Below we present a listing of several entities associated with bone marrow edema of the knee,
arranged according to the pathophysiology of the edema for each case (slightly adapted from the first classification described by Eustace[1]).
Several examples collected from our practice are used to illustrate and further discuss these entities:
1.
Traumatic/Degenerative
a.
Osteoarthritis (Fig. 2)
b.
Acute Trabecular Microfractures (Fig. 7, Fig. 8, Fig. 9, Fig. 10,
Fig. 11)
c.
Stress Fatigue Fractures (Fig. 3)
d.
Stress Insufficiency Fractures
i.
SIF (Subchondral Insufficiency Fracture) formerly known as SONK (Spontaneous osteonecrosis of the knee,
also known as Ahlback’s Disease) (Fig. 4,
Fig. 5, Fig. 6)
e.Macroscopic Fractures (Fig. 12,
Fig. 13, Fig. 14)
f.
Symptomatic Ossicles (Fig. 15)
g.Osgood-Schlatter Disease (Fig. 16)
h.Sinding-Larsen-Johansson Syndrome (Fig. 17)
i.
Osteochondritis Dissecans (OCD) (Fig. 18,
Fig. 19)
2.
Congestive
a.
Osteonecrosis (Avascular Necrosis,
AVN)
i. Hemoglobinopathies (Fig. 20)
ii.
Marrow Packing Disorders
iii.
Fat Cell Hypertrophy
iv.
Steroids Use
v.
Pregnancy
3.
Hyperemic/Vasogenic
a.
Inflammatory/Infectious Arthritis (Fig. 21)
b.
Posttraumatic Reflex Sympathetic Dystrophy
c.
Chronic Recurrent Multifocal Osteomyelitis (CRMO)
4.
Tumorigenic
(e.g. Osteosarcoma (Fig. 22), Giant Cell Tumor (Fig. 23), Osteoid Osteoma (Fig. 24),
...)
5.
Atypical/Others
a.
Metabolic Changes
i.
Chronic Kidney Disease
b.
Neurological Changes
i.
Charcot’s Joint
c.
Iatrogenic Changes
i.
Radiotherapy
d.
Idiopathic/Multifactorial Edema
i.
Transient Bone Marrow Edema Syndrome
(also known as Transient Osteoporosis (TO)) (Fig. 25)
a.
Regional Migratory Osteoporosis (RMO)
i.
Extra-articular
ii.
Intra-articular (Fig. 26)
2.
Complex Regional Pain Syndrome (CRPS,
comprehends the terms Reflex Sympathetic Dystrophy,
Algoneurodystrophy,
and Sudeck’s Atrophy)
3.
Calcineurin Inhibitor Pain Syndrome (CIPS)
ii.
Disuse Osteoporosis (DO) (Fig. 27)
1.
Traumatic/Degenerative
a.
Osteoarthritis
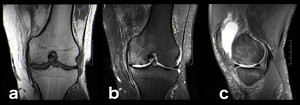
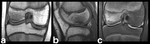
Fig. 2: Osteoarthritis (OA): 58-year-old man with extensive chondral defect at the weight-bearing area of the medial femoral condyle, associated with small marginal osteophytes and secondary subchondral bone marrow edema. Osteoarthritis isn’t usually classified alongside traumatic etiologies but rather as a degenerative condition, truth is that the development of BME on OA is secondary to repetitive microtrauma on weight-bearing structures[2] (a: coronal T1-WI; b: coronal PD-FS-WI; c: sagittal PD-FS-WI).
1.
Traumatic/Degenerative
b.
Acute Trabecular Microfractures
c.
Stress Fatigue Fractures
d.
Stress Insufficiency Fractures
i.
SIF (Subchondral Insufficiency Fracture) formerly known as SONK (Spontaneous osteonecrosis of the knee,
also known as Ahlback’s Disease)
MRI findings of traumatic bone edema (commonly referred to as a bone bruise,
contusion or trabecular microfracture) becomes evident within hours of the injury.[4]
It is important to note that acute trabecular microfractures and stress fractures are not synonyms.
It’s even common to witness “stress fractures” or “insufficiency fractures” reported as the same entity,
although insufficiency fractures are actually a subtype of stress fractures.
