathologic processes specific to this population.
Errors in interpretation may lead to inappropriate further imaging and cost,
as well as psychologic effects on the patients and their families.
Following issues should be addressed in a systematical manner [7,8]:
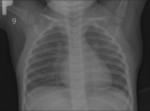
Heart size:
Heart size is often difficult to evaluate because of the supine AP projection and thymic shadow.
As a rule of thumb,
transverse diameter of the heart is half that of the thorax.
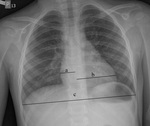
It is described using the cardiothoracic ratio,
calculated by dividing the greatest transverse diameter of the heart by the greatest inner diameter of the thorax (Fig.
2).
Cardiothoracic ratio of over 0,5 is a sign of cardiomegaly except in neonates,
where it should be <60%.
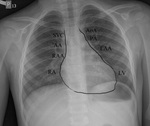
The right border of the heart silhouette is formed by superior vena cava,
ascending aorta,
right atrial appendage and right atrium.
The left consists aortic knob,
pulmonary trunk,
left atrial appendage and the left ventricle in AP projection (Fig.
3).
In a neonate,
the left border of the heart may still be formed by the right ventricle.
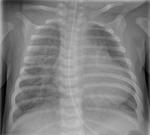
In lateral projection,
cardiac silhouette is oval in shape and the right ventricle is located directly behind the sternum.
The posterior border is formed by the left atrium and the left ventricle [8,9].
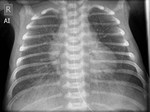
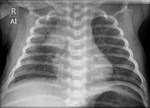
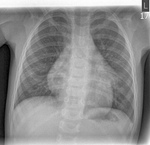
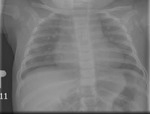
Figure 4 presents normal CXR findings in a neonate in AP view.
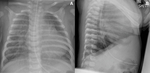
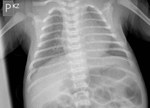
Figure 5 shows both AP and lateral views.
Other anatomical features:
Aeration of the normal neonatal lung is complete within two or three respiratory cycles after birth and the lung fields should appear symmetrically aerated on the initial X-ray [10].
Gradual change in the normal appearance of the CXR from infancy to adulthood must be appreciated.
At first chest configuration is more triangular in shape and deeper in the AP diameter.
Heart size and mediastinal width may be exaggerated by both supine position and AP projection.
Air bronchograms projecting through cardiac shadow in neonate and young infant may not be significant but should be considered pathological when seen more peripherally.
Anterior aspects of the diaphragms are higher,
with diaphragms at the level of the 8th ribs posteriorly and 5th to 7th ribs anteriorly in a well inspired examination,
costo-phrenic angles are relatively shallower,
and the lower zones may be obscured.
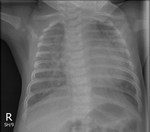
In neonates cardiac silhouette is frequently concealed by the presence of a still large thymus located in the anterior and superior mediastinum.
Thymus is recognized by its characteristic ‘sail’ shape or wavy margins resulting from the interdigitation of the soft thymic tissue in the horizontal tissue or intercostal spaces (Fig.
6).
It gradually becomes less evident between the ages of 2–8 [4,5,7,8].
2.
Review of selected CHD conditions in chest X-ray
Table 1 presents a brief summary of alterations within cardiac silhouette in common shunt lesions like Atrial Septal Defect (ASD),
Ventricular Septal Defect (VSD) and Patent Ductus Arteriosus (PDA) - figures 7-10.
Aortic Coarctation (CoA),
Figure of Three
Coarctations account for 5%–10% of all congenital heart defects and occur more frequently in males.
Increased frequency among patients with Turner syndrome is also seen - up to 20%–36% [1].
Clinical manifestations vary from congestive heart failure in infancy to hypertension with varrying blood pressures between upper and lower extremities in adulthood.
