A-Imaging techniques
Neuroblastoma can be staged with either Magnetic Resonance Imaging (MRI) or contrast-enhanced computed tomography (CECT).
Each technique has its advantages and drawbacks,
and the use of each of them depends upon the availability of each imaging technique in the radiology Department (Fig.3) (Fig.4).
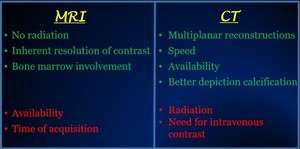
Fig. 3: Summarized table with the advantages and disadvantages of using Magnetic Resonance Imaging (MRI) and computed tomography (CT) in the staging of neuroblastomas.
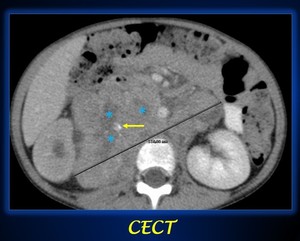
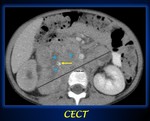
Fig. 4: Axial contrast-enhanced abdominal CT shows a large heterogeneus mass (measurement line) with necrotic areas (blue asterisks), midline crossing and presence of calcium (yellow arrow) in keeping with a neuroblastoma.
References: University Hospital Niño JesĂșs
Ultrasonography (US) is usually performed as a first line in the setting of an abdominal mass in a children because of its wide availability and noninvasiveness.
However,
it is operator dependent,
does not provide an only image of the whole abdominal compartment,
and the relationship of the mass with the vessels is usually difficult to assess.
Therefore,
US is not a suitable imaging method to stage this tumor.
On ultrasound images,
as in MRI and CECT,
neuroblastoma may have a very variable appearance.
(Fig.
5).
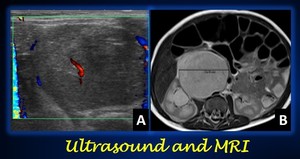
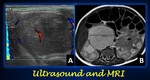
Fig. 5: Neuroblastoma in a 2-month old girl in ultrasound and MRI. (A) Axial ultrasound at the level of the right flank shows an heterogeneous predominantly hypoecoic round mass with scarse vascularity in its centre and periphery. (B) T2-weighted abdominal MRI provides better depiction of the mas (measurement line) and its relationships with the neighboring structures.
References: University Hospital Niño JesĂșs
Study protocol
On MRI,
appropriate MR sequences are spin-echo,
fast spin-echo,
inversion-recovery,
and spoiled gradient-echo T1-weighted sequences,
and fast spin-echo and short-tau inversion-recovery T2-weighted sequences.
The tissue contrast on T2-weighted images can be increased with use of fat saturation or water-excitation options.
The use of gadolinium-enhanced T1-weighted sequences is optional,
but if these sequences are used,
they should be performed with fat saturation to increase the contrast against fatty tissues.
The acquisition planes should always include the axial plane and at least one longitudinal plane (coronal or sagittal).
A three-plane study is strongly recommended for paraspinal neuroblastomas to assess foraminal and intraspinal extensions.
The orientation of the coronal views should be adjusted on the sagittal view to yield images that demonstrate the full foraminal and intraspinal tumor extent on the same coronal image.
If the staging wants to be performed with CT,
intravenous contrast is mandatory,
unlike MRI.
In both MRI and CECT,
imaging should include the primary tumor compartment and the adjacents superior and inferior anatomic compartments.
The main diagnostic challenge in the staging of neuroblastoma is to differentiate it from Wilms tumor when it is located in the abdomen.
Midline crossing,
vessel encasement and presence of calcium are typical features of neuroblastoma that may help in its diagnosis.
B-Staging
With the aim to homogenize the staging criteria of neuroblastoma,
investigators from the major cooperative groups formed the INRG (International Neuroblastoma Risk Group Task Force),
which developed the INRGSS (International Neuroblastoma Risk Group Staging System).
The INRGSS,
published in 2009,
is a staging system based on preoperative diagnostic images.
Since imaging can be retrospectively and centrally reviewed by experts in the field,
a system based on baseline imaging features should be more robust and reproducible than one based on surgical resection.