Stress fractures can be classified as fatigue or insufficiency fractures,
the first ones are caused by repeated overloading of normal bony structures while the latter ones occur spontaneously on an already impaired bone (e.g. because of osteoporosis or osteomalacia),
without trauma or overloading.[5]
Trabecular microfractures (traumatic) and stress-related fractures (chronic overloading on normal bone or normal loading on impaired bone) can’t be distinguished on MRI,
only the patient’s age and history can help to differentiate them.[5]
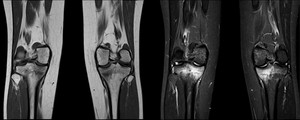
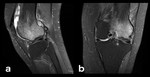
Fig. 3: Stress (fatigue) fractures: T1-weighted and STIR coronal images of the knees of a 25-year-old woman, who had recently engaged in strenuous physical activity at the gym without adequate conditioning. She complained of medial tibial pain bilaterally. Medial proximal stress fractures are evident in both tibias.
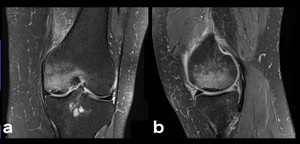
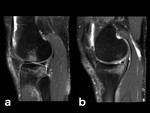
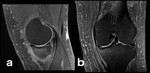
Fig. 4: SIF: in the past, every time a subchondral lesion as this one was observed in the medial femoral condyle it was labeled as spontaneous osteonecrosis of the knee (SONK). Now it’s considered to be an insufficiency fracture.[5] These insufficiency fractures typically develop at the medial femoral condyle and in the femoral head and are present in older individuals (they are related to osteoporosis and altered biomechanics, as in unstable meniscal lesions).[5] Similar to other stress fractures, a hypointense fracture line is surrounded by hyperintense bone marrow edema on water-sensitive sequences (a, b: coronal and sagittal PD-FS-WI).
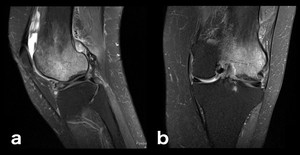
Fig. 5: Long-standing SIF with subchondral collapse and associated large area of edema on the lateral femoral condyle (less common than the medial location shown on the previous image).
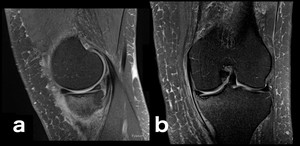
Fig. 6: Less common form of SIF on the medial tibial plateau (a, b: sagittal and coronal PD-FS-WI).
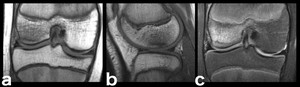
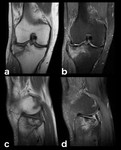
Fig. 7: Trabecular microfracture: 15-year-old male adolescent with acute pain following traumatic event while playing soccer: hypointense microfracture on femoral medial condyle both on T1-WI (a, b) and on T2-FS-WI (c). On the water-sensitive sequence (T2-FS-WI) the hyperintense bone marrow edema surrounding the fracture is evident, while on the T1-WI’s it translates as an ill-defined hypointense area on the otherwise hyperintense marrow of the distal femoral epiphysis and proximal tibia.
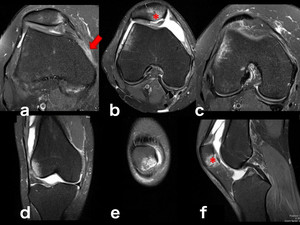
Fig. 8: Traumatic bone edema, patellar dislocation typical pattern: PD-FS-WI on axial (a, b, c), coronal (d, e) and sagittal planes (f) of a young adult with transient patellar dislocation and resulting typical contusion pattern: anterolateral aspect of the lateral femoral condyle and inferomedial aspect of the patella. It’s more common on teenagers or young adults who perform sports that require a twisting motion of the knee while it’s flexed. Associated lesions can be present, as in this patient with osteochondral injury of the patella (*) and tear of the medial patellofemoral ligament (MPFL) (arrow).
