Institutional review board approval and waiver for informed consent was obtained for this retrospective study.
Patients
A computer search in our hospital database was performed,
identifying patients with unresectable PDA treated with FOLFIRINOX regimen between November 2010 and February 2017.
Inclusion criteria were as follows: (1) histological evidence of PDA; (2) unresectability of the pancreatic primary tumour established by a gastro-intestinal multidisciplinary team; (3) FOLFIRINOX therapy duration of at least 1 month or 2 cycles; (4) baseline CECT study including a portal-venous scan of thorax-abdomen-pelvis performed within 8 weeks from therapy start; (5) at least one follow-up CECT study including a portal-venous scan of thorax-abdomen-pelvis performed 8-12 weeks after therapy start.
The exclusion criteria were as follows: (1) therapy duration inferior to 1 month or 2 cycles; (2) no baseline CECT study available for review; (3) unenhanced baseline or follow-up CT studies (4) CECT study inadequate for CTTA (e.g.
suboptimal enhancement or beam-hardening artifacts projecting over the PDA); (5) no measurable disease at baseline as per RECIST 1.1; (6) no distinguishable PDA at baseline.
Treatment was administered every 2 weeks as per FOLFIRINOX protocol.
All CECT follow-up studies were obtained within 8-12 from each other,
until therapy stop in the event of disease progression (either clinical or radiologically assessed) or unacceptable toxicity.
Based on baseline PDA staging,
patients were divided into LAPC and MPC.
Image acquisition
Multi-detector-row helical CT scanners were used (Siemens,
Berlin,
Germany),
and images of the thorax,
abdomen and pelvis were acquired before and after intravenous contrast administration of iohexol (300 mg iodine per milliliter,
Omnipaque 300; GE Healthcare,
Cork,
Ireland) using a power injector at a flow-rate of 2-3 mL/sec,
depending on the cannula size at a dose of 2ml per kg of patient body weight.
Pancreatic and portal venous phase imaging were performed in the craniocaudal direction,
with a 40- and 80-second delay respectively (120 kVp; 170–350 mAs; collimation,
0.2 mm).
Routine dataset reconstructions at 5-mm section thickness were obtained from the scanner raw data for lesion measurements purposes.
Image analysis
Portal-venous phase images were manually imported from PACS into a commercially available software (Mint LesionTM v3.2.3,
Mint Medical,
Heidelberg,
Germany) for response assessment and CTTA purposes.
Target lesions were chosen following RECIST 1.1 criteria (no more than five target lesions,
up to two target lesions per organ) by two radiologists in consensus.
Each lesion was measured in its longest axis (short axis for lymph nodes selected as target lesions) at each timepoint; the sum of diameters (SOD) of all target lesions (millimeters – mm) was computed automatically by the software at each timepoint.
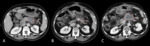
Additionally,
a polygonal region of interest (ROI) was drawn around the primary pancreatic tumour excluding vessels and other adjacent structures (e.g.
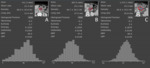
duodenum or biliary stents) on the single slice where it was best represented (Figure 1); first-order lesional CTTA metrics (kurtosis,
entropy and uniformity) derived from the voxels included in the ROI were automatically calculated by the software for each timepoint (Figure 2).
Response assessment
For every follow-up study,
therapy response from baseline was evaluated as per RECIST 1.1 criteria,
based on the SOD and the relative percentage change from baseline and nadir calculated by the software.
Each study was graded as complete response (CR),
partial response (PR),
stable disease (SD) or progressive disease (PD) following RECIST 1.1 criteria.
For each patient,
the follow-up recording the smallest SOD was considered as best response (BR).
Patients were divided into responders (R) and non-responders (NR) based on their BR (R=CR+PR; NR=SD+PD).
Texture analysis
First-order CTTA metrics values were recorded at baseline (BL),
first assessment (FA),
BR and PD; percentage changes from BL to BR and from BR to PD were calculated and recorded.
Statistical Analysis
All data were analysed using a statistical software (Stata 13; StataCorp,
College Station,
Tex) for the LAPC and MPC groups separately.
Mann-Withney U test was used to assess differences between responders and non-responders by RECIST 1.1 in CTTA metrics values recorded at BL and FA separately; the same was done for the CTTA metrics percentage change recorded from BL to BR and from BR to PD.
A p value equal or inferior to .05 was considered as statistically significant.
To assess the diagnostic performance of CTTA metrics and their percentage changes a receiver operating characteristic (ROC) analysis was performed.
The cut-off values were calculated from the Youden Index.
CTTA quantitative parameters are reported as median value and interquartile range (IQR).