Therefore,
with the INRGSS,
the focus has shifted from surgico-pathologic staging to imaging.
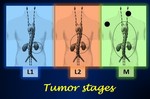
This new system divides the tumors in localized (stage L1 or L2 disease) on the basis of wether one ore more of the 20 IDRFs were present,
and metastatic (stage M if distant metastasis are present,
and stage MS in metastatic disease in children younger than 18 months,
with metastases confined to skin,
liver and/or < 10% of the bone marrow,
which resolve spontaneously).
Stages (Fig.6)
L1: Localized tumor not involving vital structures,
as defined by the list of IDRFs,
and confined to one body compartment.
*The tumor must be confined within one body compartment (neck,
chest,
abdomen or pelvis).
L2: Local-regional tumor with presence of one ore more IDRFs.
*The tumor may be ipsilaterally contiguous within body compartments.
M: Distant metastatic disease (except MS tumor)
*Non regional (distant) lymph node involvement is metastatic disease.
Primary tumors in an upper or lower anatomic compartment with linfadenopathies in the upper or lower adjacent anatomic compartment is considered loco-regional,
and therefore,
not metastatic.
MS: Metastatic disease in children younger than 18 months,
with metastases confined to skin,
liver and/or < 10% of the bone marrow.
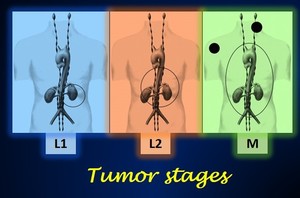
Fig. 6: Illustration summarizing the stages of neuroblastoma according to the INRGSS (International Neuroblastoma Risk Group Staging System). Stage MS is not inlcuded in the picture.
Modified images from Brisse J., et al Guidelines for Imaging and Staging of Neuroblastic Tumors: Consensus Report from the International Neuroblastoma Risk Group Project. Radiology 2011; 261 (1):243-257.
IDRFs
IDRFs are surgical risk factors,
detected on images,
that predict the resection will be very risky or difficult at the time of diagnosis.
Describing an imaging feature as an IDRFs depends on two main things: the type of relationship with the primary tumor,
and the vital structure involved
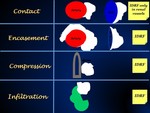
a)Types of relationships with vital structures
There are 4 different types of relationships a neuroblastoma may have with the neighboring vital structures (Fig.
7) :
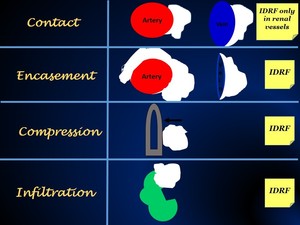
Fig. 7: Illustration showing the terms to describe relationships between primary tumor and vital structures.
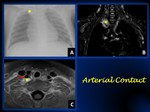
I.Contact. In an artery,
there is contact when the tumor surrounds less than 50% of its circumference without a visible layer between them.
In a vein,
there is contact when the tumor flattens its circumference but there is still a visible lumen.
Contact is an IDRF ONLY in the renal vessels.
(Fig.
8).
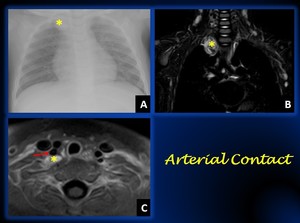
Fig. 8: Figure A shows a mass in the medium or posterior mediastinum- cervical region (yellow arrow). Coronal T1 weighted fat-sat contrast-enhanced MRI (B) shows the mass in the cervicothoracic junction Core-needle biopsy proved it to be a neuroblastoma.
Figure C shows an axial T1-weighted MRI at the level of the cervicothoracic junction. There is conctat but not encasement of the vertebray artery (yellow arrow), therefore, an IDRF is not present.
References: University Hospital Niño JesĂșs
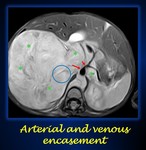
II.Encasement. In an artery,
there is encasement when the tumor surrounds more than 50% of its circumeference without a visible layer between them.
In a vein,
there is encasement when the tumor narrows its circumference and a lumen is not visible.