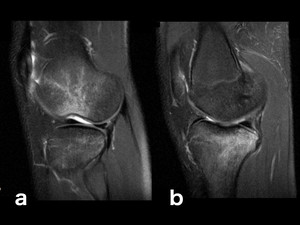
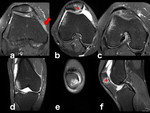
Fig. 9: Traumatic bone edema, typical pivot shift pattern: PD-FS-WI on sagittal planes. This type of non-contact injury is commonly seen in skiers or American football players.[3] This pattern results from a valgus load applied to the flexed or semi-flexed knee, combined with external rotation of the tibia or internal rotation of the femur.[3] The edema pattern involves the posterior aspect of the lateral tibial plateau and the midportion of the lateral femoral condyle near the condylopatellar sulcus (a).[3] The degree of flexion of the knee determines the exact location of the lateral femoral condyle injury (more flexion results in a more posterior bruise, whereas less flexion results in a more anteriorly located edema).[3] Sometimes, as shown in a, a lateral femoral condyle impaction fracture occurs. Another frequently associated site of bone contusion is the posterior aspect of the medial tibial plateau (as shown in b), thought to result from contrecoup forces in the medial compartment at the resolution of the forced valgus forces (b). The ACL is frequently disrupted in this kind of trauma[3], therefore when this pattern is recognized a thorough evaluation of the ligament should be performed.
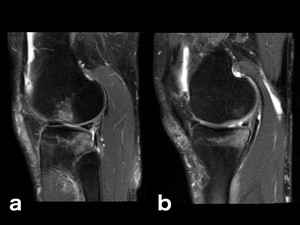
Fig. 10: Traumatic bone edema, typical pivot shift pattern: T2-FS-WI on sagittal planes, different patient from figure 6, same mechanism and similar findings (albeit without fracture on this case).
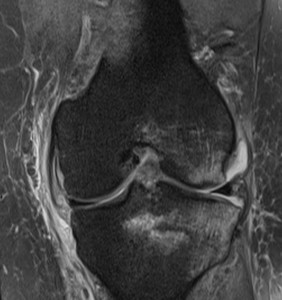
Fig. 11: Traumatic bone edema, typical clip injury pattern: Trauma sustained by a 23-year-old man while playing rugby. Coronal intermediate-weighted fat suppressed MR image reveals bone marrow edema in the lateral femoral condyle and lateral tibial plateau secondary to direct contusion. The valgus force associated with the mechanism of trauma stresses the MCL, which may tear, as documented on this patient.
1.
Traumatic/Degenerative
e.
Macroscopic Fractures
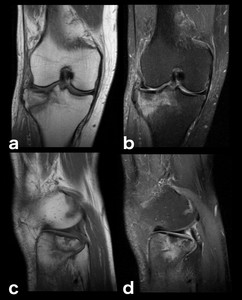
Fig. 12: Traumatic bone edema, macroscopic fracture (with cortical disruption): This patient had a very faint fracture line on conventional radiography, barely noticeable (not shown here). On MR imaging the contusional bone edema surrounding the fracture makes it impossible to miss (PD-FS-WI hyperintensity and T1-WI hypointensity on the lateral tibial plateau). The fracture line is hypointense on T1-WI (a, c) and isointense on PD-FS-WI (b, d).
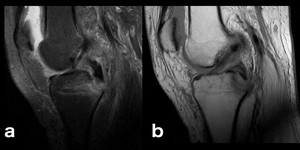
Fig. 13: Traumatic bone edema, macroscopic fracture (PCL avulsion): When the posterior cruciate ligament is not retracted it can be hard to identify an avulsion fracture of its insertion. The surrounding bone edema makes it an easier diagnosis.
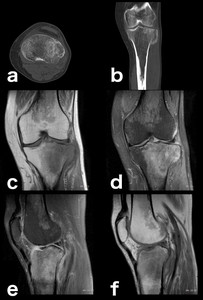
Fig. 14: Traumatic bone edema, macroscopic subacute fracture: Even with a little less than obvious fracture line on the CT scan (a, b), MRI bone edema points out where to look (c, d, e, f).
1.
Traumatic/Degenerative
f. Symptomatic Ossicles
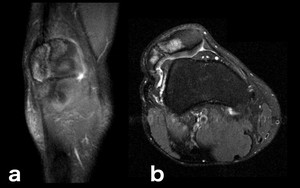
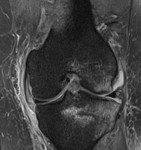
Fig. 15: Up to 2% of all bipartite patellae become painful because of stress across the synchondrosis between the ossicle and the patella, resulting from repetitive pull by the vastus lateralis muscle.[5] On this patient the repetitive overload on the synchondrosis has resulted in local bruising (hyperintense edema) in both the unfused accessory ossification center and the remaining patella (a, b: coronal and axial PD-FS-WI).
