Ultrasonography,
although having poor overall sensitivity in the detection of Retroperitoneal Fibrosis,
is usually the first-line study performed to assess the presence of hydronephrosis or associated aortic aneurysm.
A well-defined hypoechoic or isoechoic band anterior to the aorta,
IVC and iliac vessels may be depicted,
sometimes misinterpreted as a retroperitoneal haematoma,
lymphadenopathies or solid neoplasm [Fig.
1].
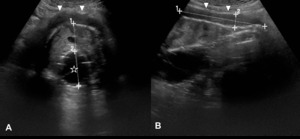
Fig. 1: Idiopathic Retroperitoneal Fibrosis on ultrasound (arrowheads) in a patient with an aortic aneurysm submitted to an endovascular aneurysm repair (star). Note that it presents as a homogeneously hypoechoic band enfolding the aorta on axial [A] and longitudinal [B] planes. This finding was initially interpreted as an endoleak.
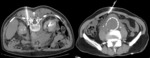
Computed tomography (CT) is the pillar of noninvasive diagnosis of Idiopathic RPF,
as it allows evaluation of its location,
extent and effect on adjacent organs and vascular structures.
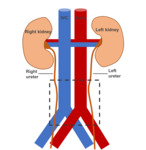
The typical findings are a well-delimited but irregular soft-tissue mass that is isodense to the surrounding muscles and extends from the infra-renal aorta to the iliac vesses,
wraping the great vessels but sparing their posterior aspect.
The centre of the mass is generally at the level of the aortic bifurcation and it often envelops the IVC and the ureters,
causing medial deviation of the latter [Figs.
2 and 3].
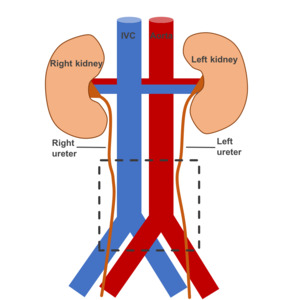
Fig. 2: Typical location of Idiopathic Retroperitoneal Fibrosis, represented by the area inside the dashed rectangle.
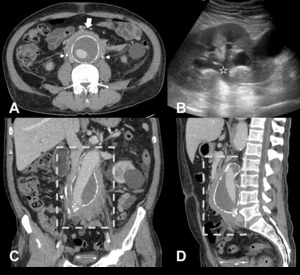
Fig. 3: Idiopathic retroperitoneal fibrosis causing bilateral ureteral obstruction in a 68-year old male patient with an aortic aneurysm. [A] - the retroperitoneal mass (arrow) wraps the infrarenal aorta while sparing the posterior border. Two ureteral stents are depicted (arrowheads). [B] – Ultrasound shows hydronephrosis (star) secondary to ureteral obstruction. [C] and [D] – Coronal and sagittal reformations show the typical extent of Idiopathic RPF (dashed rectangle).
The degree of soft tissue enhancement after intravenous contrast administration correlates with the activity of the disease.
Avid enhancement is seen in the early stages due to the marked inflammatory/fibrotic process whereas in the late inactive stages there is discrete or absent enhancement [Figs.
4 and 5].
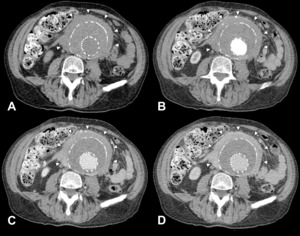
Fig. 4: Idiopathic Retroperitoneal Fibrosis on CT (same case illustrated in Fig. 1). The mass wrapping the infra-renal aorta (arrowheads) has spontaneous attenuation similar to the paraspinal muscles [A]. The dynamic study in the arterial [B], portal [C] and late [D] phases demonstrated a mild, homogeneous and progressive enhancement.
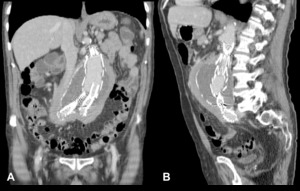
Fig. 5: Coronal [A] and sagittal [B] CT reformations of the case illustrated in Fig. 4. The typical location of Idiopathic RPF is depicted, with the centre of the mass at the level of the aortic bifurcation.
Atypical locations of Idiopathic RPF (eg.
caudal extension to the pelvis or cranially to the level of the renal hila) are uncommon and represent a diagnostic challenge.
RPF secondary to drugs (namely ergot alkaloids,
dopamine agonists and asbestos) is usually radiologically indistinguishable from the idiopathic form.
Secondary forms due to infection,
trauma/surgery or radiotherapy depend respectively on the extent of the disease,
location of the hematoma and the radiation field utilized [Fig.
6].
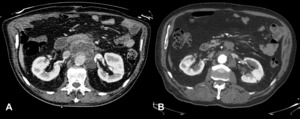
Fig. 6: [A] – Contrast-enhanced CT revealed an infected aortic graft leak with abscesses (stars) collected along the anterior and left lateral borders of the aorta in a 75 year-old male patient. [B] – Secondary Retroperitoneal Fibrosis (arrowheads) was depicted in the same location on follow-up examination (one year later, after antibiotherapy and percutaneous drainage of the collections).