CoA results from a deformity of aortic media and intima, causing a prominent posterior infolding of the aortic lumen.
It characteristically occurs at or near the junction of aortic arch and descending thoracic aorta [11].
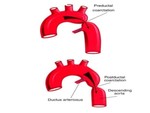
There are two main types of aortic coarctation - figure 11.
1.
Localized aortic coarctation (postductal/adult-type coarctation) is the most common type characterized by a focal narrowing of aorta just beyond the origin of the left subclavian artery or the ligamentum arteriosum.
Dilated and tortuous intercostal vessels may form deep grooves on undersurfaces of ribs (rib notching; usually ribs 3/4-8) appreciated in chronic disease thus unusual in patients under 5 years of age.
Pulmonary flow is normal unless left ventricular decompensation has occurred.
Contour of the aorta is deformed by both pre- and poststenotic dilatation,
the left subclavian artery is dilated.
2.
Tubular hypoplasia (preductal/infantile-type coarctation) is the second most common cause of heart failure in newborns - figure 12.
Long aortic segment beyond the origin of the innominate artery is narrowed.
This abnormality is combined with a superimposed focal constriction before the level of ductus arteriosus and usually associated with an intracardiac defect [12-15].
Classic radiologic sign associated with CoA and seen in 50%–66% of young adults is the figure-of-three sign - figure 13.
Aortic segment affected by coarctation has a shape that resembles the number „3” on frontal CXRs.
Number 3 is formed by dilatation of left subclavian artery and aorta proximal to the site of coarctation,
indentation at the site,
and dilatation of aorta distal to it.
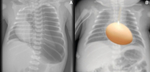
Transposition of the Great Arteries (TGA), Egg-on-a-String Sign
TGA is the most common cause of cyanotic CHD in newborns,
with incidence of 20-30 per 100,000 live births.
It is isolated in 90%,
being rarely associated with a syndrome or an extra-cardiac malformation.
The hallmark of TGA is ventriculoarterial discordance,
in which the aorta arises from the morphologic right ventricle and the pulmonary artery arises from the morphologic left ventricle.
Two separate circuits are formed-one that circulates oxygen-poor blood from the body back to the body,
and another that recirculates oxygen-rich blood from the lungs back to the lungs.
A communication (e.g.
patent foramen ovale (PFO),
ASD,
VSD,
or a combination) is required between the systemic and the pulmonary circulation to sustain life,
in addition to systemic collateral arteries.
About two-thirds of TGA patients have simple transposition and may only have a small PDA and/or PFO.
In presence of large communication,
prominent vessels and cardiomegaly are seen unless the shunt is balanced or impeded by an obstruction of the pulmonary artery [16].
Specific radiologic features are determined by the extent to which the great arteries are superposed in the plane of imaging,
the size of the communication between pulmonary and systemic circulation,
and the presence and severity of obstruction to pulmonary flow.
Pulmonary artery is situated to the right of its normal location and is obscured by aorta on AP view.
This malposition,
in association with stress-induced thymic atrophy and hyperinflated lungs,
results in apparent narrowing of superior mediastinum.
Cardiovascular silhouette varies from normal in the first few days after birth to enlarged and globular,
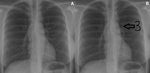
with the classic appearance described as an „egg on a string” - figures 14-15.
Right atrial border is abnormally convex,
and left atrium is enlarged because of increased pulmonary blood flow [12,17].
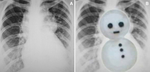
Total anomalous pulmonary venous return (TAPVR),
Snowman sign,
Figure-of-eight sign
TAPVR represents around 0.7 to 1.5% of CHD cases.
Pulmonary veins fail to drain into the left atrium and instead form an aberrant connection with some other cardiovascular structure.
Both systemic and pulmonary venous blood enters the right atrium.
Systemic desaturation occurs as the result of mixing of two circulations.
Survival is maintained by a right-to-left shunt through a communication at the level of atrial septum.
