Computed Tomography (CT) is an excellent means for visualizing the imaging features of GP [3].
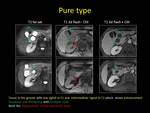
In the pure form a poorly enhancing plate-like hypodense lesion (which may range from ill-defined fat stranding to frank soft tissue) is depicted between the pancreatic head and the duodenum,
near the minor papilla [3,5].
This lesion shows delayed enhancement which is due to delayed blood circulation,
caused by fibrous tissue proliferation and secondary arteries constriction.
Duodenal wall thickening and stenosis as well as cystic-like lesions of variable sizes in the duodenal wall or in the groove area are usually observed.
Thickening of the medial duodenal wall and small cysts either within the duodenal wall or the groove are better demonstrated on coronal images [4].
The latter are also useful in the assessment of the widening of the distance between the pancreas and the duodenum,
caused by the thickened duodenal wall and the presence of fibrous component in the groove [3,5,7].
Fig. 4: Pure form of Groove pancreatitis in pancreatic phase CT
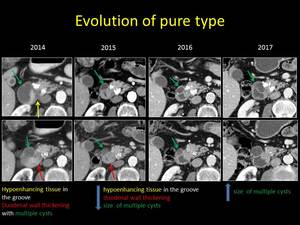
Fig. 5: Evolution of the pure form of Groove pancreatitis over time
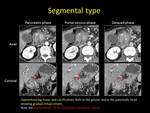
In the segmental form, CT reveals a focal hypodense lesion in the pancreatic head in close proximity to the duodenal wall.
In this case the mass-like enlargement of the pancreatic head obscures the involvement of the groove itself,
making the diagnosis even more difficult [5]. The main pancreatic duct may appear mildly dilated in the body and tail,
while in the pure form it is usually normally sized.
The CBD is narrowed at its distal part in both forms,
leading to dilatation of the intra- and extra-hepatic biliary ductal system [3,5].
A dilated banana-shaped gallbladder has also been described as an ancillary finding [5].
In most cases,
the constriction of CBD is relatively smooth,
tapered and regular [5,7].
The peripancreatic vessels do not show signs of thrombosis or infiltration.
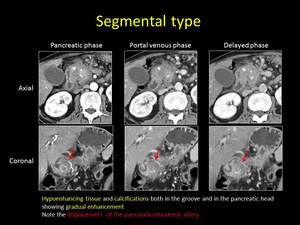
Fig. 6: CT findings of segmental form of Groove pancreatitis (axial and coronal reformations)
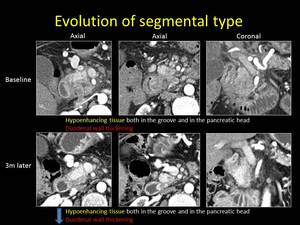
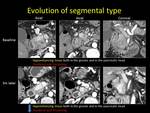
Fig. 7: Evolution of the segmental type of Groove pancreatitis over time (axial and coronal reformations)
The most characteristic feature in MRI is a “sheet-like” crescentic mass between the pancreas and the duodenum associated with duodenal wall thickening [3].
The soft tissue is typically hypointense on T1-weighted images and has variable signal intensity on T2-weighted images depending on the acuity of the disease [5].
In the acute phase,
the tissue tends to be more hyperintense because of oedema and fluid,
while in a more advanced stage it becomes progressively more hypointense,
due to accumulation of fibrous component.
In the segmental form,
a focal mass-like lesion with progressive loss of T1 signal intensity in the pancreatic head is visualized,
as a result of parenchymal atrophy and fibrosis,
along with dilatation of the main pancreatic duct [3,5].
The medial duodenal wall appears to be thickened in both forms (pure and segmental) with high signal intensity on T2-weighted images.
Multiple T2 hyperintense cysts can be depicted in the duodenal wall and the pancreaticoduodenal groove.
Gadolinium-enhanced T1-weighted images show progressive enhancement of the mass in the groove and/or the pancreatic head and the thickened duodenal wall.
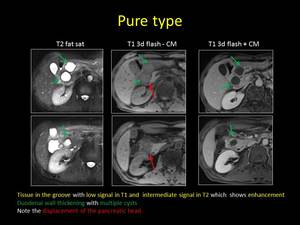
Fig. 8: MRI findings of pure form of Groove pancreatitis
MRCP delineates abnormalities of the distal CBD and the downstream pancreatic duct,
both of which tend to be narrowed near the ampulla.
