The pattern and distribution of blooming signal loss is a good differentiator for several pathologies on SWI.
This poster will explore this differentiation by examining a range of conditions incorporating vascular,
infectious,
inflammatory,
neoplastic,
neurodegenerative and traumatic pathologies.
In this section we will explore the clinical applications of SWI with a particular focus on differentiating features for various pathologies.
Case contents:
Vascular
Hypertensive angiopathy (Fig. 4 )
Venous Sinus Thrombosis ( Fig. 5 )
Cavernous malformation ( Fig. 6 )
Telangectasia
Delopmental venous anomaly
Superficial Siderosis
Stroke
Infectious
Abscess ( Fig. 7 )
Neurocysticercosis
Inflammatory
MS
Neoplastic
Primary
Traumatic
Traumatic brain injury ( Fig. 8 , Fig. 9 )
Neurodegenerative
Cerebral Amyloid Angiopathy ( Fig. 10 , Fig. 11 )
Alzheimer’s Disease,
Parkinson’s Disease,
Huntington’s Disease,
Amyotrophic Lateral Slerosis
Miscellaneous
Drug-induced ( Fig. 12 )
Wilson’s Disease ( Fig. 13 )
Neurodegeneration with brain iron accumulation ( Fig. 14 )
Fat emboli post tibio-fibular fracture ( Fig. 15 )
Multiple emboli associated with congenital cardiac disease ( Fig. 16 )
VASCULAR - Hypertensive microangiopathy
- Hypertensive microangiopathy (also known as chronic hypertensive encephalopathy) is the term given to parenchyma changes in the brain caused by the longstanding effects of untreated/treatment resistant systemic hypertension.
- Chronic hypertension is thought to cause irreversible structural changes in the walls of small sized arteries in the brain parenchyma leading to lacunar infarcts as well as changes in the white matter.
- It is also associated with the deposition of hyaline within small arteries with subsequent predisposition to thrombotic occlusion.
- Hypertensive microangiopathy is a well-recognised and important cause of cognitive deficits attributed to vascular disease (vascular dementia).
- General findings include:
o Diffuse white matter lesions (hypoattenuating on CT,
hyperintense on T2WI)
o Microhaemorrhages
- Other imaging features:
o Lacunae (basal ganglia,
thalami,
brainstem)
o Grey matter lesions (basal ganglia,
thalami,
brainstem)
- SWI differentiators:
o Type – blooming signal loss due to cerebral
microhaemorrhage
§ Histologically correspond with sites of
haemosiderin containing macrophages in the
perivascular spaces of associated vessels.
o Distribution –
§ Basal ganglia,
thalami,
pons,
cerebellum
§ Subcortical white matter,
especially of the
posterior brain regions
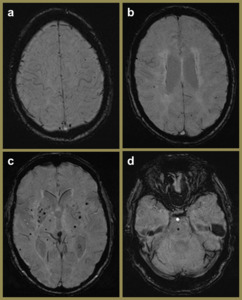
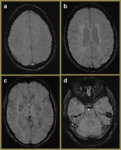
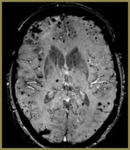
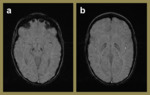
Fig. 4: SWI images of a 57-year-old female demonstrating hypertensive angiopathy. Note the preference of the foci of blooming signal loss for the basal ganglia (c) and pons (d) relative to the superficial (a) and periventricular (b) regions.
VASCULAR - Cerebral venous sinus thrombosis
- Cerebral venous sinus thrombosis is an infrequent pathology affecting both adult and paediatric populations.
- Diagnosis is challenging due to the non-specific nature of the presenting clinical findings.
- In addition,
primary signs related to the thrombus,
such as the “empty delta” sign on CT and loss of the normal flow voids on conventional MR sequences can be subtle findings and normally require active seeking by the radiologist to be identified.
- The thrombus causes venous hypertension with associated engorgement of the cerebral veins and can cause venous ischaemia and infarction.
