Meningiomas are non-glial neoplasms originating from the meningocytes or the arachnoid cap cells of the meninges.
They are the most frequent primary brain tumors and represent approximately 20-30% of all primary intracranial neoplasms [1-2] with an annual incidence of about 4-5 per 100.000 individuals [3].
The incidence increases progressively with age,
change by race (with a black to white ratio of 2:1 [7]) and sex,
being more common in women (female to male ratio of about 3:1 [8]).
Meningiomas can be multiple in 5-40% of cases,
especially in association to Neurofibromatosis type 2 (NF2).
Meningiomas are usually slow growing and tend to be asymptomatic,
being their clinical behavior,
mortality and morbidity rates difficult to assess.
Symptoms develop depending on the location of the mass and time of evolution.
Meningiomas can develop from anywhere in the dura,
being common in sites of dural reflection (falx cerebri,
tentorium cerebelli,
venous sinuses).
On 2007,
WHO classified meningiomas into 3 histological grades and 15 subtypes ( Table 1 )[1-2].
Most meningiomas correspond to WHO Grade I Meningiomas (Benign) [1] and show a benign biologic behavior.
Grade II Meningiomas (Atypical) and Grade III Meningiomas (Anaplastic) account for 4.7 - 7.2% and 1.0 - 2.8% of all meningiomas respectively [4].
Of these groups (WHO Grades II and III Meningiomas),
40-80% show an aggressive behavior,
defined as recurrence (after surgical treatment) or progression (after surgical treatment or initial diagnosed) [1].
Generally,
meningiomas are easily diagnosed,
being Computed Tomography (CT) usually the first modality on which meningiomas are detected.
On non-contrast CT,
they are slightly hyperdense to normal brain,
having some calcification in 20-30%.
On post-contrast CT,
meningiomas tend to have brightly homogeneous enhancement.
Some of them demonstrate more heterogeneity and less intense enhancement.
Hyperostosis can also be present on CT.
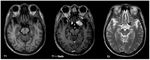
On MRI,
meningiomas typically appear as homogeneous and well circumscribed extra-axial masses with dural base.
( Table 2 ) and ( Fig. 1 ) [9-10] show MRI signal characteristic of typical meningiomas:
A few studies have investigated the MRI features of WHO II and III meningiomas and their correlation with anatomopathological findings assessed using WHO classification,
in order to predict aggressive biological meningioma behavior (recurrence/progression) but there is a lack of longitudinal follow-up studies to clearly distinguish these tumors.
It is known that meningiomas show high uptake of choline due to the overall increased metabolism of cell membranes [5] within these tumors.
18F-Choline shows a low physiological uptake in the brain and is thus able to show a tumor-to-background ratio much higher than that of 18F-FDG [6].
Recently,
it has been suggested that increased 18F-choline uptake in meningiomas relates to a higher mitotic index (cellularity) [11] so that 18F-choline PET could provide added value in identifying recurrent/progressive meningiomas,
but the evidence here is very limited.
Our purpose has been therefore to study MRI features of those meningiomas having increased 18F-choline PET uptake,
in order to further characterize recurrent/progressive meningiomas.