When there is encasament,
an IDRF is present (Fig.9).
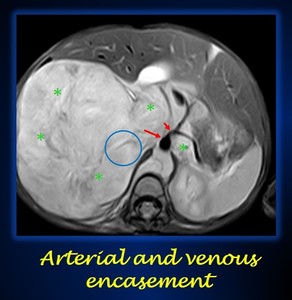
Fig. 9: Axial T2 weighted MRI in a 9 years-old female with a giant neuroblastoma in the right abdomen that crosses the midline (green asterisks). The Inferior vena cava is not depicted in its usual location, as it is collapsed by the lesion (round blue circle). The abdominal aorta (large red arrow), and celiac axis and its branches (small red arrow) are completely surrounded by the tumor. Thus, there is arterial and venous encasement, and IDRFs are present.
References: University Hospital Niño JesĂșs
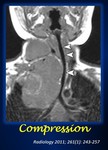
III.Compression. This term is only used when referring to the airways.
Compression occurs when the tumors causes the short axis of the airways to be reduced.
When there is compression,
an IDRF is present (Fig.10).
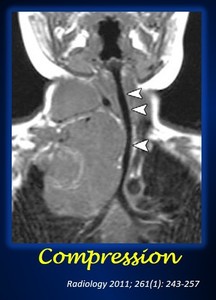
Fig. 10: Coronal T1-weighted MR image shows a reduced and displaced trachea (arrowheads). An IDRF is therefore present.
References: Brisse J., et al Guidelines for Imaging and Staging of Neuroblastic Tumors: Consensus Report from the International Neuroblastoma Risk Group Project. Radiology 2011; 261 (1):243-257
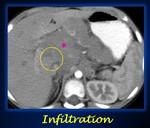
IV.Infiltration. This term refers to vital structures other than vessels or airways.
The presence of ill-defined margins between the tumor and the neighboring structure is considered infiltration.
When a tumor infiltrates a vital structure,
an IDRF is present (Fig.11).
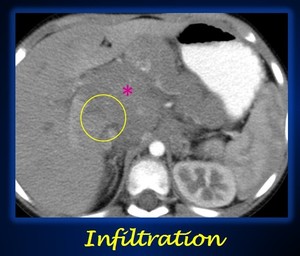
Fig. 11: Axial abdominal contrast-enhanced CT shows ill-defined borders (yellow circle) between the necrotic abdominal mass (pink asterisk) and the right hepatic lobe, a finding suggestive of infiltration.
References: University Hospital Niño JesĂșs
b) Vital structures involved
In order to provide a better comprehension,
IDRFs are described upon their anatomic compartment.
Anatomic region Description
Neck -Tumor encasing carotid artery,
vertebral aratery, and/or internal jugular vein.
-Tumor extending to skull base
-Tumor compressing trachea
Cervicothoracic junc -Tumor encasing brachial plexus roots
-Tumor encasing subclavian vessels,
vertebral artery,
and/or carotid artery
-Tumor compressing trachea
Thorax -Tumor encasing aorta and/or major branches
-Tumor compressing trachea and/or principal bronchi
-Lower mediastinal tumor infiltrating costovertebral junction between T9 and T12 vertebral levels
Thoracoabdominal -Tumor encasing aorta and/or vena cava
Junction
Abdomen and pelvis -Tumor infiltrating porta hepatis and/or hepatoduodenal ligament
-Tumor encasing branches of superior mesenteric artery at mesenteric root
-Tumor encasing origin of celiac axis and/or origin of superior mesenteric artery
-Tumor invading one or both renal pedicles
-Tumor encasing aorta and/or vena cava
-Tumor encasing iliac vessels
-Pelvic tumor crossing sciatic notch
Intraspinal extension -Intraespinal tumor extension (whatever the location) provided that more than one-third of spinal canal in axial plane is invaded,
the
perimedullary leptomeningeal spaces are not
visible,
or the spinal cord signal intensity is abnormal (Fig.12).
Infiltration of -Pericardium,
diaphragm,
kidney,
liver, duodenopancreatic block, mesentery.
