INDICATIONS
- Indeterminate pulmonary lesions in patients with or without known malignancy,
especially those that would require treatments other than surgery (chemotherapy,
radiotherapy,
antibiotics)
- Molecular testing in a known malignacy,
mainly non-small cell lung cancer (NSCLC),
to assess the presence of driver mutation (ALK,
ROS 1,
EGFR,
PDL1…) in order to offer a personalized targeted therapy.
CONTRAINDICATIONS
- A unique functional lung.
- Uncooperative patients (mental status,
mechanical ventilation,
intractable cough…)
- Lesions suspected of being vascular in origin.
- Uncorrectable coagulopathy.
Anticoagulant or antiplatelet therapy should be suspendend for a period depending on their pharmacological properties.
We don´t consider necessary to suspend aspirin.
- Respiratory compromise (i.e.
severe emphysema).
PRIOR TO PROCEDURE
For a proper selection of candidates we recommend a thorough consideration of the indications and patient history.
A careful review of recent studies (chest CT or PET-CT) is necessary for planning the best approach to sample the lesion (in case of multiple lesions it should be the largest and more accesible).
More over,
PET-CT findings can potentially improve the diagnostic accuracy of the biopsy as it allows a functional point on view and helps to differentiate necrotic zones from viable tumor areas.
It's best to avoid necrotic tissue as the diagnostic value of the samples is low.
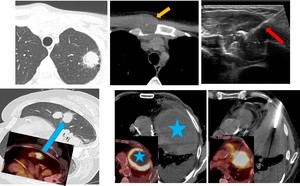
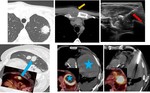
Fig. 1: Review of recent images is necesary to plann the procedure. Biopsy of possible sites of nodal or extranodal metastasis instead of the lung mass can establish histology and stage the patient in a single procedure.
If multiple nodule are encountered, PET-CT may help to choose the more suitable in base of 18F-FDG uptake. Also display the tumor vaibility areas when we are dealing with a large necrotic mass.
If there is suspicion of metastatic lung cancer,
sampling the metastases has proven to be less risky while providing tissular diagnosis and staging information (i.e.
supraclavicular nodes,
bone or liver metastases…).
The goal should be to obtain an histological diagnosis with minimal invasion and complications.
Never forget to obtain an informed consent prior to the procedure.
TECHNIQUE - PREPARATION
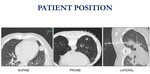
Patient positioning (prone,
supine or lateral decubitus) depends on tumor location and previously planned entry site and needle trajectory.
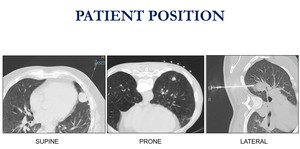
Fig. 2: Patient position depends on lesion location.
The prone decubitus is preferred.
The posterior ribs move less and the intercostal spaces are wider compared to anterior ribs.
In addition,
it prevents the patient from seeing the needle.
Also it is easier to put the entry site down when it is posterior,
with recovery in a more comfortable supine position.
Patient collaboration is essential for the success of the procedure.
We instruct the patient to not move nor cough and to breathe freely,
but superficially.
During the procedure we encourage to avoid sudden respiratory movements and we teach the patient to hold his breath during the cannula inserction and prior to each shot of the needle.
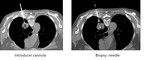
TECHNIQUE – THE PROCEDURE
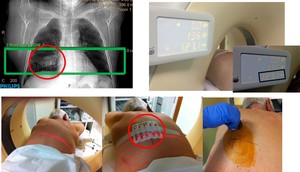
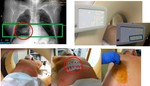
Fig. 3: 1-Noncontrast CT with a metallic grid pattern on the skin. 2-The needle entry site is determined using CT gantry lights and the grid on the patiens's skin. 3-Prepare the zone in a sterile fashion.
1.
Noncontrast CT images of 3 mm slice thickness are obtained with a metallic grid pattern on the skin above the region of interest to localize the target.
Extension is limited to the region of interest.
2. The pathway,
puncture site and the distance from de skin to the lesion and to the pleura are determined.
The route should be the shortest and safest technically feasible,
and the most vertical to the pleural surface as possible.
Avoid vessels in the chest wall,
mediastinum and large pulmonary vessels,
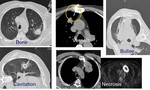
and visible bronchi and bullae.
Whenever possible,
do not traverse fissures. On cavitary lesions the needle must be aimed at the solid wall.
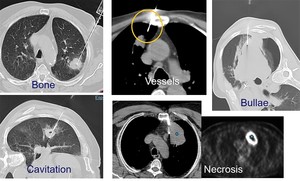
Fig. 4: The path should be addapted to avoid thoracic skeleton and vessels, bullae, cavitating or necrotic areas
3.
The needle entry site is determined using the CT gantry lights and the grid on the patient´s skin.
4.
Proper surgical skin asepsis technique.
5.