- General findings include:
o "Empty delta" sign (on CECT,
T1WI C+ MR)
o Hyperdense sinus on NECT
o Filling defect in the affected dural sinus(es)/cerebral vein(s)
- Other imaging features:
o Hypointense thrombus "blooms" on T2* GRE
o Absence of flow in occluded vein/sinus on 2D TOF MRV
- SWI differentiators:
o Type –
§ Hypointense SWI signal due to increased
deoxyhaemoglobin concentration in the involved veins
(venous stasis and slow collateral flow).
§ Parenchymal or extra-axial haemorrhage secondary to
venous infarction.
o Distribution –
§ Dependent on the site/extent of the thrombosis.
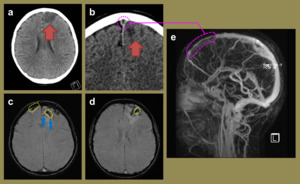
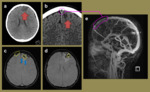
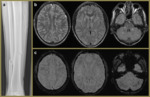
Fig. 5: Imaging relates to a 16-year-old female with a history of acute lymphoblastic leukaemia who presented with a new seizure. The initial unenhanced CT head (a) demonstrated focal hypoattenuation and oedema in left frontal lobe (red arrows) which was also present on the subsequent contrast enhanced CT (b). This also demonstrated a filling defect in the superior sagittal sinus anteriorly (pink dashed circle) consistent with a dural sinus thrombosis with secondary venous ischaemia. The SWI MR images (c, d) highlight the engorged frontal cortical veins (yellow dashed circles) as well as a couple of foci of blooming loss, consistent with venous cerebral microhaemorrhages (blue arrows). The reconstructed MRV image (e) illustrates the confinement of the extent of the thrombosis to the anterior superior sagittal sinus.
VASCULAR – Cavernous malformation
- Cavernous malformations are common cerebral vascular malformations which are also known as cavernomata or cavernous haemangiomata.
- Most are asymptomatic and incidental findings,
however haemorrhage of a lesion can cause focal neurological deficits or seizures.
- A familial form exists with a greater number of lesions in patients and thus associated with a higher annual risk of associated haemorrhage.
- The lesions represent benign vascular hamartomas with intralesional haemorrhage and contain no neural tissue.
- Can exhibit variable dynamic behaviours including enlargement,
regression and de novo formation.
- General findings include:
o “Popcorn ball” or “berry” appearance with a complete
hypointense haemosiderin rim on T2WI.
o Acute haemorrhage with non-specific features and associated
oedema
- Other imaging features:
o Associated with developmental venous anomalies
- SWI differentiators:
o Type –
§ Hypointense “blooming” due to prominent susceptibility
effect
§ Multiple “black dots” (punctate hypointense foci) due to
multiple lesions
o Distribution –
§ Cerebral hemispheres more commonly involved than the
brainstem or cerebellum.
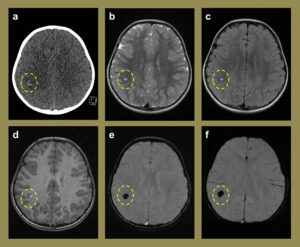
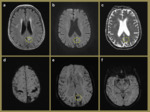
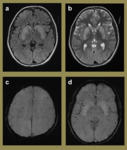
Fig. 6: Imaging relating to a 3-year-old girl who presented with seizures that were clinically thought to be due to febrile convulsions secondary to tonsillitis. The initial CT (a) demonstrated a small right inferior parietal hyperdense lesion. The subsequent MR study performed demonstrated imaging characteristics on the T2 (b), FLAIR (c) and T1 (d) sequences that were consistent with a cerebral cavernous haemangioma. The SWI (e) and mIP (f) sequences confirmed the isolated nature of the lesion with no further cavernomata identified.
VASCULAR – Telangiectasia
- Capillary telangiectasias are less common than cavernous malformations and are usually smaller in size.
- Occurrence may be sporadic,
associated with syndromes (such as hereditary haemorrhagic telangiectasia) or resultant from endothelial injury such as post radiotherapy.