1.
Traumatic/Degenerative
g.
Osgood-Schlatter Disease
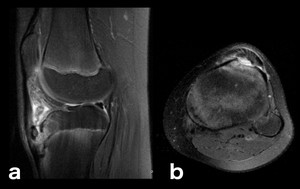
Fig. 16: Osgood-Schlatter’s disease is a chronic avulsion injury thought to result either from repetitive microtrauma and traction on the tibial tubercle or direct trauma to the patellar tendon at its insertion at the tibial tubercle. It’s typically seen in active adolescents, particularly those who participate in sports with strenuous involvement of the knee.[6] The resulting edema on the avulsion site can be an important clue to make this diagnosis, as shown above (a, b: sagittal and axial PD-FS-WI).
1.
Traumatic/Degenerative
h.
Sinding-Larsen-Johansson Syndrome
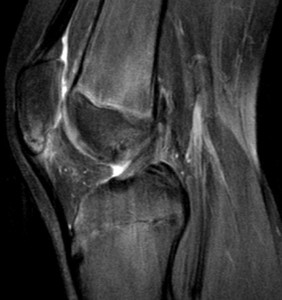
Fig. 17: This 13-year-old boy has Sinding-Larsen-Johansson Syndrome, a chronic traction injury of the immature osteotendinous junction of the proximal end of the patellar tendon as it inserts into the inferior pole of the patella (signaled by the lower pole edema on this sagittal PD-FS-WI).
1.
Traumatic/Degenerative
i.
Osteochondritis Dissecans (OCD)
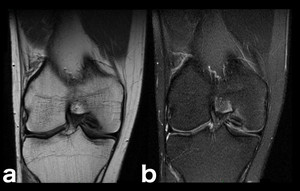
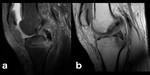
Fig. 18: Osteochondritis dissecans (OCD). OCD etiology is unknown and likely multifactorial, with injury, vascular factors, pressure changes, developmental differences, and genetics all contributing.[5] The most common site for OCD of the knee is the lateral aspect of the medial femoral condyle (75%).[5] Here is shown a characteristic MRI appearance, typically without additional lesions: a small area of bone marrow edema is evident on b, surrounding the osteochondral fragment. (a, b: coronal T1-WI and coronal PD-FS-WI).
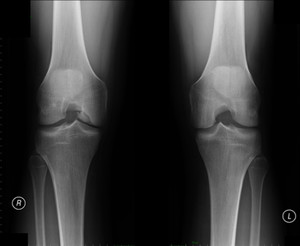
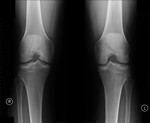
Fig. 19: Osteochondritis dissecans (OCD). Same patient as in fig. 18. An osteochondral fragment on the lateral aspect of the right femoral medial condyle is seen on conventional radiography.
2.
Congestive
a.
Osteonecrosis (Avascular Necrosis,
AVN)
i.
Hemoglobinopathies
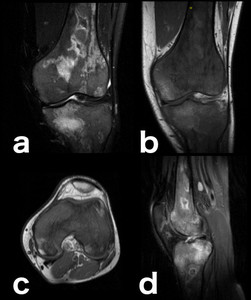
Fig. 20: Patients with hemoglobinopathies (for instance sickle-cell anemia, as in this case) are prone to develop osteonecrosis secondary to capillary obstruction. This obstruction leads to raised capillary hydrostatic pressure, which in turn is responsible for the hyperintense edema surrounding the hypointense bone infarcts on water-sensitive sequences. This is an example of BME of a congestive nature (due to impaired removal of fluid from the marrow space capillary bed). This patient also presents extensive red marrow reconversion as depicted on the marrow signal in T1-WI’s (much lower than surrounding fat tissue and slightly higher than muscle tissue) and DP-FS-WI’s (doesn’t fully saturate, remaining slightly hyperintense) (a: coronal DP-FS-WI; b: coronal T1-WI; c: axial T1-WI; d: sagittal DP-FS-WI).
3.
Hyperemic/Vasogenic
a.
