Management of PPH
Patients with PPH require aggressive measures to restore and maintain the circulating blood volume to vital structures.
All medical units involved in the care of pregnant women must have a protocol for the management of severe PPH.
Implement the protocol in a manner similar to a cardiac arrest protocol,
with the same attention to detail and documentation.
The diagnosis of PPH is established by observing the amount of bleeding and the patient’s clinical status.
The amount of blood lost and the patient’s level of consciousness and vital signs are continually assessed.
The magnitude and underlying cause of the bleeding to some degree dictate which specialized personnel are called,
but a minimum of one obstetrician and one anesthetist is necessary.
Skilled nurses can be indispensable.
Notifying blood transfusion services is essential because the timely availability of blood products is likely to be critical.
As in a cardiac arrest,
designate an experienced person to document critical information and times.
Ensure to activate CATH Lab/operating room.
The speed with which PPH occurs,
becomes life-threatening,
and can be successfully managed with relatively simple interventions sometimes makes it difficult to decide when to institute the full protocol.
Fluid resuscitation
Fluid resuscitation of women experiencing obstetric hemorrhage is sometimes overly conservative.
Possible reasons for this include
-
Blood loss being generally underestimated both in volume and rapidity
-
Patient initially compensating well for losses because of their good health and the hypervolemia of pregnancy
-
Concerns that over resuscitation leads to pulmonary edema
-
Failure to appreciate the dynamics of fluid shifts in the body.
Immediately commence resuscitation.
Raising the legs improves venous return and is consistent with the positioning used to diagnose and treat the underlying causes of bleeding.
Administer oxygen and obtain intravenous access.
All intravenous lines started on the labor ward for other reasons must be placed with cannulas of sufficient gauge if PPH develops.
Twice as much fluid can be infused through a 14-gauge intravenous line compared with an 18-gauge intravenous line over the same time period.
Perform the initial resuscitation with large volumes of Crystalloid solution,
either normal saline (NS) or Ringer’s Lactated solution (RL),
through peripheral intravenous sites.
Central venous access is not required for the vast majority of patients with PPH,
but do not delay establishing such access if necessary.
Draw blood for baseline measurements at this time.
NS is a reasonable solution in the labor ward setting because of its low cost and compatibility with most drugs and blood transfusions.
The risk of hyperchloremic acidosis is very low in the setting of PPH.
If large amounts of crystalloid are being infused,
a change to RLS can be considered.
Dextrose-containing solutions,
such as 5% dextrose in water or diluted NS in 5% dextrose in water,
have no role in the management of PPH.
Remember that the loss of 1 L of blood requires replacement with 4-5 L of crystalloid because most of the infused fluid is not retained in the intravascular space but instead shifts to the interstitial space.
This shift,
along with oxytocin use,
may result in peripheral edema in the days following PPH.
Healthy kidneys easily excrete this excess fluid.
Use wide-open initial infusion rates,
with the goal of infusing the required replacement volume over minutes rather than hours.
PPH of up to 1500 ml in a healthy pregnant woman can usually be managed by crystalloid infusion alone if the cause of bleeding is arrested.
Blood loss in excess of this usually requires the addition of a PRBC transfusion.
Because a large portion of crystalloid fluid volume is lost to the interstitial space,
the use of colloids in resuscitation has been examined.
These solutions are largely retained within the intravascular space and include albumin,
dextran,
hydroxyethyl starch,
and modified fluid gelatin.
Large volumes of colloid solutions (>1000-1500 mL/d) can have an adverse effect on hemostasis.
No colloid solution has been demonstrated to be superior to NS,
and,
because of the expense and the risk of adverse effects with colloids,
crystalloid is recommended.
Blood transfusion
Order blood transfusions if blood loss is ongoing and thought to be in excess of 1500 ml or if the patient having poor clinical status.
Whole blood is no longer available in most settings,
and for many reasons,
PRBCs are initially used with other blood components and given only if indicated.
In PPH,
uncrossmatched ABO- and Rh-compatible blood is usually available because a blood group and antibody screen has already been performed.
Have full crossmatched blood available for transfusion within 30 minutes.