Adjacent organs
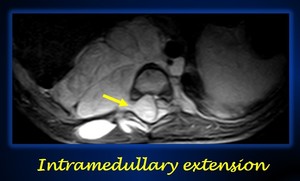
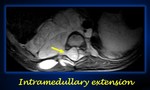
Fig. 12: Axial T2-weighted MR imaging shows an IDRF (intramedullar extension (yellow arrow) in a thoracic neuroblastoma.
References: University Hospital Niño JesĂșs
Special considerations
-The presence of ascities and pleural effusion,
even if malignant cells are present,
is not considered metastasis.
-In case of multifocality,
which usually occurs in familiar or genetic neuroblastomas,
the bigger neuroblastoma is the one to be staged (Fig.
13).
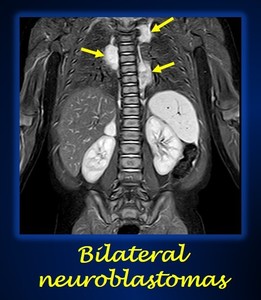
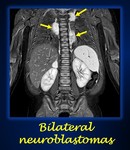
Fig. 13: Coronal STIR image shows multiple bilateral paraspinal thoracic neuroblastomas (yellow arrows) in a patient with PHOX2B mutations.
References: University Hospital Niño JesĂșs
Study of metastasis
Metastasis of this tumor may occur by lymphatic via (linfadenopathies are present in up to 30% cases as previously mentioned),
or hematogenously.
The most common site of metastasis is the bone marrow (70%),
and its involvement has to be assessed by both imaging and bone marrow aspirate and biopsy.
Other sites of metastasis in frequence are the skeletal bone (50%),
and the liver,
where massive liver metastases can occcur leading to the so called âPepper syndromeâ.
Metastatic disease to the lungs and brain parenchyma is rare and more frequently seen in end-stage disease,
and therefore,
it is not systematically screened at diagnosis.
(Figs.
14,15,16).
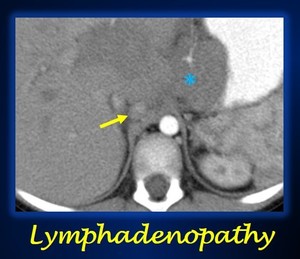
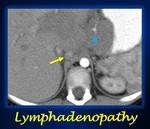
Fig. 14: Axial contrast enhanced abdominal CT in arterial phase shows a retrocaval linfadenopathy (yellow arrow) in a patient with a neuroblastoma (blue asterisk).
References: University Hospital Niño JesĂșs
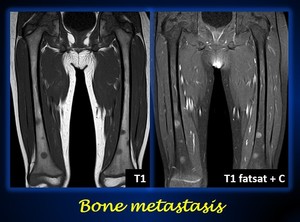
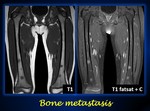
Fig. 15: Bone metastasis of neuroblastoma of the patient of Fig. 14
Coronal T1-weighted MRI of both femurs show multiple round hypointense lesions in the medullary bone. The lesions show enhancement after administration of intravenous gadolinium.
References: University Hospital Niño JesĂșs
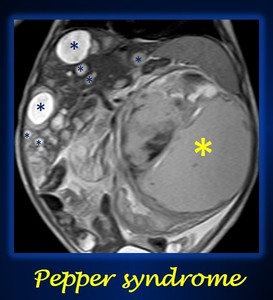
Fig. 16: Coronal T2 weighted abdominal MRI shows multiple cystic liver metastasis (blue asterisks) in an abdominal left neuroblastoma (yellow asterisk).
References: University Hospital Niño JesĂșs
Distant metastasis must be assessed with MIBG nuclear scintography,
as neuroblastoma is a tumor that secretes cathecolamines and shows MIBG avidity.
As 10% of neuroblastomas are non-MIBG avid,
MIBG imaging must be performed before resection of the primary tumor is performed.
Patients with non-MIBG avidity should be assessed with 99m-TC- MDP bone scintigraphy.
Brain scans are only performed if neurological symptoms other than spinal cord compression or MIBG/bone scan uptake of the skull base or orbits are present.