Inflammatory/Infectious Arthritis
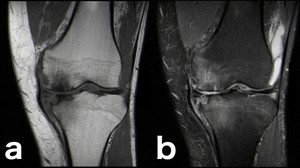
Fig. 21: This patient’s inflammatory arthritis is responsible for the bone marrow edema surrounding the erosive changes on the medial femoro-tibial compartment. It’s an example of BME of a vasogenic nature (secondary to an increased delivery of blood and serum to the marrow space capillary bed) (a, b: coronal T1-WI and PD-FS-WI).
4.
Tumorigenic
(e.g. Osteosarcoma, Giant Cell Tumor, Osteoid Osteoma,
...)
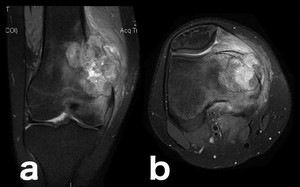
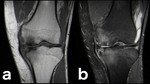
Fig. 22: Osteosarcoma (a, b: coronal and axial PD-FS-WI): tumorigenic edema is secondary to direct capillary trauma from trabecular destruction with the release of intravascular fluid and associated hemorrhage.[2] It may be difficult to differentiate surrounding edema from tumor invasion.
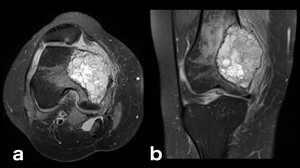
Fig. 23: Giant Cell Tumor (PD-FS-WI): tumorigenic edema is also present.
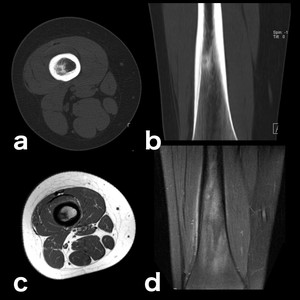
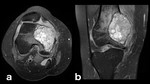
Fig. 24: Osteoid Osteoma: in these benign tumors edema can be secondary to an induced inflammatory response by prostaglandin E2 [2] (a, b: axial and coronal CT scan; c, d: axial T1-WI and coronal PD-FS-WI).
5.
Atypical/Others
d.
Idiopathic/Multifactorial Edema
i. Transient Bone Marrow Edema Syndrome
(also known as Transient Osteoporosis (TO))
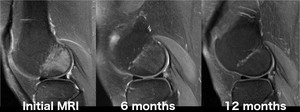
Fig. 25: Transient Bone Marrow Edema Syndrome (Transient Osteoporosis): sagittal intermediate-weighted fat-suppressed MR image of a 30-year-old woman with acute knee pain shows bone marrow edema involving the non-weight-bearing area of the lateral femoral condyle, which underwent almost complete spontaneous resolution over the course of 1 year. Transient Bone Marrow Edema Syndromes are self-limiting entities which manifest as arthralgia associated with bone marrow edema evidence on MRI studies.[8] Its etiology remains uncertain.[8]
5.
Atypical/Others
d.
Idiopathic/Multifactorial Edema
i. Transient Bone Marrow Edema Syndrome
(also known as Transient Osteoporosis (TO))
a. Regional Migratory Osteoporosis (RMO)
ii. Intra-articular
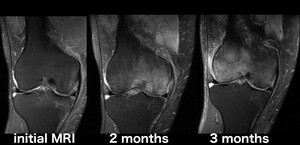
Fig. 26: Regional Migratory Osteoporosis (a Transient Bone Marrow Edema Syndrome with location shifting) (serial coronal PD-FS-WI): In some cases a location shift of the bone marrow edema is seen on serial MRIs, this is known as Regional Migratory Osteoporosis (RMO).[8] Although it’s more common to witness a spontaneous resolution of the transient bone marrow edema syndrome, a shift to a different joint can occur in a small fraction of cases, or (even rarer) a shift to a different site within the same articulation (i.e. intra-articular shifting), as seen on this patient.[8] On this intra-articular RMO case there was no trauma history, the edema started on the medial femoral condyle and 3 months later the findings had shifted to the lateral condyle.
5.
Atypical/Others
d.