- May be asymptomatic but mild neurological symptoms do occur which would bear no findings on conventional MR imaging.
- Appear on SWI as “black dots” or blooming signal loss.
VASCULAR – Developmental venous anomalies
- Developmental venous anomalies are similar to cavernous malformations and telangiectasias in that they are lesions containing low velocity and multidirectional flow.
- These features attribute to their reduced conspicuity on conventional MR techniques,
compared to high-flow arterio-venous malformations.
- They also share another feature in their potential to lead to haemorrhage.
- The incorporation of phase information in SWI results in improved sensitivity in detecting such vascular lesions.
VASCULAR – Superficial Siderosis
- T2WI and FLAIR sequences are the currently most utilised mode of detecting haemosiderin deposition in the subpial regions in cases of suspected superficial siderosis syndrome.
- Despite its conspicuity on T2,
FLAIR and T2* sequences,
the haemosiderin deposition has been shown to be more readily detected on SWI suggesting an increased sensitivity.
VASCULAR – Stroke
- Many modalities are utilised in the investigation of acute thrombotic stroke including CT,
CT perfusion,
and multiple MRI sequences.
- SWI may add value to MR protocols in more ways than one:
o Detection of intra-arterial thrombus
o Small volume haemorrhagic transformations (not visible on CT)
INFECTION - Cerebral abscess
- The role of SWI imaging in the imaging of cerebral infection is not established.
- Some have suggested its ability to detect calcification in atypical infections such as neurocysticercosis or those caused by fungal pathogens.
However,
calcification may be detected on CT imaging.
- One potential role for SWI is to aid in the differentiation of pyogenic brain abscess from necrotic glioblastoma,
where the utility of DWI is less valuable than usual.
- Toh et al.
compared several morphological differences between T2WI and SWI and concluded that SWI can aid differentiation between the above mentioned pathologies.
- The “dual rim sign” was described by them as the most specific imaging feature to utilise.
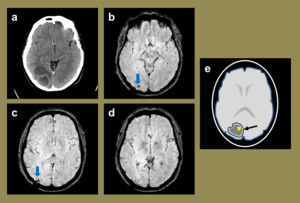
Fig. 7: A case of a 41-year-old intravenous drug user who’s initial post contrast CT head (a) demonstrated a right occipital rim enhancing lesion, and given the clinical history, a diagnosis of a cerebral abscess was made. This was surgically drained and post procedure MR SWI images (b-d) demonstrate a few foci of blooming signal loss (blue arrows) at the prior site of the abscess which are likely to represent small iatrogenic haemorrhagic foci. The illustration represents the expected SWI appearances of the cerebral abscess, had it been imaged prior to removal (e), highlighting the ‘dual rim sign’. This sign describes the presence of two rims bordering the abscess, with the inner rim being hyper-intense (yellow arrowhead), and the outer one being hypo-intense relative to the abscess.
References: Leeds Teaching Hospitals NHS Trust - Leeds/UK, H. Nejadhamzeeigilani
INFLAMMATORY – Multiple sclerosis (MS)
- SWI is able to depict small veins more readily and thus enables the observation of the perivenous MS plaque deposition.
- In addition,
intralesion iron deposition,
depicted by low signal foci on SWI may indicate chronicity and offer more accurate assessment of the plaque burden and potentially of monitoring disease activity.
NEOPLASTIC – Primary CNS malignancy
- Widespread susceptibility effects within a lesion are suggestive of vascular correlation,
with several studies reporting correlation with high-grade lesions.
- Careful consideration should be given to the appearances of calcification,
and phase sequences must be utilised in order to distinguish this feature which is more of a low-grade lesion.
- Lymphomas rarely show haemorrhage and thus would give differing SWI appearances to diffusely infiltrative glioblastomas.
TRAUMA - Diffuse axonal injury/traumatic brain injury
- Traumatic brain injury is a leading cause of death and disability in children and young adults.
- It is a consequence of sudden deceleration and abrupt changes in angular momentum in a closed head injury.