1% lidocaine hydrochloride is injected for local anesthesia through the chest wall without passing the pleura.
6.
A small stab skin incision is made to aid introduction of the needle in CNB.
7.
The needle is introduced into the lesion or its edge in a stepwise manner.
Consecutive CT images are performed as needed to check out the needle progression to the target.
If the needle is misaligned,
the introducer must be realigned without exiting the lung if possible (more feasible with larger needles for CNB than the thinner needles employed in FNA).
Once the tip has reached the lesion,
the samples are obtained.
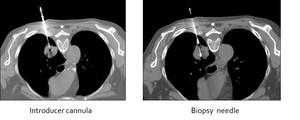
Fig. 5: Progressive images of CNB of a pulmonary nodule. Once the cannula is into the lesion or its margin (left picture), the biopsy gun is passed trough the canula (rigth picture). In semi-automathic mode you can see exactly the hole biopsy tract.
TECHNIQUE – NEEDLE GAUGE SELECTION
There is some controversy in the literature when comparing the diagnostic accuracy and complication rate of fine-needle aspiration (FNA) versus core-needle biopsy (CNB).
The decision to perform FNA or CNB depends on the location and size of the lesion,
presence of mild coagulopathy and the amount of tissue needed in each particular case. CNB allows to obtain larger samples for tumor architecture evaluation,
necessary for bening diagnosis.
Also,
although a diagnosis of malignancy can be easily establish on cellular material,
larger samples are often needed for inmunohistochemical and molecular testing.
We prefer to perform CNB with coaxial tecnique whenever possible,
since it allows multiple samples with a single pleural puncture.
Slight deviations of the cannula into the lesion permits to obtain samples from different parts of the lesion.
We preffer FNA in small nodules near to major vessels or next to the diaphragm or very peripheral location.
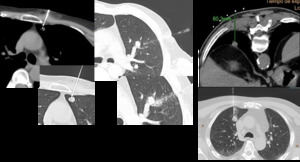
Fig. 6: FNAB indications
The technique for FNA and CNB are displayed in fig.7 and fig.8 respectively.
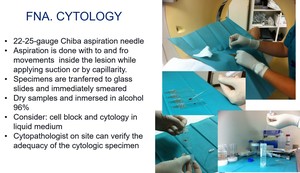
Fig. 7: Different steps during FNA
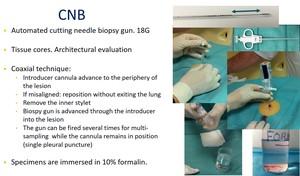
Fig. 8: Different steps during CNB
Attention: During coaxial tecnique,
remember the outer cannula should never be left inside the patient's chest without the inner stylet because an air embolism may occur if a small pulmonary vein branch is punctured.
The presence of the cannula without the stylet inside the pleural space is also an important risk factor for pneumothorax.
POST-PROCEDURE CARE
- Immediate postbiopsy CT: pneumothorax or pulmonary hemorrhage?
- Upright expiratory chest radiograph 1-4 hours after the biopsy,
to assess a delayed pneumotorax.
- Treatment of complications if needed.
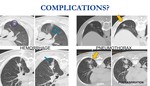
COMPLICATION
1.
PNEUMOTHORAX

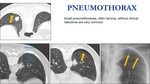
Fig. 9: Pneumothorax is the most frequent complication, usually with a laminar morphology and clinically asymptomatic. In this patient we observed a small pneumothorax without clinical relevance. Follow-up Chest X Ray demonstrated it stability.
Pneumothorax is the most common complication,
with reported rates extremely variable (8-69%). Ussually occur during or inmediately after the procedure,
but can be deferred.
Biopsy can still be done under stable pneumothorax when it appears during the procedure.
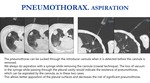
Small pneumothoraces can be aspirated through the introducer cannula in case of coaxial technique when it is detected prior to the cannula withdrawal.
The risk factors for the development of a pneumothorax:
- Small lesion size (<1cm)
- Increasing lesion depth
- Number of needle passes
- Increasing number of pleural surfaces crossed
- Underlying lung disease
Small,
asymptomatic pneumothoraces are treated conservatively.
Patient's vital signs are closely monitored and follow-up chest radiograph is recommended to evaluate the stability of the pneumotorax.
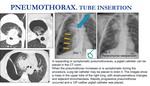
In expanding or symptomatic pneumotoraces,
a pigtail catheter can be placed in the CT room.
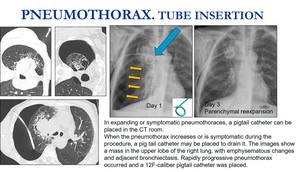
Fig. 11: Extensive pneumothorax that was sympthomatic. A pig tail catheter was placed in the CT room. Follow-up Chest X Ray showed a total reexpansion on day 3.