Erdheim-Chester disease may also cause secondary RPF,
normally with a more cranial location and a characteristic perirenal predilection [Fig.
7].
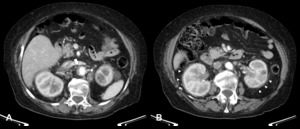
Fig. 7: Secondary Retroperitoneal Fibrosis due to Erdheim-Chester disease in a 83-year-old female patient. When the aorta is involved, it is normally at a higher level than IRPF [A, arrow]. The peri-renal location is characteristic [B, arrowheads].
Imaging features aside from the atypical location that should prompt suspicion for a malignant aetiology [Figs.
8-10] are:
- Peripheral lobulation or nodularity;
- Eccentric location in the retroperitoneum (not centred on the aorta);
- Heterogeneous texture or enhancement;
- Mass effect on neighbouring structures (e.g.
anterior dislocation of the great vessels - floating aorta sign - or lateral deviation of the ureters)
- Presence of lympadenopathies.
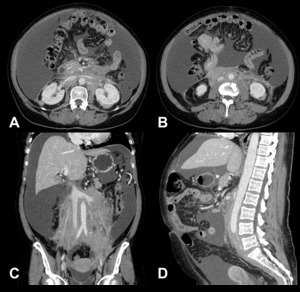
Fig. 8: Secondary Retroperitoneal fibrosis in a 59-year-old female patient due to a metastatic carcinoma of unknown primary origin (at the time of publication). Note the involvement at the level of the renal hila [A], the floating aorta sign [B, arrow] and the extent of the disease on coronal [C] and sagittal [D] reformations. Massive ascites is evident.
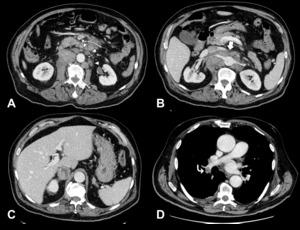
Fig. 9: Secondary Retroperitoneal fibrosis due to a Diffuse Large B-Cell Lymphoma. From caudal to cranial, it is possible to identify multiple lymphadenopathies [A, arrowheads], retroperitoneal fibrosis at the level of the renal arteries with minimal floating aorta sign [B, arrow], tumoural invasion of the inferior vena cava [C, star] and subsequent pulmonary embolism [D, curved arrow].
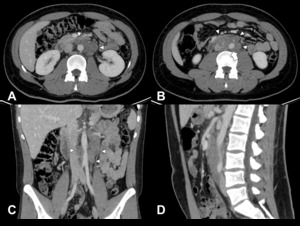
Fig. 10: Secondary Retroperitoneal Fibrosis in a 21 year-old male patient due to a metastatic testicular mixed germ cell tumour with predominant embryonal carcinoma component. Note the peripheral lobulation / nodularity of the mass [A-C, arrowheads] and the ”floating aorta” sign [B, arrow]. Coronal [C] and sagittal [D] CT reformations show the craniocaudal extent of the disease.
Malignant RPF is usually bulkier and its mass effect causes anterior displacement of the aorta and inferior vena cava.
This vascular displacement is likely to be caused by lymph nodes that lie posterior to the great vessels and its commonly called the floating aorta sign.
In Idiopathic RPF (and some benign secondary forms),
as there is a purely fibrotic process,
the great vessels are tethered to the underlying vertebrae,
which explains the usual absence of this sign [Fig.
11].
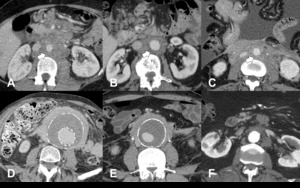
Fig. 11: The floating aorta sign (arrows) is represented in the top three cases, which have a malignant ethiology [A-C]. Compare them with the benign cases from the bottom row [D-F] where this sign is absent.
[A and B] – Lymphomas. [C] – Metastatic carcinoma of unknown origin (same case as in Fig. 8). [D and E] – Idiopathic RPF (same cases as in Figs. 3 and 4). [F] – Secundary RPF due to an infected aortic graft leak (same case as in Fig. 6).
Magnetic ressonance has some advantages over CT,
particularly in patients with severe impairment of the renal patient in which intravenous contrast agents should be avoided.
MRI also shows superior contrast resolution and T2-weighted images allows estimation of the degree of active inflammation (high singal intensity in T2 reflects edema and hypercellularity).
F-FDG PET,
as a whole-body examination,
allows assessment of the full extent and distribution of the inflammatory involvement in idiopathic RPF.
It can also demonstrate infectious,
neoplastic,
or other autoimmune processes with which RPF may be associated.
Biopsy and histopathologic examination of the retroperitoneal tissue [Fig.
12] is indicated to establish the definitive diagnosis in the presence of:
- Suspicious imaging features;
- Atypical location of the mass;
- Progression of the mass;
- Clinical or laboratory finding suggesting underlying malignancy.