Clinicians must be aware of the capabilities of their blood bank regarding timing,
type and amount of blood products available in emergencies.
Good communication with the blood transfusion service is essential and the nature of the emergency and the potential amount of blood products required must be stressed.
The goal is to rapidly transfuse 2-4 U of PRBCs to replace lost oxygen-carrying capacity and to restore circulating volume.
Administer the blood transfusion through a set with an integrated filter and use a blood warmer if the infusion rate (>100 mL/min) or the total volume infused is high.
A rapid infusion set with an integrated warmer or a pressure cuff may be used to increase the infusion rate.
PRBCs are very viscous,
reducing the infusion rate.
This problem may be overcome by adding 100 ml of NS to each unit.
Do not use RL for this purpose because the calcium contained in the solution may cause clotting.
The risks of transfusion are well known and are covered elsewhere,
but they include infection,
transfusion reaction,
and development of atypical antibodies.
Several other complications may be noted in large-volume transfusions.
The risk of hypothermia is minimized by the use of blood warmers.
Monitor electrolyte and acid-base status if the situation is ongoing.
Hypocalcemia due to citrate intoxication is also seldom observed.
Patients may refuse a transfusion of blood products based on religious or other grounds.
A patient's refusal of blood products must be respected and must not be equated with a desire for no intervention or be seen as an excuse for suboptimal care.
Several options,
including the use of autotransfusion can be considered for the management of these patients.
Ensure that a care plan is in place.
Coagulopathy
Women experiencing PPH do not usually have a pre-existing disorder of hemostasis,
however initial blood work includes a coagulation screen and platelet count.
Regularly monitor hemostatic test results in all patient who require a massive transfusion.
If findings are abnormal in conjunction with ongoing bleeding additional blood products are required.
Infuse fresh frozen plasma (FFP),
beginning with 4U and following with additional units to normalize the coagulation test findings. International standarised ratio for infusion recommend the addition of 1Unit of FFP for every 5Unit of PRBCs for patients who require continued transfusion.
Keep the platelet count more than 1Lakh by using platelet transfusion.
After giving each unit of PRBC monitor the platelet count,
the increases in platelet count depends upon the patient condition.
If bleeding is continuing and the platelet count is less than 50 Thousand - 1 Lakh,
administrate to give platelet.
If surgical intervention is necessary,
maintain the platelet count more than 1 Lakh.
Cryoprecipitate may be useful along with FFP because of the markedly decreased fibrinogen levels.
Cryoprecipitate provides a more concentrated form of fibrinogen and other clotting factors (VIII,
XIII,
von Willebrand factor) and is faster to prepare in the blood bank.
It is commonly given in 2-5Units and may also be helpful immediately before any surgical intervention in patients with abnormal coagulation test results.
Response to resuscitation
Pay close attention to the patient’s level of consciousness,
Pulse rate,
Blood pressure,
Saturation and Urine output during the course of the management of massive hemorrhage.
A urine output of 30 ml/h or more likely indicates adequate renal perfusion.
Closely monitor the CBC count,
coagulation,
and blood values in addition to acid-base status.
A Cardiac monitor is useful for evaluating tissue perfusion and oxygen saturation.
Frequent auscultation of the lung fields helps detect pulmonary edema or the development of adult respiratory distress syndrome.
For patients in critical condition with ongoing bleeding,
the placement of a central venous line may be helpful for resuscitation.
Arterial line placement also may aid in monitoring blood pressure and allowing easy access for blood work.
Few patients experiencing PPH require such invasive monitoring.
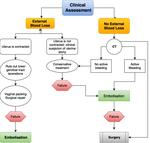
Management of massive PPH
The following is a plan for managing massive obstetric hemorrhage,
Organization
- Call experienced staff (including obstetrician and anesthetist).
- Alert the blood bank and hematologist.
- Designate a nurse to record vital signs,
urine output,
and fluids and drugs administered.
- Activate CATH Lab on standby.
Resuscitation
- Administer oxygen by mask.
- Place 2 large-bore (14-gauge) intravenous lines.