Common findings in the pure and segmental form include a long,
smooth stenosis of the distal or intrapancreatic CBD and a medial shift of the duct contracted by fibrosis [3].
The main pancreatic duct is usually normal in the pure form,
but in the segmental form a stricture is frequently observed within the pancreatic head with proximal pancreatic duct dilatation.
The distance between the ampulla and the duodenal lumen is widened.
US findings of groove pancreatitis are not well described in the literature.
In early stages,
when inflammation prevails,
a hypoechoic bandlike thickening of the pancreaticoduodenal groove or a heterogeneous hypoechoic pancreatic head can be visualized [5].
Thickening of the adjacent duodenum can also be apparent.
In chronic stages,
fibrosis dominates over inflammation and an hyperechoic band in the groove contiguous with an hyperechoic thickening of the duodenum and an increasingly hyperechoic pancreatic head are encountered.
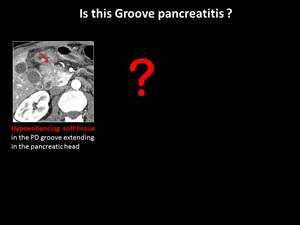
Fig. 9: Differential diagnosis of Groove pancreatitis
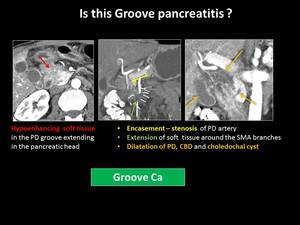
Fig. 10: Groove adenocarcinoma - key findings for correct diagnosis
The differential diagnosis between groove pancreatitis and pancreatic adenocarcinoma can be extremely difficult and many cases with groove pancreatitis inevitably proceed to surgery [3,5].
Both lesions appear hypovascular with delayed enhancement on MDCT and MRI studies.
However,
Ishigami et al.
suggested that patchy focal enhancement in the portal venous phase was more indicative of groove pancreatitis,
reflecting pancreatic tissues within the inflammatory mass [8].
On the contrary,
peripheral enhancement was only seen in groove pancreatic carcinomas,
because tumor cells predominantly proliferate in the peripheral portion around a central area of fibrosis and necrosis.
The latter feature may not be seen if the cancer cells proliferate in a scirrhous fashion and the tumor tends to be more homogeneously hypodense on MDCT.
In groove pancreatitis,
displacement of the major vessels can occur,
but no signs of obstruction or encasement are encountered as in adenocarcinomas [3].
Graziani et al.
reported that the gastroduodenal artery is displaced leftward in GP,
while in adenocarcinoma it is located between the lesion and the duodenum [9].
In GP,
CBD narrowing tends to be long and tapered,
without evidence of “shouldering”,
irregularity or abrupt margins,
while upstream dilatation is not marked [3,5]. In the segmental form of GP,
the pancreatic duct stricture is longer than in cases of pancreatic adenocarcinoma.
Widening of the space between distal pancreatic and common bile ducts is not commonly seen in cases of pancreatic cancer [3]. However,
in malignancy arising immediately adjacent to the groove,
pancreatic ductal cutoff,
CBD obstruction and upstream atrophy may be absent [5].
Moreover,
both GP and pancreatic carcinoma arising in the groove area can be associated with duodenal wall thickening and lumen stenosis,
but the presence of cystic lesions is in favor of GP [3,10].

Fig. 11: Differential diagnosis of Groove pancreatitis
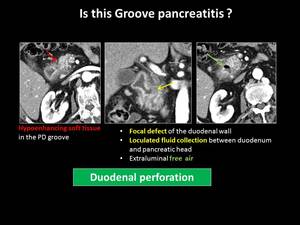
Fig. 12: Contained duodenal perforation - key findings for correct diagnosis
The differential diagnosis of groove pancreatitis also includes duodenal carcinoma,
common bile duct cholangiocarcinoma,
duodenal gastrointestinal stromal tumors and neuroendocrine tumors,
oedematous pancreatitis with involvement of the groove,
chronic pancreatitis in acute transformation with pseudocysts in the duodenal wall,
cystic dystrophy of the duodenum and contained rupture of the duodenum [3,10].