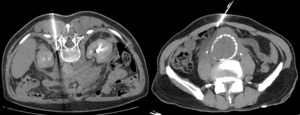
Fig. 12: CT-guided biopsy of two retroperitoneal masses. A – Histopathological study revealed retroperitoneal fibrosis with metastasis from a carcinoma of unknown primary origin (same patient as in Fig. 8). B – Histopathological study confirmed retroperitoneal fibrosis without neoplastic cells (same patient as in Fig. 3).
Treatment in Idiopathic RPF is normally aimed to relieve ureteral obstruction and prevent disease recurrence,
mainly with corticosteroids and/or other immunossupressive drugs.
Surgery (ureterolysis and ureteral transposition) is reserved only for refractory cases.
Management in secondary forms of RPF requires an approach based on the specific cause,
when identified.

![Fig. 1: Idiopathic Retroperitoneal Fibrosis on ultrasound (arrowheads) in a patient with an aortic aneurysm submitted to an endovascular aneurysm repair (star). Note that it presents as a homogeneously hypoechoic band enfolding the aorta on axial [A] and longitudinal [B] planes. This finding was initially interpreted as an endoleak.](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742359?maxheight=150&maxwidth=150)
![Fig. 3: Idiopathic retroperitoneal fibrosis causing bilateral ureteral obstruction in a 68-year old male patient with an aortic aneurysm. [A] - the retroperitoneal mass (arrow) wraps the infrarenal aorta while sparing the posterior border. Two ureteral stents are depicted (arrowheads). [B] – Ultrasound shows hydronephrosis (star) secondary to ureteral obstruction. [C] and [D] – Coronal and sagittal reformations show the typical extent of Idiopathic RPF (dashed rectangle).](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742362?maxheight=150&maxwidth=150)
![Fig. 4: Idiopathic Retroperitoneal Fibrosis on CT (same case illustrated in Fig. 1). The mass wrapping the infra-renal aorta (arrowheads) has spontaneous attenuation similar to the paraspinal muscles [A]. The dynamic study in the arterial [B], portal [C] and late [D] phases demonstrated a mild, homogeneous and progressive enhancement.](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742360?maxheight=150&maxwidth=150)
![Fig. 5: Coronal [A] and sagittal [B] CT reformations of the case illustrated in Fig. 4. The typical location of Idiopathic RPF is depicted, with the centre of the mass at the level of the aortic bifurcation.](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742361?maxheight=150&maxwidth=150)
![Fig. 6: [A] – Contrast-enhanced CT revealed an infected aortic graft leak with abscesses (stars) collected along the anterior and left lateral borders of the aorta in a 75 year-old male patient. [B] – Secondary Retroperitoneal Fibrosis (arrowheads) was depicted in the same location on follow-up examination (one year later, after antibiotherapy and percutaneous drainage of the collections).](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742367?maxheight=150&maxwidth=150)
![Fig. 7: Secondary Retroperitoneal Fibrosis due to Erdheim-Chester disease in a 83-year-old female patient. When the aorta is involved, it is normally at a higher level than IRPF [A, arrow]. The peri-renal location is characteristic [B, arrowheads].](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742366?maxheight=150&maxwidth=150)
![Fig. 8: Secondary Retroperitoneal fibrosis in a 59-year-old female patient due to a metastatic carcinoma of unknown primary origin (at the time of publication). Note the involvement at the level of the renal hila [A], the floating aorta sign [B, arrow] and the extent of the disease on coronal [C] and sagittal [D] reformations. Massive ascites is evident.](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742365?maxheight=150&maxwidth=150)
![Fig. 9: Secondary Retroperitoneal fibrosis due to a Diffuse Large B-Cell Lymphoma. From caudal to cranial, it is possible to identify multiple lymphadenopathies [A, arrowheads], retroperitoneal fibrosis at the level of the renal arteries with minimal floating aorta sign [B, arrow], tumoural invasion of the inferior vena cava [C, star] and subsequent pulmonary embolism [D, curved arrow].](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742363?maxheight=150&maxwidth=150)
![Fig. 10: Secondary Retroperitoneal Fibrosis in a 21 year-old male patient due to a metastatic testicular mixed germ cell tumour with predominant embryonal carcinoma component. Note the peripheral lobulation / nodularity of the mass [A-C, arrowheads] and the ”floating aorta” sign [B, arrow]. Coronal [C] and sagittal [D] CT reformations show the craniocaudal extent of the disease.](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/742368?maxheight=150&maxwidth=150)
![Fig. 11: The floating aorta sign (arrows) is represented in the top three cases, which have a malignant ethiology [A-C]. Compare them with the benign cases from the bottom row [D-F] where this sign is absent.
[A and B] – Lymphomas. [C] – Metastatic carcinoma of unknown origin (same case as in Fig. 8). [D and E] – Idiopathic RPF (same cases as in Figs. 3 and 4). [F] – Secundary RPF due to an infected aortic graft leak (same case as in Fig. 6).](https://epos.myesr.org/posterimage/esr/ecr2018/143863/media/746680?maxheight=150&maxwidth=150)