- Due to the different densities of the grey and white matter,
these structures rotate at different speeds in a closed head injury,
leading to axonal stretching (and possible shearing injury).
- Adams and Gennarelli system used for staging of severity.
Increased severity of traumatic forces correlate with deeper brain involvement.
- SWI enables the full extent of diffuse axonal injury to be revealed due to its high sensitivity for detection of small amounts of haemorrhage.
- Several studies have revealed its greater sensitivity over T2*WI in this realm.
- General findings include:
o Microhaemorrhages
o Punctate lesions at corticomedullary junction,
deep grey
matter,
brainstem and corpus callosum.
- SWI differentiators:
o Type –
§ Multiple “black dots” (punctate hypointense foci) due to
multiple microhaemorrhages
o Distribution –
§ Grey-white matter junction (in particular
frontotemporal lobes)
§ Corpus callosum (particularly the splenium)
§ Brainstem (dorsolateral midbrain and upper pons in
particular)

Fig. 8: Adams and Gennarelli severity grading system for traumatic brain injury
References: H. Nejadhamzeeigilani
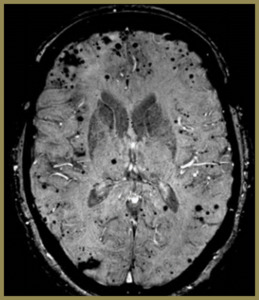
Fig. 9: SWI image of a patient who presented with trauma which demonstrates blooming signal loss which predominates at the grey white matter junction in keeping with diffuse axonal injury.
NEURODEGENERATIVE - Cerebral amyloid angiopathy
- Sporadic cerebral amyloid angiopathy is a common cerebrovascular condition of the elderly.
- Characterised by the deposition of the cerebral amyloid-β within the walls of small and medium sized cortical and leptomeningeal vessels.
- This is thought to lead to increased fragility of the vessel walls leading to haemorrhages.
- Most likely has a significant role in the development of dementia and Alzheimer’s disease.
- Histological diagnosis dependent on biopsy (rarely performed) or post mortem examination.
- Modified Boston criteria used as a tool to aid and standardise diagnosis.
- General findings include:
o Dementia in a normotensive patient (2/3 normotensive,
1/3 hypertensive)
o Lobar haemorrhages (of varying ages)
o Cerebral microhaemorrhages
- Other imaging features:
o Pial siderosis
o Occipital dominant white matter hyper intensities on MR
- SWI differentiators:
o Type – blooming signal loss due to cerebral
microhaemorrhage
§ Histologically correspond with sites of haemosiderin
containing macrophages in the perivascular spaces of
associated vessels.
o Distribution –
§ Cortex and subcortical white matter
§ Usually involve the frontal and parietal lobes
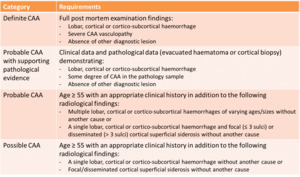
Fig. 10: Modified Boston criteria for the diagnosis of CAA
References: H. Nejadhamzeeigilani
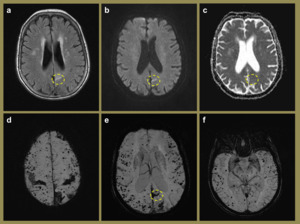
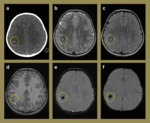
Fig. 11: MR imaging of a 72-year-old male. Linear hyper intensity (yellow dashed circle) in the left parafalcine parietal region, apparent on the FLAIR (a) and B1000 (b) axial images with no associated ADC abnormality (c). SWI images (d-f) of the same patient demonstrate diffuse cortical and subcortical blooming artefacts, predominantly involving the frontal and parietal lobes with relative sparing of the basal ganglia and thalami. Appearances are consistent with probable CAA.
NEURODEGENERATIVE – others
- Pathological variations in the iron content within the brain have been previously demonstrated in histopathology samples of numerous neurodegenerative conditions.
- Some of these include Alzheimer’s disease,
Parkinson’s disease,
Huntington’s disease and amyotrophic lateral sclerosis.