Idiopathic/Multifactorial Edema
ii. Disuse Osteoporosis (DO)
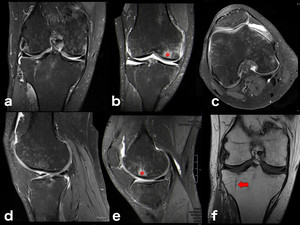
Fig. 27: Disuse Osteoporosis (DO): the confluent and patchy moderately hyperintense pattern here shown in all sequences on the epiphysis is typical of transient osteoporosis following prolonged immobilization. Resuming activity produces bigger stress in the disused bone than in the normal bone because the trabeculae, which have to support the load, are less and weaker, hence bone edema ensues (a stress reaction is also seen on the medial condyle and marked with an *).[7] An healed lateral tibial plateau fracture, responsible for the immobilization, is also apparent (arrow). (a-e: PD-FS-WI; f: T1-WI). It’s also worth noting that Complex Regional Pain Syndrome (CRPS, comprehending the terms reflex sympathetic dystrophy, algoneurodystrophy, and Sudeck’s atrophy) has the same MRI bone edema pattern as DO but it’s pathophysiology is not related to disuse demineralization and it’s always painful (DO is an incidental finding in an asymptomatic patient with known immobilization history).[8]
Bone Marrow Edema Pitfalls
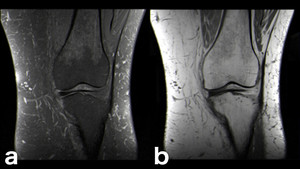
Fig. 28: Normal red marrow: this is a possible pitfall when looking for bone marrow edema. Hematopoietic red marrow (either due to normal distribution in younger patients or marrow reconversion in adults) has intermediate signal intensity both on T1-WI (b) and on water-sensitive sequences (a: PD-FS-WI).[4] The characteristic pattern of distribution (usually on the metaphysis) and the higher signal intensity of normal red marrow relative to muscle on T1-WI (as opposed to pathologic marrow that is typically iso- or hypointense to muscle tissue on T1-WI) should enable prompt recognition of this normal finding.[4]

![Fig. 2: Osteoarthritis (OA): 58-year-old man with extensive chondral defect at the weight-bearing area of the medial femoral condyle, associated with small marginal osteophytes and secondary subchondral bone marrow edema. Osteoarthritis isn’t usually classified alongside traumatic etiologies but rather as a degenerative condition, truth is that the development of BME on OA is secondary to repetitive microtrauma on weight-bearing structures[2] (a: coronal T1-WI; b: coronal PD-FS-WI; c: sagittal PD-FS-WI).](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764847?maxheight=150&maxwidth=150)
![Fig. 9: Traumatic bone edema, typical pivot shift pattern: PD-FS-WI on sagittal planes. This type of non-contact injury is commonly seen in skiers or American football players.[3] This pattern results from a valgus load applied to the flexed or semi-flexed knee, combined with external rotation of the tibia or internal rotation of the femur.[3] The edema pattern involves the posterior aspect of the lateral tibial plateau and the midportion of the lateral femoral condyle near the condylopatellar sulcus (a).[3] The degree of flexion of the knee determines the exact location of the lateral femoral condyle injury (more flexion results in a more posterior bruise, whereas less flexion results in a more anteriorly located edema).[3] Sometimes, as shown in a, a lateral femoral condyle impaction fracture occurs. Another frequently associated site of bone contusion is the posterior aspect of the medial tibial plateau (as shown in b), thought to result from contrecoup forces in the medial compartment at the resolution of the forced valgus forces (b). The ACL is frequently disrupted in this kind of trauma[3], therefore when this pattern is recognized a thorough evaluation of the ligament should be performed.](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764885?maxheight=150&maxwidth=150)
![Fig. 4: SIF: in the past, every time a subchondral lesion as this one was observed in the medial femoral condyle it was labeled as spontaneous osteonecrosis of the knee (SONK). Now it’s considered to be an insufficiency fracture.[5] These insufficiency fractures typically develop at the medial femoral condyle and in the femoral head and are present in older individuals (they are related to osteoporosis and altered biomechanics, as in unstable meniscal lesions).[5] Similar to other stress fractures, a hypointense fracture line is surrounded by hyperintense bone marrow edema on water-sensitive sequences (a, b: coronal and sagittal PD-FS-WI).](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764879?maxheight=150&maxwidth=150)
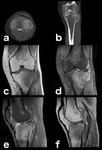
![Fig. 15: Up to 2% of all bipartite patellae become painful because of stress across the synchondrosis between the ossicle and the patella, resulting from repetitive pull by the vastus lateralis muscle.[5] On this patient the repetitive overload on the synchondrosis has resulted in local bruising (hyperintense edema) in both the unfused accessory ossification center and the remaining patella (a, b: coronal and axial PD-FS-WI).](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764893?maxheight=150&maxwidth=150)