Several measures have been described to reduce the rate,
if not the incidence,
and the relevance of pneumothorax (meaning chest tube insertion):
- Autologous intraparenchymal blood patch
- Rapid roll over of the patient to the biopsy-side-down- position
- Self-expanding tract sealant device (BioSentryTM)
2.
PARENCHYMAL HEMORRHAGE AND HEMOPTYSIS.
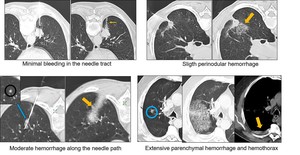
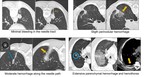
Fig. 12: Hemorrhage is one of the most frequent complication. It can be mild (in needle tract or perinodular), moderate and extensive, being mild asymptomatic hemorrhage the most common.
Parenchimal hemorrhage is the second most common complication,
and it may occur with or without haemoptysis.
Incidence 4-30%.
The cause of parenchymal hemorrhage is due to the proximity of small vessels to the lesion and the shock wave injury in the adjacent parenchyma to the needle during the firing of the device.
The risk factors to develop parenchymal hemorrahge are:
- Small lesion size.
- Increasing lesion depth.
- Emphysematous parenchyma.
- Pulmonary hypertension.
Usually this complication does not need any treatment.
When a larger parenchymal hemorrhage appears,
oxygen and pro-coagulative therapy may be needed.
Hemothorax is a more severe complication but it is extremely rare,
usually due to puncture of an intercostal or mammary vessel.
One patient in our series had a rapidly progressive hemothorax that needed urgent insertion of a coarse chest tube for drainage by the thoracic surgeon and a day of intensive care stay.
3.
OTHER COMPLICATIONS
Air embolism and tumour seeding are extremely rare complications with reported incidences of 0.061% and 0.012% respectively.
None were found in our series.
The mechanism of air embolism is:
- Placement of the needle tip within a pulmonary vein,
followed the entry of air into the needle.
- Placement of the needle through both a bronchus and an adjacent pulmonary vein,
creating a fistula.
OUR EXPERIENCE
From january 2013 to september 2017,
577 CT-guided lung biopsies were performed at our institution (395 males and 182 females),
with age from 29 to 89 years (average 65.23).
All biopsies were guided by a 16-slice CT-scanner.
FNA was performed in 117 cases,
CNB in 397,
and 63 procedures consisted in FNA plus CNB.
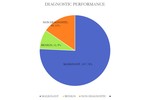
Diagnostic performance and complications are reflected in fig.13 and fig.14.
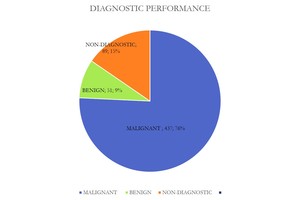
Fig. 13: Chart showing biopsy results.
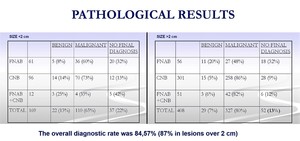
Fig. 14: Pathological results in nodules under 2 cm and over 2 cm.
Lesions were located: 154 in the right upper lobe; 140 in the right lower lobe; 33 in the middle lobe; 134 in the left upper lobe; 116 in the left lower lobe.
There were 169 lesions up to 2 cm,
and 408 larger than 2cm.
Overall diagnostic accuracy was 84.57% (78.1% for lesions ≤ 2cm and 87.25 for those over 2 cm),
with 15.42% of non-diagnostic procedures.
The most frequent malignant diagnosis in both lesion sizes considered was adenocarcinoma (206 cases,
47.13% of all malignancies),
followed by squamosus cell tumor in lesions over 2 cm,
and mestastasis in nodules under 2 cm.
Pneumothorax ocurred in 20.1% of all procedures (n 116),
usually laminar and limited to the area of pleural puncture.
25 patients required a thoracostomy tube placement (4.33%).
Any degree of hemorrhage was seen in 148 cases (25.64%),
most commonly asymptomatic as a perilesional opacity or along the needle path.
18 patients had self-limiting hemoptysis,
up to 50-100 ml en two cases.

Fig. 15: Types and percentages of complications in nodules under and over 2 cm.
Most of pneumothoraces were laminar and asympthomatic. Most of hemorrhages were mild and perinodular.
It can appear a high complication rate in our series.
Perhaps because any complication visualized in the immediate post biopsy CT scan is included,
regardless of the clinical relevance.
In addition,
we don´t refuse biopsy indication in basis of nodule size,
deep location,
ground-glass appearence,
presence of moderate emphysema or other considered risk factors when the expected benifit justifies it.
We should wonder whether any amount of gas in the pleural space or blood in the parenchyma should be considered a complication.
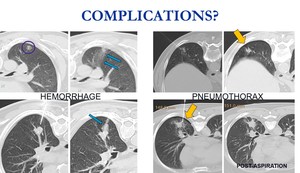
Fig. 16: Minimal pneumothorax at the site of puncture, without clinical significance, are common. Also small bleeding along the needle tract or around the lesion. Should it be considered a complication or a common side efect?