- Unfortunately,
the lack of normal quantification methods and ranges for iron content in healthy individuals on SWI imaging can make its utility here.
- However,
given its high sensitivity and the increasing incidence of dementia,
in particular Alzheimer’s,
a future role for SWI in the assessment of neurodegenerative conditions seems very possible.
MISCELLANEOUS – Drug-induced
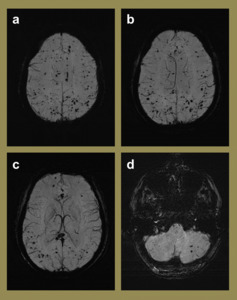
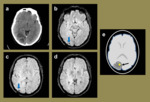
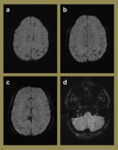
Fig. 12: Imaging of a 31-year-old male with an extensive prior history of recreational drug use who presented with increasing frequency of seizures. mIP SWI images (a-d) demonstrated innumerable supra- and infra-tentorial microhaemorrhages in the brain parenchyma and given the clinical history, were felt to be most likely due to drug toxicity. Given a different clinical history, other considerations for such widespread microhaemorrhages in this age group should include vasculitis and numerous cavernomata.
MISCELLANEOUS – Wilson’s disease
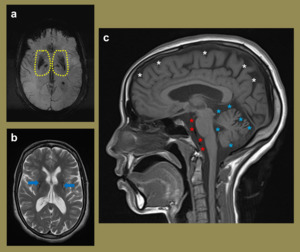
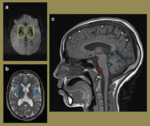
Fig. 13: 27-year-old female with biochemical findings in keeping with Wilson’s. The SWI mIP image (a) demonstrates increased susceptibility in the putamena and globus pallidi. Note the perivascular spaces on the axial FLAIR image (b). The sagittal T1 image (c) illustrates Increased conspicuity of the convexity sulci (white asterisks) in keeping with cerebral atrophy with more significant atrophy involving the cerebrellum as evident with the increased surrounding CSF spaces (blue and red asterisks).
MISCELLANEOUS – Neurodegeneration with brain iron accumulation
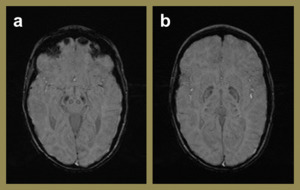
Fig. 14: SWI images (a-b)of a 6 year old female with global delay and neurological regression following a febrile illness at 10 months. Increased signal dropout within the globus pallidi, subthalamic nuclei and substantia nigra are more than one would expect for this age group. This is likely due to increased iron accumulation in these regions.
MISCELLANEOUS – Fat emboli post tibio-fibular fracture
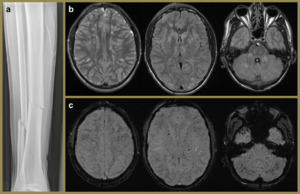
Fig. 15: 22-year-old male presenting following a tibio-fibular fracture (a) complicated by fat embolism. T2 and FLAIR axial images demonstrate multifocal hyper intensities, involving the deep white matter, basal ganglia and cerebellar hemispheres. SWI images show extensive blooming artefacts.
MISCELLANEOUS – Multiple emboli associated with congenital cardiac disease
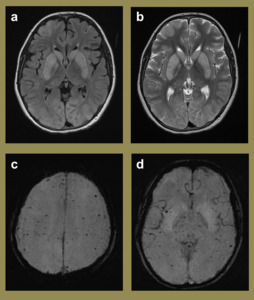
Fig. 16: Axial FLAIR (a) and T2 (b) images of a 9 year old female, following cardiac bypass surgery, highlight striking abnormal hyper intensities involving both caudate and lentiform nuclei. Given the history of CPR, these are likely to represent normalisation following recent ischaemic insult. In addition, the SWI mIP images (c-d) demonstrate multifocal areas of parenchymal microhaemorrhage. This phenomenon is often seen in patients with a significant congenital cardiac history and is thought to be embolic.