![Fig. 16: Osgood-Schlatter’s disease is a chronic avulsion injury thought to result either from repetitive microtrauma and traction on the tibial tubercle or direct trauma to the patellar tendon at its insertion at the tibial tubercle. It’s typically seen in active adolescents, particularly those who participate in sports with strenuous involvement of the knee.[6] The resulting edema on the avulsion site can be an important clue to make this diagnosis, as shown above (a, b: sagittal and axial PD-FS-WI).](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764927?maxheight=150&maxwidth=150)
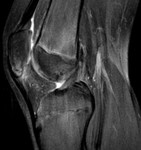
![Fig. 18: Osteochondritis dissecans (OCD). OCD etiology is unknown and likely multifactorial, with injury, vascular factors, pressure changes, developmental differences, and genetics all contributing.[5] The most common site for OCD of the knee is the lateral aspect of the medial femoral condyle (75%).[5] Here is shown a characteristic MRI appearance, typically without additional lesions: a small area of bone marrow edema is evident on b, surrounding the osteochondral fragment. (a, b: coronal T1-WI and coronal PD-FS-WI).](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764934?maxheight=150&maxwidth=150)
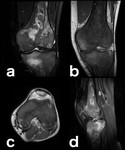
![Fig. 22: Osteosarcoma (a, b: coronal and axial PD-FS-WI): tumorigenic edema is secondary to direct capillary trauma from trabecular destruction with the release of intravascular fluid and associated hemorrhage.[2] It may be difficult to differentiate surrounding edema from tumor invasion.](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764946?maxheight=150&maxwidth=150)
![Fig. 24: Osteoid Osteoma: in these benign tumors edema can be secondary to an induced inflammatory response by prostaglandin E2 [2] (a, b: axial and coronal CT scan; c, d: axial T1-WI and coronal PD-FS-WI).](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764948?maxheight=150&maxwidth=150)
![Fig. 25: Transient Bone Marrow Edema Syndrome (Transient Osteoporosis): sagittal intermediate-weighted fat-suppressed MR image of a 30-year-old woman with acute knee pain shows bone marrow edema involving the non-weight-bearing area of the lateral femoral condyle, which underwent almost complete spontaneous resolution over the course of 1 year. Transient Bone Marrow Edema Syndromes are self-limiting entities which manifest as arthralgia associated with bone marrow edema evidence on MRI studies.[8] Its etiology remains uncertain.[8]](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764951?maxheight=150&maxwidth=150)
![Fig. 26: Regional Migratory Osteoporosis (a Transient Bone Marrow Edema Syndrome with location shifting) (serial coronal PD-FS-WI): In some cases a location shift of the bone marrow edema is seen on serial MRIs, this is known as Regional Migratory Osteoporosis (RMO).[8] Although it’s more common to witness a spontaneous resolution of the transient bone marrow edema syndrome, a shift to a different joint can occur in a small fraction of cases, or (even rarer) a shift to a different site within the same articulation (i.e. intra-articular shifting), as seen on this patient.[8] On this intra-articular RMO case there was no trauma history, the edema started on the medial femoral condyle and 3 months later the findings had shifted to the lateral condyle.](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764952?maxheight=150&maxwidth=150)
![Fig. 27: Disuse Osteoporosis (DO): the confluent and patchy moderately hyperintense pattern here shown in all sequences on the epiphysis is typical of transient osteoporosis following prolonged immobilization. Resuming activity produces bigger stress in the disused bone than in the normal bone because the trabeculae, which have to support the load, are less and weaker, hence bone edema ensues (a stress reaction is also seen on the medial condyle and marked with an *).[7] An healed lateral tibial plateau fracture, responsible for the immobilization, is also apparent (arrow). (a-e: PD-FS-WI; f: T1-WI). It’s also worth noting that Complex Regional Pain Syndrome (CRPS, comprehending the terms reflex sympathetic dystrophy, algoneurodystrophy, and Sudeck’s atrophy) has the same MRI bone edema pattern as DO but it’s pathophysiology is not related to disuse demineralization and it’s always painful (DO is an incidental finding in an asymptomatic patient with known immobilization history).[8]](https://epos.myesr.org/posterimage/esr/ecr2018/143516/media/764953?maxheight=150&maxwidth=150)



