Four types of TAPVR can be defined in decreasing order of frequency depending on the site of anomalous venous union [3].
In supracardiac type (Type 1,
most common 52%) anomalous pulmonary veins terminate at the supracardiac level.
On a AP CXRs,
this anomaly resembles a snowman - figure 16,
also known as figure of 8 heart or cottage loaf heart sign.
Dilated vertical vein on the left,
the innominate vein on the top,
and the superior vena cava on the right form the head of the snowman while the body is formed by the enlarged right atrium.
The sign is nowadays rarely seen in the era of rapid diagnosis and early repair [12,13,17-22].
Another types of TAPVR are: infradiaphragmatic (20%-30%),
cardiac (20%) with heart size usually normal,
but with severe interstitial pulmonary edema,
thymic atrophy,
and depression of the diaphragm,
and mixed type (10%).
In.
30% of cases of TAPVR associated cardiac lesions and heterotaxy syndromes,
particularly asplenia,
are encountered.
Type III is associated with thoracic lymphangiectasia and pulmonary congestion [23].
Tetralogy of Fallot (ToF),
Boot-shaped Heart
ToF accounts for 5 to 10% of all CDH with a prevalence of 1 in 2000 births.
The four established components of ToF include ventricular septal defect,
infundibular pulmonary stenosis,
overriding aorta and right ventricular hypertrophy.
On CXRs heart has a shape of a wooden shoe or boot (in French,
coeur en sabot) - figures 17-18 - due to upturned of cardiac apex because of right ventricular hypertrophy and concavity of main pulmonary artery [12,13,24].
Shadow of the pulmonary arterial trunk is almost invariably absent,
blood flow to the lungs is usually reduced (pulmonary oligaemia).
Small,
hypoplastic or atretic pulmonary arteries will cause the mediastinum to appear narrow.
Right-sided aortic arch is seen in 25%.
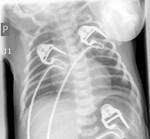
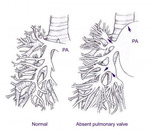
ToF With Absent Pulmonary Valve
ToF with absent pulmonary valve is seen in up to 3% of patients diagnosed with ToF and presents features of ToF with either rudimentary ridges or a complete absence of pulmonic valve tissue and usually with hypoplastic pulmonary valve annulus.
Absence of mature functioning pulmonary valve leads to severe pulmonary insufficiency,
often associated with aneurysmal dilatation of the proximal branch pulmonary arteries - figure 19.
CXR usually reveals aneurysmally dilated central pulmonary arteries with otherwise normal peripheral pulmonary vascularity.
Cardiomegaly results from dilation of the right ventricle,
particularly its infundibulum - figure 20.
Other abnormalities may be evident (eg.
atelectasis,
pneumonic infiltrates,
lobar emphysema,
hyperinflation).
Air trapping may cause a herniation of a lobe.
Right aortic arch may be also found [25-29].
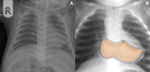
Ebstein anomaly,
Box-shaped heart
Ebstein anomaly accounts for approx.
0.5%-0,7% of CHD.
Its true prevalence is unknown because mild forms frequently are undiagnosed.
It is the only cyanotic congenital malformation of the heart in which both aorta and pulmonary trunk are smaller than normal.
Pulmonary flow may vary from normal to borderline to diminished.
If normal,
the patient is usually acyanotic.
Ebstein anomaly is characterized by apical displacement of the septal and posterior tricuspid valve leaflets,
leading to atrialization of the right ventricle with a variable degree of malformation and displacement of the anterior leaflet.
The most consistent imaging feature is right atrial enlargement; the right atrium may be huge and fill the entire right hemithorax.
Left atrium is normal in size,
but the left cardiac contour has a shelved appearance because of dilated right ventricular outflow tract.
Aorta is small,
and pulmonary trunk outline is absent.
This combination of features presents with a cardiac silhouette described as box shaped [22,30-34] - figure 21.