- Take blood for crossmatch of 6U PRBCs,
and obtain a CBC count,
coagulation screen,
urea level,
creatinine value,
and electrolyte status.
- Begin immediate rapid fluid replacement with NS or RL solution.
- Transfuse with PRBCs as available and appropriate.
Defective blood coagulation
- Order coagulation screen (International Normalized Ratio,
activated partial thromboplastin time) if fibrinogen,
thrombin time,
blood film,
and D-dimer results are abnormal.
- Give FFP if coagulation test results are abnormal and sites are oozing.
- Give cryoprecipitate if abnormal coagulation test results are not corrected with FFP and bleeding continues.
- Give PRBC if the platelet count is less than 1 lakh and bleeding continues.
- Use cryoprecipitate and PRBC before intervention.
Evaluation of response
- Monitor pulse,
blood pressure,
blood gas status,
and acid-base status,
and consider monitoring central venous pressure.
- Measure urine output using an indwelling catheter.
- Order regular CBC counts and coagulation tests to guide blood component therapy.
Remedy the cause of bleeding
- If postpartum,
use oxytocin,
prostaglandin,
or ergonovine.
- Explore and empty the uterine cavity,
and consider uterine packing.
- Examine the cervix and vagina,
ligate any bleeding vessels,
and repair trauma.
- Ligate the uterine blood supply
- Consider arterial embolization.
Uterine Artery Embolisation Technique
UAE for the treatment of PPH was performed only after all usual obstetric maneuvers for the treatment of PPH were used.
The protocol and technique for UAE for PPH is relatively standardized in our division and each procedure was performed by a fellowship-trained Interventional radiologist and his team. The procedure is performed in CATH lab.
A digital subtraction angiography of the bilateral internal iliac arteries was performed by the interventional radiologist.
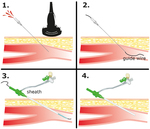
Under Ultrasound guidance the femoral punture site is idenitfied and puntured using a 18G needle and a 5F sheath introduced into the right-side femoral approach with 4F cobra-shaped catheter and a 0.035-inch-diameter hydrophilic polymer-coated angled guide-wire (TERUMO) and is advanced into the uterine artery using a digital road map for guidance.
The uterine artery usually arises as either the first branch off the bifurcation of the inferior gluteal artery or as part of a genitourinary trunk at the level of the bifurcation of the superior gluteal and inferior gluteal arteries.
Oblique positioning of the angiographic unit is essential for the best visualization of the vessel because it is important to have good visualization in uterine arteries origin to allow easy access.
Usually left anterior oblique (LAO) is used for both uterine arteries.
If one oblique does not show the origin of the vessel well,
then the opposite oblique should be used.
Bilateral uterine arteries were also catheterized using a 5F Roberts Uterine Catheter (RUC).
After superselective angiography confirmed catheter position in the distal uterine artery,
often in the horizontal portion of the artery,
embolization was performed.
As long as a pseudoaneurysm was not visualized with selective uterine angiography,
Embolisation of the uterine arteries was carried out preferentially with Absorbable Polyvinyl Alcohol Particle 300μm-500μm(Cook India Medical Devices) or Absorable Haemostatic Gelatin Sponge pieces (Spongostan,
Ethicon INC USA) or both,
administered in a slurry with little contrast medium.
Embolization was performed until stasis of flow in the uterine artery was achieved.
With the same femoral arterial access,
the uterine artery was then selected and embolized in a similar manner in all cases.
If a pseudoaneurysm was identified,
it was embolized with MicroNester Embolization Coil (Cook India Medical Devices) through a RUC.
Subsequently,
vascular occlusion was confirmed by repeat angiography,
and uterotonic agents were administered,
further treatments were performed when necessary.
Then the patient is shifted to an intensive care unit for close monitoring.
the patient will be immobilised becouse of the arterial puncture ,
there is high chance of groin hematoma if the patient is mobilised.
The groin bandage is removed after 12hrs.
The prior to the next day of procedure blood investigation will be done like CBC and Creatine level to rule out any further fall counts and an Ultrasound Abdomen portable is done for any arterial injury.
If the bleeding persist and the patient have hemodynamically instability will go for Hysterctomy.